Abstract
The space environment is regularly used for experiments addressing astrobiology research goals. The specific conditions prevailing in Earth orbit and beyond, notably the radiative environment (photons and energetic particles) and the possibility to conduct long-duration measurements, have been the main motivations for developing experimental concepts to expose chemical or biological samples to outer space, or to use the reentry of a spacecraft on Earth to simulate the fall of a meteorite. This paper represents an overview of past and current research in astrobiology conducted in Earth orbit and beyond, with a special focus on ESA missions such as Biopan, STONE (on Russian FOTON capsules) and EXPOSE facilities (outside the International Space Station). The future of exposure platforms is discussed, notably how they can be improved for better science return, and how to incorporate the use of small satellites such as those built in cubesat format.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
Science experiments designed to benefit from the unique conditions provided in situ by the space environment began almost at the same time as the conquest of space in the late 1950s. When the word “exobiology” was coined by J. Lederberg in 1960 (Lederberg 1960), at a time when the search for life beyond Earth started to settle on the scientific foundation that prevails today (Cottin et al. 2015a), microorganisms were intentionally placed in space as part of the scientific payloads of Sputniks, Vostoks and Gemini spacecraft, principally to study the effects of microgravity (Taylor et al. 1974). The very first exposure of microorganisms to space radiation, proving that life could survive the extremely harsh conditions of open space, were conducted on sounding rockets in 1965 (150 km) (Hotchin et al. 1967), extended to the Gemini 9 and 12 missions in 1966 (300 km) (Hotchin et al. 1968) and finally as the Apollo 16 mission was flying back to Earth from the Moon (Taylor et al. 1974).
After Apollo 16, space was used episodically as a tool for astrobiology in the 1980s (Long Duration Exposure Facility—LDEF) and in the early 1990s (EUropean REtrievable CArrier—EURECA). In parallel with the increasing number of organic molecules detected in the interstellar medium and better understanding of the chemical complexity of carbonaceous chondrites, comets, and planetary environments such as the atmosphere of Titan, the number of experiments addressing chemistry with an astrobiological perspective increased. It is now quite common to have both astrochemistry and biology experiments on a given facility. With the Biopan, STONE, and now the EXPOSE facilities on the International Space Station (ISS), the European Space Agency (ESA) has shown sustained interest since the mid-1990s in granting its science community regular access to the space environment. Two main scientific questions related to astrobiology motivate the experiments supported by those facilities:
-
What does the resistance of microorganisms to space conditions tell us about the possibility to find life beyond Earth and what can we learn from space effects on microbes that is pertinent to planetary protection?
-
How was the chemistry leading to the origin of life on Earth influenced by processes in space?
-
What can we learn from these types of experiments to support future exploration missions?
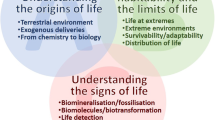
Scientists involved in these types of complex and costly projects are often asked why space access is necessary for such studies, what are the benefits, and why a more classical laboratory approach is not preferred. In 2011 ESA funded a topical team on astrobiology to address those questions. The team was asked to produce an update about the recent profound achievements and transformations in the field of astrobiology that have occurred in the last years (Cottin et al. 2015a) and to focus specifically on experimental studies either in the field (i.e. using Earth as a tool for astrobiology) or in space, (i.e. using space as a tool for astrobiology). The present paper, an interdisciplinary review of the latter subject, reports on the history of some 60 years of research for astrobiology in space, focusing on the most recent developments and results. It shows that the field is active and that many exciting new experiments are planned in alignment with the current expectations of the science community. To meet modern science requirements while guaranteeing added value similar to that obtained from ground-based experimentation, passive exposure facilities in space will have to be progressively modified to support more complex, active experiments including real-time measurements. Orbits will have to be changed to provide larger doses of multiple types of energetic particles, which cannot be provided in combination with solar simulators in Earth laboratories.
This paper describes the benefits of the use of space radiation environment and research in field of astrobiology conducted in space. Experimental space facilities, hardware, and samples are presented and discussed. It must be noted that the organization of the paper is such that first the exposure platforms common for both chemistry and biology are presented, and then two distinct main chapters are dedicated to a selected number of specific experiments related respectively to chemistry and biology, using sometimes the common facilities presented in the first section. We have tried to give sufficient context information in each chapter to provide independent self-consistent sections. Then, in both in the chemistry and biology chapters, details about the experiment hardware and results for each experiment are presented. A perspective for future developments in this research field is proposed and recommendations from the Topical Team to ESA are presented.
2 The Space Radiation Environment
By definition, “outer space” (or more simply “space”) begins 100 km above the Earth’s sea level, at the so called Kármán line. At this altitude, the Earth atmosphere is so thin that the speed required for a plane to fly would equal or exceed orbital velocity. This is therefore the boundary between aeronautics and astronautics, as accepted by the Fédération Aéronautique Internationale. However, there is no strict physical boundary between Earth’s atmosphere and space, since the atmosphere extends well beyond the 100 km limit. Figure 1 shows that the residual Earth atmosphere stretches above this arbitrary boundary and that the International Space Station and other artificial satellites orbiting the Earth at the lower levels of altitudes, referred to as low Earth orbit (LEO, ∼400 km), are in an environment dominated by O radicals (NASA 1976). Total pressure at 400 km is about \(10^{-8}\) mbar (\(10^{-6}\) Pa); it is \(\sim10^{-10}\) mbar (\(10^{-8}\) Pa) at 1000 km.
Composition of the Earth’s atmosphere from 100 to 700 km (data from NASA 1976). The average altitude of the International Space Station is shown with the horizontal red line
Outer space provides a challenging environment with regard to temperature: in absence of appropriate system design, thermal extremes far exceeding Earth’s hottest and coldest locales are to be expected. In general, the temperature to which a biological sample is exposed in space is a strong function of the design of its exposure and containment system: insulation, emissivity, duration of exposure to direct or indirect sunlight and eclipse (determined in part by the orbit), provision of active heating and/or cooling, strength of thermal linkages to a parent structure or spacecraft, and so forth. In space-exposure experiments, temperature is a parameter to be managed by careful design.
Terrestrial orbits are described by their altitude (from sea level) and inclination (tilt of the orbital plane relative to Earth’s equator) (Stark and Swinerd 2003). Defining a non-circular orbit also requires knowledge of perigee and apogee, the respective points of closest and furthest distance from the Earth. Table 1 provides a number of parameters that summarize Earth orbits and other locations at which inhabited space stations and satellites have been deployed, or are likely in the coming decade to voyage, in order to provide a particular set of environmental conditions for a given science experiment. This table will also be discussed in Sect. 2.2 since it includes data related to particle radiation sources and doses.
Two physical phenomena that differ significantly in outer space relative to Earth’s surface, often with great relevance for biological, astrobiological, and astrochemical studies, are gravitation and radiation. The gravity field decreases slowly from sea level to classical orbits around the Earth. It is a common misunderstanding to believe that objects and astronauts in Earth orbit are weightless because they have escaped Earth gravity. Gravity caused by our planet is still some \(8.6~\mbox{m}\,\mbox{s}^{-2}\) at 400 km altitude and \(7.3~\mbox{m}\,\mbox{s}^{-2}\) at 1000 km. Without that strong pull from the Earth, the ISS would not remain in orbit and wander away into space. Weightlessness in Earth orbit is a consequence of the fact that such objects are freely falling within a gravity environment. Instead of weightlessness the word microgravity is often used. This leads to even more confusion because it has nothing to do with micro (\(= 10^{-6}\)), neither with a reduction of gravity. It is more appropriate to call it weightlessness or (more accurately) near-weightlessness (see also van Loon 2007).
The space radiation environment can be divided into two main categories: photons emitted by the Sun, and energetic particles from the solar wind and galactic cosmic rays. Photons and energetic particles are progressively filtered through Earth’s atmospheric layers. Photons are not subject to significant variations or concentration processes due to interaction with Earth’s electromagnetic field; they are only filtered once they enter the atmosphere. In the following sections, the space radiation environment will be described in detail, and we will address the relevance of conducting experiments in space as a complement to ground laboratory investigations.
2.1 Photons
Electromagnetic radiation from the Sun is divided into spectral categories ranging from gamma rays to radio waves (Table 2). At 1 astronomical unit (AU), i.e. in Earth’s vicinity, the total solar irradiance (TSI) is equal to \(1.361~\mbox{kW}\,\mbox{m}^{-2}\) (measured during the 2008 solar minimum period). This energetic input at the top of the atmosphere is made up of approximately 46% IR radiation, 46% visible light, and only 8% UV light (calculated from Thuillier et al. 2004b (Fig. 2)). The amplitude of variation in the monthly average value of TSI over an 11-year solar cycle is about \(1.6~\mbox{W}\,\mbox{m}^{-2}\) (\(0.12\%\)), with rapid fluctuations on the time scales of days or weeks superimposed that can reach \(4.6~\mbox{W}\,\mbox{m}^{-2}\) (\(0.34\%\)) (Kopp and Lean 2011). These variations may appear rather small and negligible in the context of astrobiology studies (photochemistry of organic molecules, resistance of microorganisms to damage or death), but irradiance variations are not uniformly distributed over the whole electromagnetic spectrum: they are especially intense in the VUV domain, where photolytic processes are predominant. For instance, Lyman \(\alpha\) daily average values can vary from \(6 \mbox{ to } 11~\mbox{mW}\,\mbox{m}^{-2}\,\mbox{nm}^{-1}\) over one solar cycle (\(+83\%\)), while they vary from \(7.3 \mbox{ to } 8.0~\mbox{mW}\,\mbox{m}^{-2}\,\mbox{nm}^{-1}\) (\(+10\% \)) at 200 nm over the same period (Fig. 3). Important variations are also measured over the 27-day rotation period of the Sun: during a solar maximum, they can reach \(+30\%\) at Lyman \(\alpha\), \(+5\%\) at 200 nm, and are much less variable at longer wavelengths (DeWolfe et al. 2010; Rottman et al. 2006).
Irradiance spectrum of the Sun from 100 to 2500 nm. (From the Sorce web database, http://lasp.colorado.edu/lisird/sorce/)
Time series for Lyman \(\alpha\) and 200 nm daily averaged irradiance between 2003 and 2013, measured with the SOLSTICE instrument on the SORCE spacecraft (Pankratz et al. 2005; Rottman 2005). The high frequency modulation is due to the 27 days solar rotation period. (From the Sorce web database, http://lasp.colorado.edu/lisird/sorce/)
In addition to contemporary fluctuations, it must be noted that the overall shape of the emission spectrum of the Sun has changed since its formation. This behavior is important and has to be taken into account to address astrochemistry and astrobiology questions in the early Solar System (Güdel and Kasting 2011). The overall luminosity of the Sun has been steadily increasing and it is now established that our star is 30% brighter today than when it entered its main sequence of evolution 4.5 billion years ago (Gough 1981). However, this global trend is driven by an increase within the visible and infrared regions, while the XUV and VUV emission significantly decreased since 4.5 billion years: by a \(\mbox{factor} > 1000\) at 0.1 nm, 100 at 10 nm, 10 at Lyman \(\lambda\) (Claire et al. 2012).
2.2 Radiation Other than Photons
The non-electromagnetic radiation environment in interstellar space is dominated by galactic cosmic rays (GCRs), which mainly consist of protons (95%) and helium (4%). Only a small fraction (about 1%) is contributed by heavier nuclei, electrons and positrons (Ferrari and Szuszkiewicz 2009; Gaisser 1990; Mewaldt 1996). Since these particles travel with velocities close to the speed of light, their energies range from a few million to \(10^{20}\) electron volts (Hörandel 2010; Newell and Naugle 1960). Their distribution is approximately isotropic (Fig. 4) and their flux is stable over time. The elemental composition of GCRs is very similar to the naturally occurring (hydrogen–uranium) elemental abundances in our Solar System. Most GCRs reaching our Solar System originate from supernovae remnants within the Milky Way Galaxy; however, research into other sources and acceleration mechanisms of GCRs is still an active field of astrophysics (Pasquale 2010).
In our Solar System, particles ejected by the Sun also contribute to observed background radiation levels. These solar particle events (SPEs), with particle energies up to hundreds of MeV (Potgieter 2010), are caused by eruptions on the Sun’s surface either due to Sun spot activity or coronal mass ejections and therefore depend on the local variations in solar magnetic activity. Solar activity fluctuates with well-known periodicities and is expected to reach high levels in the years 2020–2040 (Norbury 2011) after having gone through a minimum between 2006–2011 (Koshiishi and Matsumoto 2013). Changes in the magnetic field of the Sun also affect trajectories of the lower energy GCRs and therefore impact GCR radiation levels in solar proximity (including the vicinity of Earth), in a manner that is anti-cyclical to solar activity (Ferrari and Szuszkiewicz 2009). Neutral atoms with origins in the interstellar medium and Jovian electrons contribute only to the lower energy spectrum (up to 30 MeV) of particle energies found within 10 AU from the Sun (Potgieter 2010).
The critical difference among the various possible Earth orbits, and other locales, for most biological and many astrobiological space experiments, is the radiation environment (Fig. 5). In LEO the Earth’s magnetosphere provides substantial shielding from high-energy charged particles, including a majority of the charged-particle flux from SEPs, and to a lesser extent GCRs, as per the local geomagnetic cutoff rigidity. The geomagnetic cutoff rigidity (a particle’s momentum:charge ratio) “specifies the minimum rigidity a charged particle must possess to enter a specific position in the geomagnetic field from a specified direction” (Smart and Shea 1985). Magnetospheric shielding also exists to a lesser extent in higher altitude orbits, but reaches essentially zero at polar latitudes. The magnetosphere extends to varying distances relative to Earth, with strong influence by the solar wind: on the sun-facing side of Earth, it extends about 65,000 km into space, whereas on the night-facing side of Earth—in the “wake” of the solar wind—the magnetosphere extends some 6.3 million km (McElroy 2012).
The second set of phenomena that vary according to orbital location and strongly influence a space experiment’s radiation environment are the regions of magnetically trapped radiation known as the Van Allen Belts. The Outer Van Allen Belt, approximately formed as a D-shaped cross-sectional toroid, extends from 13,000–60,000 km above Earth; it is comprised mainly of 0.1–10 MeV trapped electrons. The Inner Van Allen Belt forms a more C-shaped cross-sectional toroid located principally from 1000–6000 km above Earth’s surface at equatorial latitudes, and is comprised of significant numbers of mainly 0.1–450 MeV protons, as well as electrons with energies measuring tenths of one MeV. In a region known as the South Atlantic Anomaly (SAA), the Inner Van Allen Belt reaches to within just 200 km of Earth off the coast of Brazil; this region features the most significant localized weakness in Earth’s magnetic field, and is due to the tilt between the Earth’s magnetic and rotational axes. The SAA presents significant radiation dose rates to spacecraft even in low-inclination LEO (\(\sim0\mbox{--}50^{\circ}\)) at altitudes of a few hundred km, but only for that portion of each orbit spent above the southern Atlantic Ocean and adjacent regions of South America (Fung 1996). At the altitude of the ISS (330–435 km), the dose rate is around \(12~\upmu\mbox{Gy}\,\mbox{h}^{-1}\) in the SAA.
The distances cited above for the extents of the two Van Allen Belts apply near the equator; near arctic and Antarctic latitudes, the belts approach Earth closely. Thus, high-inclination LEO (Table 1) can provide a much higher radiation ambient than low-inclination LEO due to orbital paths crossing through regions of trapped radiation in one or both belts, and due to the magnetic field arrangement, which funnels both trapped and magnetically interacting particles towards the polar regions (Walt 2005).
The final and most important aspect of the orbital environment summarized in Table 1 is the typical anticipated monthly radiation dose, reported here when the experiment is separated from the vacuum of space by either 1 mm or 5 mm of aluminum (or other materials with equivalent stopping power). The former thickness is chosen as a practical minimum shielding, particularly if samples are to be maintained at 1 bar behind a window or cover, in order to maximize radiation exposure, as discussed further below. The latter thickness represents moderate shielding that does not add excessive mass, and can be used to decrease radiation damage to electronic components in a small spacecraft in which every gram of mass competes with the experimental capacity of the payload. By comparing the two shielding thicknesses in Table 1, it should become apparent that the efficacy of shielding vs. thickness depends strongly on the energy spectrum of the radiation: at one extreme, in LEO at the orbit of the ISS, 4 mm of additional Al decreases the effective dose 80-fold, because the typical energies of trapped electrons and protons in that environment allow most of them to be effectively blocked. At the other extreme, in interplanetary space, there can be as little as a 6-fold decrease when shielding thickness is quintupled because the high-energy particles of SEPs and GCRs are much more penetrating.
When particles with high energies interact with Earth’s atmosphere (mainly with nitrogen and oxygen atoms), secondary particles are produced. These so called “air showers” are typically produced at ∼20 km altitude. Depending on the initial energy of the arriving particle, cascades of hadrons such as protons, neutrons and pions are formed. Pions decay and form muons, neutrinos and their respective antiparticles (Ferrari and Szuszkiewicz 2009). These particles eventually reach the Earth’s surface and add significantly to the natural background radiation at sea level (around \(10\mbox{--}80~\mbox{nGy}\,\mbox{h}^{-1}\)). Protons and electrons trapped within the magnetic field of the Earth increase radiation levels predominantly at the poles and can be seen as the Northern Lights due to emitted synchrotron radiation by these particles. The origin of these particles is partly solar and partly decay products from high-energy galactic cosmic rays.
Knowing the radiation environment in Earth orbit and beyond is not only important with respect to radiation protection for human space flight missions but also for the design of space hardware. The level of radiation protection required to fulfill radiation protection requirements determines the cost and design of a space mission. Radiation levels in LEO are well known due to direct measurements (Reitz et al. 2005) and can be reasonably well predicted by computer simulations (Gustafsson et al. 2009). The radiation environment in higher altitude orbits, interplanetary space, and near other planets or moons is less well characterized. Additional data from space probes and planetary missions (Hassler et al. 2012, 2014) are providing better understanding of the radiation protection measures required for future missions, thereby enabling scientists and engineers to develop space hardware and experiments to further investigate the effects of ionizing radiation in space.
2.3 Space Environment Versus Laboratory Environment
A large number of experimental programs are conducted to study the photostability of solid and gaseous organic compounds in the laboratory. As useful as they are, such experiments have limitations. For example, it is extremely difficult to simulate the whole spectrum of wavelengths corresponding to the most energetic part of solar radiation in the VUV range (Fig. 6), although recent progress in laboratories has been achieved (Chen et al. 2014; Cook et al. 2014; Es-sebbar et al. 2015). Discrepancies between the actual emission from the Sun and simulators can result in important differences between kinetics measurements related to photochemistry measured directly in space and similar experiments conducted in “classical” ground laboratories and then extrapolated to space conditions (Guan et al. 2010). For specific conditions, such as the simulation of the surface of Mars, where photons below 190 nm have been filtered by the atmosphere (Cockell et al. 2000), deuterium discharge or high pressure xenon UV lamps are able to reproduce quite faithfully the Sun emission continuum above 200 nm (Poch et al. 2013; Ten Kate et al. 2005). However, these simulations do not take into account simultaneous UV radiation, temperature variations, the solar wind, and cosmic rays. The laboratory experimental simulations are then advantageously complemented with in situ space experiments in order to evaluate to which extent they reflect the reality of the space environment.
Comparison of the solar spectrum between 100 and 200 nm (from Thuillier et al. 2004a, upper left) and typical laboratory VUV lamps (H2) (from Cottin et al. 2003—lamp 1, upper right, from Chen et al. 2014—lamp 2, middle left and Es-sebbar et al. 2015—lamp 3, middle right). To date, no laboratory lamp is able to accurately simulate the solar spectrum in the VUV, although the most recent developments are much improved and have eliminated the strong emission at 160 nm by adding He to H2. The VUV spectra emitted by the lamps are extremely sensitive to the pressure and composition of the gas circulating within the lamps. The lower panel shows a comparison between a Martian laboratory simulator (high pressure Xe lamp—lamp 4) (Poch et al. 2013) with a simulated UV spectrum reaching the surface of Mars for two extreme scenarios: (1) during northern summer low dust loading (\(\tau=0.1\)), at the equator and local noon (taken from Patel et al. 2002); (2) during spring (vernal equinox) for a dusty day (\(\tau=2.0\)), at 60°N and local noon (taken from Cockell et al. 2000). Representability of solar Martian simulator is quite satisfactory for UV radiations
Photochemical experiments related to space environments can also be conducted in gas or the solid phase using synchrotron facilities. Although they are all different with their own specificities, the SOLEIL synchrotron located in St Aubin (France) is taken as an example in the following discussion. One of the SOLEIL beamlines, the “Dichroïsme Et Spectroscopie par Interaction avec le Rayonnement Synchrotron (DESIRS)” is of particular interest for the topics discussed in this paper. It is an undulator-based VUV beamline covering range from 25 to 300 nm (i.e. 5–40 eV) (Nahon et al. 2012). It provides high spectral purity, high resolution, and variable polarization, and is thus a valuable tool for astrochemical studies. As an example, it has been used to evaluate the photochemical effect of circularly polarized light on chiral organic materials under simulated inter/circumstellar conditions. The results provide information about the importance of asymmetric interstellar photochemistry for supplying the Earth with some of the enantio-enriched organic materials needed as prebiotic building blocks of life (de Marcellus et al. 2011; Meinert et al. 2014).
However, such beamlines cannot be considered as a substitute for space exposure in Earth orbit. First of all, due to the high demand, the maximum accessibility of DESIRS is 6 days per year per project after it has approval by a committee. To deal with this short exposure time, increasing the flux compared to the Sun to simulate longer irradiation times can of course be considered. The integrated solar flux between 100 and 300 nm is of the order of \(2\times 10^{13}~\mbox{ph}\,\mbox{cm}^{-2}\,\mbox{s}^{-1}\) (Thuillier et al. 2004b) while, in the same range of wavelengths, the flux generated by the DESIRS synchrotron source can reach \(10^{15}~\mbox{ph}\,\mbox{cm}^{-2}\,\mbox{s}^{-1}\), i.e. about 50 times more intense than from the Sun. Thus, 6 days of exposure at SOLEIL can generate a dose similar to 7200 h of exposure to the Sun (i.e. about twice the amount of photons collected during the EXPOSE-R experiment on the ISS between 2009 and 2011 (Rabbow et al. 2015b)). However, it should be noted that samples are not continuously facing the Sun when they are in space. Moreover, if the irradiated surface is considered, taking into account that the synchrotron beam irradiates a maximal surface of \(0.5~\mbox{cm}^{2}\), samples similar to those exposed in space (and described below, see Sect. 3.2.2.c1) can be exposed only one by one. Hence, an experiment similar to AMINO described below, with 30 samples exposed during 3000 h to the Sun on EXPOSE-R, would require about 15 weeks of continuous use of the line, while about 37 weeks is needed in order to imitate PSS on EXPOSE-R2, with 75 samples.
Apart from this consideration regarding the duration of an experiment similar to those conducted in space, an additional limitation has to be considered. A synchrotron beam cannot be customized to reproduce at once faithfully the whole VUV spectrum emitted from the Sun. If the Solar flux can be precisely reproduced at each wavelength, relative to the others, this would only be in a sequential manner, scanning monochromatically the range desired. The beam can embrace a larger range of wavelengths, but in this case, solar spectra reproduction will be less accurate. This point is of prime importance since some organic molecules such as purines or pyrimidines absorb photons with the same efficiency (same order of magnitude for the absorption cross section) in all of this wavelength range (Saiagh et al. 2014, 2015), and are then photolyzed in space simultaneously over a very broad UV domain, a situation which is not feasible on a synchrotron beamline.
Therefore synchrotron beamlines cannot be used for the same kinds of studies as those conducted in space. They are, of course, a great tool for measuring fundamental parameters, such as photolysis quantum yield at a specific wavelength, branching ratio, and the exact photodissociation threshold of a specific molecule, thus providing in-depth and valuable additional information.
3 Current and Past Astrobiology Facilities
The exposure of microorganisms to space environment started almost with the conquest and exploration of space in the 1960s. Such experiments were conducted outside the Gemini 9 and 12 modules 1966 for a few hours (Hotchin et al. 1968). The first elaborated exposure facility was used during the Apollo 16 mission during the transearth coast, i.e. the journey back to Earth (Taylor et al. 1974) where the potential for microorganisms to survive the harsh conditions of outer space was revealed. Since then, space agencies regularly issue calls of opportunities to use space facilities to conduct experiments in Earth orbit. Space exposure facilities for both biological and chemical samples were initially conducted on the LDEF and EURECA platforms, and then outside the MIR space station. In recent years (2004, 2009, 2014), joint announcements coordinated by ESA (Europe), NASA (USA), JAXA (Japan) & CSA (Canada) entitled International Life Science Research Announcement (ILSRA) have been released and experiments addressing astrobiology have been selected. In the following section the main facilities used (past and present) are described. Specific details with respect to hardware and science results for chemistry and biology related experiments are also described.
3.1 Common Tools and Facilities
More than two decades of experiments on the International Space Station (ISS) and free-flying satellites (since the Gemini mission in the 1960s) have provided new information about the evolution of organic and biological material in space and planetary environments (Fig. 7). Research in low Earth orbit has contributed to our knowledge on extraterrestrial delivery processes, the responses of life to the space environment and crucial aspects of planetary protection (Guan et al. 2010; Horneck et al. 2010; Nicholson et al. 2011). Some of the main facilities used both for chemical and biological applications are described in this section.
Some of the past and current astrobiology devices used in Low Earth Orbit. From upper left to bottom right: LDEF, FOTON with embedded samples in its heat shield for STONE experiments, one lid of Biopan, EURECA, EXPOSE-R, and O/OREOS nanosatellite (artist’s impression). (Credits: NASA, ROSCOSMOS, ESA/Kayser-Threde, NASA, ESA & NASA)
3.1.1 LDEF
The NASA Long Duration Exposure Facility (LDEF) was a 10 ton, cylindrical (\(4 \times 9~\mbox{m}\)) exposure facility launched in April 1984 by the Space Shuttle Challenger (STS-41-C). It was initially scheduled to be retrieved after eleven months in space, however, its recovery was delayed due to the dramatic loss of Challenger in 1986. It was finally recovered in January 1990 using the Space Shuttle Columbia (STS-32), after remaining in space for 2107 days; making it the longest duration space exposure experiment. Its experiments and samples were mainly selected for testing space radiation, temperature changes and collision with space debris, on engineering materials, with the prospect of building future spacecraft and space stations. Some experiments concerning the survival of spores and tomato seeds were conducted and proved their radiation resistance after recovery (Kahn and Stoffella 1996).
3.1.2 EURECA
EURECA (EUropean REtrievable CArrier) was designed to conduct space experiments for a duration of a few months. It was a 4.5 ton satellite meant to be released and retrieved by NASA Space Shuttle. It was the first European satellite designed specifically for microgravity experiments, after the Russian Bion (since 1973) and Foton capsules (since 1985). An important feature for EURECA in its original conception was reusability: it was built to survive five flights over a 10-year period. However, due to lack of funding it was used only once (Innocenti and Mesland 1995).
EURECA-1 (including 15 experiments) was launched with the space shuttle Atlantis mission STS-46 in July 1992 and was recovered during the space shuttle Endeavour mission STS-57 in June 1993. On EURECA, an exposure tray called ERA (Exobiology and Radiation Assembly) was mounted, in which bacteria and organic materials were exposed to study their survival and evolution in space (e.g. Dose et al. 1995; Greenberg et al. 1995).
3.1.3 Salute-6,7, Bion-9,11 and MIR Space Station
The Soviet space stations Salute-6 (1977–1982) and Salut-7 (1982–1991) were used to expose a variety of samples with an astrobiology relevance. A series of exposure of 13 and 16 months on Salute-7, and similar experiments on MIR, as well as on the Cosmos-2044 (Bion-9) & Bion-11 spacecraft, is reported in Kuzicheva and Gontareva (1999, 2003).
Later, the Soviet and then Russian space station MIR orbited Earth from 1986 to 2001. In 1999, an exposure facility called Perseus Exobiology was mounted for 97 days outside the space station to conduct experiments, including both chemical (amino acids and peptides) and biological samples (Boillot et al. 2002; Rettberg et al. 2002). The Perseus mission was launched on February 20th 1999 with a Soyuz TM-29 from Baikonour (Kazakhstan) with three astronauts onboard. The scientific material built by COMAT Aerospace (Toulouse, France) was carried to MIR with a Progress cargo on April 2nd. The Exobiology experimental hardware was installed outside the station on the Kvant 2 module on April 16th 1999. The Perseus-Exobiology experiment was then run until July 23th 1999, i.e. for 97 days
3.1.4 Biopan on Foton Capsule
Biopan is a pan-shaped retrievable exposure facility (Fig. 8) for experiments in the domains of astrobiology, chemical evolution, radiation biology and radiation dosimetry (Demets et al. 2005). Externally mounted on unmanned recoverable satellites of the Foton type, Biopan flies 2-week missions in low Earth orbit at 63.0° inclination, allowing exposure of biological samples to the harsh space conditions. Six flights were completed between 1992 and 2007 with up to ten different experiments per flight. Biopan carries its experiment packages (total mass 4 kg max.) on two mounting plates (total surface area 1,080 \(\mbox{cm}^{2}\)).
After 2007 the Biopan project was stopped by ESA for political reasons. Since then, the Russian Foton programme has been continued without ESA payloads. A new generation of Foton capsules has been introduced, equipped with solar panels to stretch the flight duration from two to six weeks. For implementation in the current Foton capsule, the thermal compatibility of Biopan would need to be re-assessed because the new Fotons have for the first time a permanent hot side (pointing in zenith direction) and cold side (pointing in nadir direction).
In orbit, the hinged lid of Biopan is opened by remote control whereupon the experiments are exposed to the space environment. At the end of the flight the lid is hermetically closed and locked. During reentry into the atmosphere Biopan and its contents are protected against the frictional heat by an ablative heat shield.
Biopan is equipped with a variety of sensors to monitor and record the environmental history of the test samples. Included are ultraviolet (UV) sensors, a radiometer and a set of eight thermistors to measure the experimental temperatures. The sensor data are stored on board and retrieved after landing. The temperature profile of the experiments is selectable. A non-controlled mode can be chosen with temperatures freely oscillating between \(\leq-20\) and \(\geq+10\)°C, in synchronization with the alternating periods of solar illumination and shadowing in orbit. Alternatively, by using electrical heaters and thermal blankets, a stable temperature can be provided with a fixed set point in the 10–25°C range for experiments in the bottom half of Biopan. Organic molecules, bacterial spores and vegetative cells, archaea, plant seeds, lichens, and tardigrades have been exposed in Biopan to a combination of solar UV, space vacuum, space radiation, wide temperature fluctuations, and weightlessness. Biopan carries reference samples, which are kept under identical conditions but shielded against UV radiation. Additional control samples are maintained on the ground. The typical operational cycle of Biopan includes experiment integration at 1 week before launch, 2 weeks of orbital flight, and return of the experiments to the investigators 4 days after landing.
Biopan was designed and built for ESA by Kayser-Threde (Munich, Germany, now OHB) with Kayser Italia (Livorno, Italy) responsible for the flight software and the electronics. The heat shield is manufactured by TsSKB-Progress (Samara, Russia). A list of all the experiments conducted on Biopan is shown in Table 3.
3.1.5 EXPOSE Outside the International Space Station
The ESA EXPOSE multi-user facility is used for long-term exposure of experimental samples to extraterrestrial solar UV under LEO space vacuum or defined atmosphere on external platforms on the ISS. Two EXPOSE facilities have been used at two different locations of the ISS. The general architecture of the interior of the EXPOSE trays is based on the exposure trays developed and built by the DLR (Cologne, Germany) for accommodation in the cargo bay of Spacelab 1 and D2 as well as on ERA of EURECA (Horneck et al. 1984a, 1984b).
The EXPOSE-R facility, named after its final destination on the Russian external platform is secured to the outer hull of the Service Module Zvezda of the ISS as part of the Russian Segment. This external platform, called URM-D, provides mechanical, electrical, and data interfaces. EXPOSE-R accommodated 10 biological and biochemical experiments, mounted in 4 cavities, called compartments, in each of the three removable containers, called trays (Fig. 9). These trays can also be exchanged when EXPOSE-R is mounted on the URM-D and exposed to the open space, providing increased programmatic flexibility. Attachment, retrieval, and exchange take place by Extra Vehicular Activities (EVA) of the ISS crew (Rabbow et al. 2015b). The first EXPOSE-R and its experiment inserts were launched on flight 31P on November 26, 2008 on an unmanned PROGRESS cargo ship on a SOYUZ launcher from Baikonur, Kazakhstan. After a storage period inside the ISS, it was mounted to the external URM-D by EVA. On March 10, 2009 the exposure of the experiments to the open space environment was initiated. The complete EXPOSE-R monoblock with its three mounted trays was recovered by EVA on January 21, 2011 and brought inside the ISS; here, the trays were extracted from the monoblock and returned to Earth by one of the last Shuttle flights, STS-133/ULF 5 landing on March 9th, 2011. The 682-day period outside the ISS provided continuous exposure to the cosmic-, solar-, and trapped-particle radiation background and \(>2500~\mbox{h}\) of unshadowed solar illumination.
The monoblock of EXPOSE-R, without trays, remained on board of the station. It has been reused for the third mission, EXPOSE-R2, re-loaded with fresh trays equipped with samples of three new experiments from ESA and one from IBMP. The upload of the new trays was on July 24th 2014 on Progress 55P. EXPOSE-R2, thus consisting of a veteran monoblock and three new trays, was deployed on August 18th on the URM-D platform of the Zvezda module.
To allow for full outgassing of all volatiles without ensuing Sun-induced contamination of the window surfaces (as experienced on EXPOSE-R, see Demets et al. 2015), the Sun shield was retained on top of the trays for several weeks. On October 22nd 2014 a second EVA was performed to remove the Sun shield, starting a 16-month period of solar exposure which lasted until February 3rd 2016. On March 2nd 2016 the first tray was downloaded back to Earth with Soyuz 44S. Trays number two and three were brought back on Earth on June 18th 2016 with Soyuz 45S. A follow-on flight, EXPOSE-R3, is currently not included in the ESA plans for the coming years.
Another EXPOSE facility, EXPOSE-E, was very similar to EXPOSE-R, but provided 3 lids to cover 7 compartments of the 3 trays. EXPOSE-E was launched February 7th 2008 with the Atlantis Space Shuttle, STS-122, to the ISS. The facility was part of the European Technology Exposure Facility platform (EuTEF) and integrated from launch on the starboard cone of the European Columbus module. Seven international experiments were selected by ESA for this 18 month-long mission, including 2 dosimetry experiments. On September 2 2009, the complete EuTEF platform with EXPOSE-E was recovered and directly stowed in the cargo bay of the Discovery Space Shuttle (STS 128) for return to Earth. During the 18 months (547 days) of exposure in space, samples were submitted to 1300 to 2500 hours of illumination (depending on their actual location on EXPOSE-E) due to the orbit of the International Space Station around the Earth, its orientation toward the Sun, and various shadowing effect due to the geometry of the facility (open lids) and its local implantation on the ISS.
All three EXPOSE missions were prepared in an extensive preflight test program using the Planetary and Space Simulation Facilities (PSI) at the Deutsches Zentrum für Luft und Raumfahrt (DLR—German Aerospace Center) in Cologne.Footnote 1 This ground facility is described in further details in Martins et al. (2017); Rabbow et al. (2015a). Several Experiment Verification Tests (EVT) ensured that experiment designs and individual samples were suitable for the rough ride to and from and a long duration stay in the hostile LEO environment. Experiment Sequence Tests and Science Verification Tests (EST, SVT) were the final ground tests and rehearsal for flight: experiments were accommodated as for flight in identical ground trays to verify the whole series of procedures.
During the ground tests, the experiments were exposed to space parameters similar to those expected during the mission: high and low temperatures and temperatures repeatedly cycling through the freezing point 0°C, short wavelength polychromatic UV down to 200 nm, and vacuum, provided individually or in combination. At the same time as the EXPOSE-E and EXPOSE-R/-R2 space missions, an identical set of flight trays fully equipped with samples was exposed in the PSI at DLR as in space, as far as technically feasible and according to the data available.
The space EXPOSE facilities measured environmental data every 10 seconds, such as temperature and visible or UV photons reaching the facility, as well as housekeeping and functionality data of the facility. While EXPOSE-E data were received by telemetry regularly, EXPOSE-R data were downloaded from the ISS on PCMCIA cards and by telemetry every few months.
The two past missions EXPOSE-R and EXPOSE-E were an overall success, although some environmental data were missing due to a variety of reasons from both missions. The results of the EXPOSE-E are published in the Issue No 5, Volume 12 of Astrobiology, published in May 2012. The results of the EXPOSE-R mission and experiment results are published in Issue 1, Volume 14 of the Journal of Astrobiology, published in January 2015. A list of all the experiments conducted on EXPOSE facilities is shown in Table 4.
3.1.6 TANPOPO Outside the International Space Station
Named after dandelion in Japanese, the “Tanpopo” mission is Japan’s first astrobiology space experiment at Kibo, or the Japanese Experiment Module (JEM), Exposed Facility on the ISS. The mission utilized the Exposed Experiment Handrail Attachment Mechanism (ExHAM) designed for multipurpose exposure experiments by JAXA (Fig. 10). The official ISS experiment code name is “Astrobiology Japan” representing “Astrobiology exposure and micrometeoroid capture experiments”. The aim of this mission is to investigate the possible interplanetary transfers of prebiotic organic compounds to the Earth as well as the transfer of the terrestrial microbes out of the earth, by sample return analyses of both Capture and Exposure Panels in the low Earth orbit (Yamagishi et al. 2009).
To achieve these objectives, the Tanpopo mission consists of following six, sub-divided themes:
-
(1)
Intact capture of terrestrial aerosols that may contain microbial colonies, in the low Earth orbit.
-
(2)
Long exposure of extremophile microbes in the low Earth orbit.
-
(3)
Long exposure of the pre-biological organic analogue compounds in the low Earth orbit.
-
(4)
Intact capture of organic bearing micrometeoroids in the low Earth orbit.
-
(5)
Spaceflight evaluation of ultralow-density aerogels (\(0.01~\mbox{g}/\mbox{cm}^{3}\)) originally developed.
-
(6)
Flux measurement of meteoroids and orbital debris in sub-mm ranges at the vicinity of the ISS.
As stated above, the experimental apparatus consists of two major components: the Capture and Exposure Panels. The Capture Panels (Fig. 11a) are used for intact capture of microparticles such as terrestrial aerosols, micrometeoroids and orbital debris for the sub-themes 1, 4, 5 and 6, with ultralow-density aerogel blocks with inner and outer bulk densities of \(0.01 \mbox{ and } 0.03~\mbox{g}/\mbox{cm}^{3}\), respectively (Tabata et al. 2015). The Exposure Panels (Fig. 11b) allow pre-loaded samples of microbes and organic compounds in laboratories to be exposed for 1–3 years in the low Earth orbit for the sub-themes of 2 and 3. For each Exposure Panel, twenty Exposure Units are attached on one common base plate. All Capture and Exposure Panels to be utilized in Tanpopo’s 3-year mission plan were onboard the Space-X Dragon commercial cargo spaceship CRS-6 and then launched on April 15th, 2015 (JST) from Cape Canaveral (USA) by the Space-X Falcon-9 rocket. The panels for the first year exposure were manually installed on the space-pointing, ram, and north faces of the ExHAM-1 by the ISS crew S. Kelly inside the Kibo pressurized facility and transferred to its airlock on May 14th. The first year exposure experiment of the ExHAM-1 has started since May 26th, and currently it is planned that these panels will be recovered after approximately one-, two- and three-year exposure. The first year samples are currently under investigations.
(a) A flight model of the Tanpopo Capture Panel (Courtesy: JAXA/Tanpopo Team). (b) A flight model of the Tanpopo Exposure Panel (Kawaguchi et al. 2016). Twenty Exposure Units are attached on a base plate of an Exposure Panel. Both panels are \(100 \times100 \times19.5~\mbox{mm}\) in dimension. (Courtesy: JAXA/Tanpopo Team)
3.1.7 O/OREOS Nanosatellite
The NASA 3U cubesat Organism/Organic Exposure to Orbital Stresses (O/OREOS) was launched in November 2010 to undertake a 6-month mission to demonstrate astrobiological measurement technologies. After more than five years, the nanosatellite is still operational in LEO above 600 km and is used for educational purposes, including routine radio telemetry of spacecraft health and status; science data are no longer downlinked. O/OREOS achieved its overall goal to utilize autonomous instrumentation and sensors for the in-situ investigation of microbes and biomarkers in space conditions using a free-flying nanosatellite. Its launch to a high-inclination (72°), 650-km Earth orbit aboard a US Air Force Minotaur IV rocket from Kodiak, Alaska provided conditions where in-orbit solar and galactic particle radiation doses—much of it from significant durations spent in the inner Van Allen Belt—exceed ISS dose rates by approximately a factor of fifteen. The O/OREOS 3U satellite (\(3~\mbox{dm}^{3}\) volume total) consists of a control bus and two high-capability science payloads, each contained in 1U cubesat module (Fig. 12).
The SESLO experiment collected data on the survival and metabolic activity for microorganisms three times during the 6-month mission. This payload consists of three “bioblock” modules, each with twelve \(75\mbox{-}\upmu\mbox{L}\) sample wells connected by microfluidic channels and valves that allow the introduction of germination/growth media. Prior to spacecraft integration and flight, bacterial cells were dried onto the walls of the sample wells and the bioblocks were sealed using a gas-permeable membrane. Using 3-color LED illumination (470, 525, and 615 nm), the growth and metabolism of the microbe Bacillus subtilis was successfully measured during the mission at 2 weeks, 3 months and 6 months after launch (Nicholson et al. 2011). Results are summarized below in “O/OREOS Results” (Sect. 3.2.3.e).
The Space Environment Viability of Organics (SEVO) experiment accomplished real-time analysis of the photostability of organic biomarkers. Four different molecular classes (a polycyclic aromatic hydrocarbon, an amino acid, a quinone and a metalloporphyrin) were selected for flight based on their astrobiological and exobiological relevance. The samples were deposited as thin films by vacuum sublimation onto MgF2 windows. The SEVO payload consists of a miniaturized UV-visible-NIR spectrometer and a 24-sample carousel that houses hermetically sealed sample cells, described in more detail below in “O/OREOS-SEVO payload sample cells” (Sect. 3.1). Integrated optics enable the use of the Sun as the light source for both sample electromagnetic radiation exposure and sample spectroscopic measurement. The SEVO payload returned spectral data sets over 17 months of space exposure (Mattioda et al. 2012). Results are summarized below in “O/OREOS Results” (Sect. 3.2.3.e).
The O/OREOS mission, including launch, successful operation of both payloads, and download of collected mission data, achieved full success in May 2011 (Ehrenfreund et al. 2014; Kitts et al. 2011). The SESLO and SEVO experiments serve as precursors for experiments on the ISS, future free-flyers, and planetary surface exposure facilities. Future flight opportunities can leverage this tested and proven triple-cubesat configuration, particularly the flight engineering (power, communications, control, and data handling) and payload environmental control systems (temperature, pressure, humidity).
3.1.8 OREOcube: An ISS Hitchhiker and New In-situ Exposure Platform
Some of the technologies that have been recently demonstrated on small satellites are ideal candidates for minimal-development payloads for accommodation on the ISS. Modular, multipurpose payload racks storing and supporting ISS experiments are located on the Destiny, Columbus, and KIBO Modules of ISS. These EXpedite the PRocessing of Experiments for Space Station (EXPRESS) racks and 33% of NASA’s external research platforms will be used for science instrumentation, including cubesat-derived instruments, which are housed in Nanoracks, Inc. systems designed for that purpose. This availability enables ISS to support not only NASA, ESA, and JAXA research, but also that of the broader worldwide scientific community.
A recent example of utilizing a cubesat payload as the basis of an ISS facility instrument is the OREOcube experiment, which is based on O/OREOS-SEVO technology described above. OREOcube will be installed as an external exposure facility on the ISS under ESA’s European Program for Life and Physical Sciences in Space (ELIPS) to study the evolution of organic and prebiotic materials in space. OREOcube will consist of two SEVO cubes (Fig. 13) and can record daily changes in ultraviolet and visible light absorption spectra of organic compounds, revealing the consequences of their exposure to solar UV and visible light and space ionizing radiation. The advantages over a free-flyer experiment are that data can be downloaded from the ISS more effectively and more frequently with on-board data averaging and storage capability using a standard power-and-command interface. Additionally, the payload or its sample carousels can be retrieved to enable additional sample- and photoproduct-characterization experiments in the laboratory on Earth.
Some of the same organic materials characterized in space by SEVO (Mattioda et al. 2012) will thus be characterized in an environment with very similar levels of UV and visible light, but some 15 times less ionizing particle radiation dose rate than the SEVO samples experienced during the O/OREOS mission, assisting in the differentiation of the consequences of these two different radiation environments.
While O/OREOS SEVO studied thin films of organic molecules, OREOcube’s science goal is to understand the interaction mechanisms of organic and inorganic thin films under the influence of solar and cosmic radiation. Organic thin-film candidates are similar to the ones from O/OREOS, whereas inorganic compounds of interest are, for example, metal alloys and metal oxides. Their photocatalytic impact is highly important from an astrochemistry/astrobiology point of view and pre-flight ground-based test and simulation experiments revealed an intriguing photo-protection effect by some iron oxides (Elsaesser et al. 2014).
Possible implementation scenarios for OREOcube consider attachment on either the Columbus module, the JAXA-KIBO facility, or the NanoRacks External Payload Platform (NREP). The last of these would require little modification of the SEVO cube-format payload in terms of additional hardware and add the benefit of already-available data and power connections. NanoRacks NREP was deployed outside the ISS in August 2016, while OREOcube is schedule to be delivered to the ISS in the 2018 time frame. Recent plans at ESA also include integration of OREOcube in a newly designed and developed ‘European Exposure Facility’, which would be able to accommodate various space exposure experiments with the need for in-situ measurements.
3.1.9 STONE Experiments
The STONE experiments were conceived to determine if sedimentary meteorites from Mars could survive entry into the Earth’s atmosphere, the rationale being that it is more likely that traces of hypothesized Martian life would be associated with rocks formed in an aqueous sedimentary environment. Although there are over one hundred known meteorites from Mars, they all are igneous (although Tissint, a fall recovered in 2011, shows evidence of having been influenced by water (Chennaoui Aoudjehane et al. 2012)). The three main objectives of STONE experiments are:
-
(1)
Determine whether Martian sedimentary meteors could resist entry into the Earth’s atmosphere and reach the ground.
-
(2)
Determine whether life forms embedded in the rocks could survive entry into the Earth’s atmosphere.
-
(3)
Determine whether natural biosignatures contained in the sediments could survive entry into the Earth’s atmosphere
In these experiments, rock samples were fixed around the stagnation point of the heat shield of FOTON capsules used to carry out experiments in lower Earth orbit (Fig. 14). Of six missions flown between 1999 and 2007, three were successful (Stone 1, Stone 5 and Stone 6). The rocks exposed included dolerite, a medium-grained basaltic rock as representative of volcanic rocks on Mars, dolostone (a carbonate), an artificial sediment consisting of 80% basalt and 20% gypsum in a carbonate/sulphate cement, a quartz sandstone, a gneiss, a volcanic sandstone (with a siliceous cement), and a carbonaceous laminate (Brack et al. 2002; Brandstaetter et al. 2008; Foucher et al. 2010; Parnell et al. 2011). Both the volcanic sandstone and the laminite contained carbonaceous biosignatures. In the former case, they consisted of carbonaceous microfossils of primitive prokaryotic organisms (similar to those expected on Mars (Foucher et al. 2010; Westall et al. 2011)). For some of the samples, microorganisms (the photosynthetic endolith Chroococcidiopsis; Cockell et al. 2007) were inserted in holes drilled into the rocks or, for Stone 6, placed (painted) onto the back surfaces of the rocks, away from the exposed surface and protected from the heat of entry by 2 cm of rock. The samples were embedded into the Foton heat shield as 6 cm diameter, 1 cm thick discs except for Stone 6 which was dome shaped, having an apex 2 cm thick.
Stone samples inserted at the bottom of the heat shield of the FOTON M3 capsule in 2007. Actual photographs of the FOTON shield at the top showing the location of the samples (one of the samples was lost during reentry), accommodation scheme at bottom left, and picture of one sample in the STONE 6 configuration at bottom right
3.2 Space Experiments for Chemistry
In this section, the experiments related to astrochemistry are described (Table 5). Beyond the exposure space carriers detailed in the previous section, samples, hardware, and results specifically dedicated to chemistry are presented and discussed.
3.2.1 Diversity of Samples for Astrochemistry Experiments in Space
a. Interstellar Medium
Astronomical observations have shown that carbonaceous matter is ubiquitous in our own as well as distant galaxies. A number of organic structures that are used in contemporary biochemistry on Earth are observed in the Solar System environments as well as in circumstellar and interstellar regions. The interstellar medium (ISM) is essentially made of H and He and is about a few percent of the galactic mass. Interstellar material is dominated by gas (99%). The remaining 1% is made of silicate in the solid state and carbon-based mm-sized dust particles that can be observed throughout interstellar clouds. They provide surfaces for accretion of gas phase species and subsequent grain surface chemistry (Ehrenfreund and Charnley 2000; Herbst and van Dishoeck 2009). Fundamental physical parameters such as temperature and density vary strongly across the spectrum of interstellar clouds. Currently ∼180 molecules are detected in the interstellar and circumstellar gas although some of them are only tentatively identified and need confirmation. Fifty-three molecules are found in extragalactic sources (http://www.astro.uni-koeln.de). \(\mathrm{H}_{2}\) is by far the most abundant molecule in cold interstellar regions, followed by CO, the most abundant carbon containing species, with \(\mathrm{CO}/\mathrm{H}_{2} \sim 10^{-4}\).
Circumstellar envelopes of carbon-rich evolved stars are regions favorable to a carbon chemistry that is similar to soot formation. Polycyclic aromatic hydrocarbons (PAHs), fullerene-type material and large aromatic networks are efficiently formed in those environments. Those large carbon-bearing molecules are then observed in the interstellar medium (ISM) in various charge states (see Tielens 2008 for a review). In the ISM mixed neutral and ionized PAHs are thought to be responsible for the unidentified infrared emission bands (UIBs) and the UV and visible diffuse interstellar bands (DIBs) (Allamandola et al. 1999). Since the abundance of any PAH and complex carbonaceous molecules depends on its balance between formation and destruction, the knowledge on the survival times (or destruction rates) of these molecules is crucial.
In cold dark clouds with a temperature of 3–10 K the sticking coefficient of most atoms and molecules is close to unity and particles freeze as ice layers that contain molecules such as \(\mathrm{H}_{2} \mathrm{O}\), CO2, CO and CH3OH, with smaller admixtures of CH4, NH3, \(\mathrm{H}_{2}\)CO and HCOOH (Boogert et al. 2008; Gibb et al. 2004; Öberg et al. 2011; Pontoppidan et al. 2008). Dark clouds provide a favorable environment for the formation of larger molecules. There, density is rather high (\(\sim10^{6}\mbox{ cm}^{- 3}\)) and they experience a quite low radiation field of \(\sim10^{3}~\mbox{photons}\,\mbox{cm}^{2}\,\mbox{s}^{-1}\) induced by cosmic rays (Prasad and Tarafdar 1983). Low density (\(\sim10^{3}~\mbox{atoms}\,\mbox{cm}^{-3}\)) and temperatures \(\sim100~\mbox{K}\) characterize the diffuse interstellar medium. Diffuse clouds are filamentary structures surrounding the cold dense interstellar regions. Ices are not present in those regions and a strong radiation field of \(\sim 10^{8}~\mbox{photons}\,\mbox{cm}^{- 2}\,\mbox{s}^{-1}\) (Mathis et al. 1983) dominates the formation and evolution of molecules and dust. Small carbonaceous molecules in the gas phase are easily destroyed by radiation. Atoms with ionization potentials less than 13.6 eV are photo-ionized. Stellar sources in their late stage of evolution are injecting “Stardust,” in the form of dust and molecules, into interstellar clouds. Whereas in dense interstellar clouds ice is covering the low temperature dust, and experiences low UV radiation flux, in diffuse clouds dust it is strongly processed by UV radiation and shocks. Understanding the evolution of interstellar material, environmental conditions and dust cycling provides important insights into the nature of the material that is later incorporated into protoplanetary regions. Exposure experimentation in Earth orbit is a useful tool to study the stability and long term evolution of molecules such as PAHs, fullerenes, and material resulting from ice irradiation.
b. Planetary Atmospheres and Endogenous Sources of Organic Compounds in Planetary Environments
As reviewed in Cottin et al. (2015a), the origin of organic materials preceding the emergence of life on the Early Earth is one major topic in astrobiology. Several complementary sources are considered, among them the coupled system ocean/atmosphere (the primitive soup theory) (see e.g. Oparin 1953; Trainer et al. 2004). A key question resides in the capacity of the primitive atmosphere to produce large organic molecules enriched by nitrogen and oxygen chemical functional groups, representative of prebiotic molecules. In this context, methane (CH4) atmospheric photochemistry appears as a source of large hydrocarbons in planetary atmospheres, driving organic growth in those environments (Raulin and Bruston 1996). Unfortunately, methane photolysis has been mostly studied only at Lyman \(\alpha\) wavelength. To complete this sparse experimental knowledge, previous EXPOSE missions have enabled a direct measurement of the methane photochemical system in space conditions (Carrasco et al. 2015; Cottin et al. 2012).
Atmospheric chemistry enabling nitrogen incorporation in hydrocarbons is the next issue to be addressed to understand the production of large prebiotic molecules in planetary atmospheres. For this purpose, Titan, the largest satellite of Saturn, is a unique place in the Solar System to observe the coupling between methane photochemistry and nitrogen reactivity. Titan’s dense atmosphere, made primarily of \(\mathrm{N} _{2}\) and CH4, hosts an extremely efficient “chemical factory” in which these simplest molecule evolve towards complex nitrogen containing organic hazes (Israël et al. 2005; Waite et al. 2007). Furthermore, it was shown in the laboratory that prebiotic molecules, such as adenine (\(\mathrm{C}_{5}\mathrm{H}_{5}\mathrm{N}_{5}\)), could be formed via chemistry mimicking what occurs in the atmosphere of Titan (Hörst et al. 2012). However, the limitations of the instruments on-board the ongoing Cassini-Huygens space mission does not allow for an identification of the processes responsible for the production of compounds of high prebiotic interest. The chemistry coupling nitrogen and methane remains to date largely unknown. Moreover, observations of the high atmosphere of Titan made by Cassini’s INMS and CAPS instruments show that a complex organic chemistry is occurring in Titan’s ionosphere, potentially yielding high molecular weight compounds (Waite et al. 2007). These complex organic compounds in the atmosphere of Titan are submitted to energetic UV irradiation in the ionosphere. What is their chemical behavior under such UV bombardment? What could be the products of such irradiation? What is the effect of other energetic radiations in addition to UV radiation? Again, to answer these questions, the use of space exposure experiments using CH4 and \(\mathrm{N}_{2}\) are improving our knowledge about these complex chemical mechanisms thanks to an appropriate input of energy both in the form of photons and other energetic particles to initiate the chemical evolution.
c. Small Bodies and Exogeneous Sources of Organic Compounds in Planetary Environments
Small bodies in the Solar System are known to contain organic matter that could have been delivered into the primitive Earth, potentially playing a significant part in chemical evolution leading to the origin of life. Initiated either in the ice phase (Colangeli et al. 2004; Cottin et al. 1999; Meinert et al. 2012, 2016; Strazzulla and Palumbo 1998; Vinogradoff et al. 2013) or in the gaseous phase in the protosolar nebula (Nuth et al. 2008), complex organic compounds are observed in carbonaceous chondrite (Martins 2011), micrometeorites, UltraCarbonaceous Antarctic micrometeorites (UCAMMs) and interplanetary dust particles (IDPs) (Clemett et al. 1993; Dartois et al. 2013; Matrajt et al. 2013) and in comets (Capaccioni et al. 2015; Elsila et al. 2009; Goesmann et al. 2015; Wright et al. 2015; Altwegg et al. 2016; Fray et al. 2016). The surfaces of comets, asteroids, and their fragments (i.e. meteorites, micrometeorites and IDPs) are exposed to ultraviolet radiation and cosmic rays, which transform and/or degrade any organic molecule present on their surface. High energy processes in the surface of those planetary bodies may break molecular bonds, leading to new molecular rearrangements and new molecular species, or to the destruction of the organic content of the object. Recent data from the Rosetta comet rendezvous mission show a large number of organic molecules, in particular those containing N and O on the surface (Goesmann et al. 2015; Wright et al. 2015). A study of the stability of organic species in a relevant space environment is crucial to constrain the amount of organic material that might have been imported to the primitive Earth, especially on small particles such as IDPs in which the organic content is much less protected from radiations than in the larger bodies where only the surface is affected by radiation (Muñoz Caro et al. 2006). In addition cosmic rays change the elemental and isotopic composition in meteorites (Marti and Graf 1992). However, certain minerals protect organic molecules against degradation by radiation (i.e. they have a shielding effect), with organic molecules being able to survive for geologically long periods (billions of years) (Aubrey et al. 2006; Garry et al. 2006; Peeters et al. 2009). Sulphates such as gypsum and jarosite (Aubrey et al. 2006; dos Santos et al. 2016) and clay minerals (dos Santos et al. 2016; Martins et al. 2011; Poch et al. 2015) seem to have a shielding effect protecting organic molecules against destruction. Sulphates protect amino acids likely because of their opacity to UV radiation (dos Santos et al. 2016).Therefore, samples including amino acids, small peptides, nitrogenated bases, or organic residues resulting from laboratory irradiated ice mixtures (Baratta et al. 2015), and exposed to real space environments provide crucial information about the energetic processes that comets, asteroids, meteorites and IDPs are exposed to, as well as the role of mineral surfaces in the photochemical stability of organic molecules (Saiagh et al. 2014, 2015).
d. Organic Molecules and Biosignatures at Mars Surface
Mars is a primary astrobiological target (Cottin et al. 2015a) because its past environmental conditions may have been favorable to the emergence of a prebiotic chemistry, and maybe even of a biological activity. Since 2004, several space missions (Mars Exploration Rovers, Mars Express, Phoenix, Mars Reconnaissance Orbiter and Mars Science Laboratory) have provided data emphasizing the past presence of liquid water on the surface of the planet, an essential ingredient for life as we know it. Thus, Mars is certainly the best target in the Solar System to search for past and, eventually, even for present extraterrestrial life since the Red Planet harboured more liquid water on its surface about 4 billion years ago (Bibring et al. 2006; Squyres et al. 2004). At that time, environments on both Mars and the early Earth showed more similarities than today (Westall 2005; Westall and Cockell 2015; Westall et al. 2011, 2013). Moreover, there is a good possibility that records of prebiotic chemistry or primitive life could still be present, even after 4 billion years since the tectonic activity that has eliminated almost all rocks older than about 4 Gy on Earth was either non-existent or of extremely limited extent on Mars.
The present MSL mission and the future ExoMars 2020 and Mars 2020 missions to Mars aim to search for traces of past life on the planet. As in situ exploration currently takes place at the surface of Mars, specific laboratory studies are implemented to determine if life could exist or have existed under martian surface conditions. The objective of such studies is to investigate the potential records that the prebiotic chemistry and/or biological activity are/were able to produce and their ability to resist and be preserved in the Martian environment.
Since terrestrial life consists of organic molecules, a logical step after the “follow the water” strategy is “follow the organic compounds”. The detection of organic molecules is a key objective because they are among the best indicators for prebiotic chemistry and even past or present biological activity. These organic signatures should therefore be detectable near the surface or in the surface rocks. In addition, since Mars is still experiencing bombardment of meteoritic and cometary material, its surface should also include organic matter imported through these processes (Flynn 1996). However, the amount and forms of organics present on the surface of Mars remains an open question because harsh surface conditions, such as UV radiation and the production of oxidants are most likely a key factor determining their evolution and/or destruction (Stalport et al. 2008, 2009; Ten Kate et al. 2005; Poch et al. 2015). A combination of both environmental factors and alteration during in situ sample analysis may explain why only few chlorinated compounds have been detected in Martian soil to date (Freissinet et al. 2015; Leshin et al. 2013).
As life also produces biominerals (i.e. minerals resulting from a biological activity such as bones, shells or stromatolites), an alternative approach would be to “follow the minerals”. Carbonates are among the most important biominerals that are produced by terrestrial life today (Lowenstam 1981; Mann 2001; Weiner and Dove 2003) but were uncommon on the early Earth (Habicht et al. 2002). Moreover, to date, no large deposits of carbonates have been detected at Mars, only low amounts at very specific local areas (Boynton et al. 2009; Ehlmann et al. 2008). UV radiation has been proposed to explain the photodecomposition of the carbonates and hence their possible evolution (Mukhin et al. 1996), however, other research has shown that carbonates are photochemically stable under Mars-like conditions (Quinn et al. 2006). On Earth, biocarbonates are stable over millions or billions of years. It is reasonable to assume that if they exist at Mars, they could also remain over long period of time (especially in absence of tectonic activity).
Beyond organic compounds and biominerals, the signatures of microbial life can be preserved in the geological record when they are entombed in a mineral matrix. These signatures may be organic in nature, textural (or morphological), or geochemical (Westall and Cavalazzi 2011; Westall and Cockell 2015). The degraded organic molecules of the organisms can be concentrated in and chelated to fine-grained, anaerobic sediments, such as clays and silts. Another possibility is that the organisms may be replaced by a mineral that preserves their morphological shape and, in anaerobic conditions, also the organic molecules that made up the cell. The latter will degrade with time, eventually after about 2 billion years, becoming simple aromatic structures that cannot be related to a specific microbial component. Other signatures of microbial life include the fractionation of carbon and other life-essential elements, such as S. There are also a host of more ephemeral signatures that include minerals or corrosion features in minerals and rocks formed through microbial activity.
Mars is considered to have had habitable conditions conducive to the appearance of life in its very early history. About 3.8 Ga ago, the conditions deteriorated at the surface of the planet with putative viable cells relegated to subsurface habitats, although there could have been brief moments of habitability at the surface at any time since. However, in order to have been inhabited, viable cells would have had to have been transported to these locations from the subsurface refuges (Westall and Cavalazzi 2011; Westall and Cockell 2015).
In this scenario of “punctuated” habitability, in suitable conditions living cells could have been preserved encased in a mineral and rocky matrix. Missions to search for traces of Martian life are concentrating their efforts on ancient terranes dating from the early period (Noachian) when there was a greater likelihood of life on the surface of the planet. Rocks containing the signatures of microbial life will have been exposed at the surface of the planet to radiation for variable lengths of time, depending upon how long they have been uncovered by erosion. While UV radiation only penetrates a few mm into the surface of a rock, cosmic and galactic radiations go much deeper (Pavlov et al. 2012).
Therefore, in order to investigate the impact of radiation reaching the surface of Mars, various samples have been exposed to space conditions using LEO experiments. Among the organic molecules those selected for Mars case studies can be related either directly or indirectly through a set of diagenetic alterations to biogenic sources and cannot be synthesized by abiotic processes (Simoneit et al. 1998). The study of the stability of prokaryotic bio-indicators (such as hopanoids and hopanes like diploptene and diplopterol) is of prime interest because, if life occurred on Mars, it was likely under a primitive form (such as terrestrial prokaryotic organisms), due to the short period of favorable conditions. Hopanoids and hopanes can resist terrestrial alteration by reductive or oxidative environments, diagenesis or catagenesis processes up to a couple of billion years (Brocks et al. 2003, 1999). It is therefore of great interest to investigate their stability versus radiation and/or to determine whether they produce new resistant organic compounds with regard to the search for terrestrial prokaryotic-like life on Mars. The stability of mineral bio-indicators, such as biogenic carbonates, is also of interest. On the other hand, life would not be the only source of organic material at Mars surface, since there is another source from interplanetary infall (meteorites, micrometeorites, IDPs and comets). This exogenous source is ubiquitous on the surface of Mars (as on Earth) and it is important to determine the evolution of these abiotic molecules (like PAHs, nucleobases, amino acids for instance) in order to distinguish them from biotic ones. The protective or activating effect of mineral matrices such as clays (nontronite, montmorillonite), sulphates (jarosite) or silicates (olivine) can be also studied. Of course, the atmosphere of Mars filters the most energetic part of the UV (below 190 nm, see Fig. 26), therefore specific filters are used and will be discussed in the next section. Energetic particles, however, reach the surface of Mars.
3.2.2 Hardware for Chemistry
a. Salute-6,7, Kosmos 2044, Bio-11 and MIR
Astrochemistry experiments have been conducted since the late seventies by Soviet Union outside the Salute-6 and Salute-7 space stations. Kuzicheva and Gontareva (2003) describe a series of experiments studying the formation of nucleoside and nucleotide under space conditions that were conducted outside the Salute 6 & 7 space stations (Khenokh et al. 1979; Kuzicheva et al. 1989), and were continued using the Kosmos 2044 (Bion-9) (Kuzicheva and Gontareva 1999) & Bion 11 (Kuzicheva and Simakov 1999) spacecraft, as well as sharing the Perseus-Exobiology hardware described below, outside the MIR space station.
The experiments conducted outside the Salute-7 space station were performed (for 13 and 16 months) using a device called Meduza shown in Fig. 15. In this device, samples were loaded in the experiment and covered by quartz lids characterized by a cutoff at 220 nm. Samples consisted in mixture of nucleosides, mixed with phosphate, in order to study the efficiency of nucleotide formation in space conditions (Kuzicheva et al. 1989). Later, similar experiments were performed outside the Kosmos 2044 (in 1989—14 days) and Bion 11 (in 1996–1997—14 days) in an outside container (OC) (see Fig. 16) adjusted on the spacecraft before launch, which was automatically opened once the satellites were in orbit, and closed before return to Earth (on a similar principle than the Biopan ESA facility).
Photograph of the Meduza exposure facility used for experiments outside the Salute-7 space station. plate: \(620\times490\times110~\mbox{mm}\) (picture credit: Kuzicheva and Gontareva 2003)
The outside container \((\mathit{OC})\) for exobiological experiments was set outside Cosmos-2044 and Bion-11 satellites. (A) baseplates with a sample holders; (B) temperature sensor; (C) \(\gamma\)-radiation dosimeter; and (D) circular glasses with dry samples (picture credit: Kuzicheva and Gontareva 2003)
The MIR/Perseus-Exobiology (Boillot et al. 2002) experimental setup consisted of a large piece of anodized aluminum shown in Fig. 18. The two opposite faces accommodated 66 symmetrical cells with sample holders and magnesium fluoride (MgF2) windows glued at one end. A typical transmission spectrum of MgF2 in VUV-UV is shown in Fig. 17. Samples were deposited as solid films (by evaporation) in the cavity facing the window. The outer surface of the hardware was covered with white silicon, light-reflecting paint. Temperature sensors installed inside two opposing cavities measured the temperature in both exposed and light protected cells every 5 min during the mission. The temperature inside the cavities varied between \(-14^{\circ}\mbox{C}\) and \(+ 44^{\circ}\mbox{C}\) with a \(5^{\circ}\mbox{C}\) difference between the exposed and dark cavities. The radiation flux was meant to be recorded outside the cavities by two UV sensors, but this direct measurement failed and finally the UV dose was estimated from an exposure time reconstructed by summing up all the periods of high temperature, resulting in a total irradiance of \(5.16\times 10^{9}~\mbox{J}\,\mbox{m}^{-2}\) and a UV flux of \(2.9\times10^{21}~\mbox{photons}\,\mbox{cm}^{-2}\).
Typical transmission spectrum of a MgF2 windows measured between 110 and 230 nm with a Horiba-Jobin Yvon VUV spectrometer is shown (more details on this instrument in Saiagh et al. 2014). It must be noted that MgF2 is transparent up to \(10~\upmu\mbox{m}\) (\(1000~\mbox{cm}^{-1}\)) in the infrared (not shown)
Photograph of the exposure facility used for the Perseus-Exobiolgy experiment outside the MIR space station. Plate: \(620\times490\times110~\mbox{mm}\). (Picture credit: Boillot et al. 2002)
The samples (amino acids and small peptide molecules) were exposed either unprotected or associated with different mineral simulating micrometeorites such as montmorillonite clay, powdered basalt and the Allende meteorite. Various thicknesses of mineral films were used to estimate a protection threshold. Some of the samples cells were loaded by mixtures of nucleoside and phosphate as a follow up of the experiments on Salute-7 (Kuzicheva and Gontareva 2003).
b. Biopan (Dust/Organic/Uvolution)
The Biopan facility is described in Sect. 3.1 and Fig. 8. Various kinds of exposure cells for samples have been used in the Biopan chemistry related experimental programs: open or closed cells (Fig. 19). In the open cells, gaseous fragments resulting from the photolytic processes on the exposed samples are released into space and lost for analysis. In this case, those volatile molecules cannot further interact with the solid phase sample deposited on the window. Vented cells were used for the UVolution experiment. They are made of an aluminum cylindrical body onto which a 9 mm (diameter) by 1 mm (thickness) MgF2 or quartz window is glued (epoxy glue). The walls of the cell are coated with Alodine to ensure electrical conductivity and prevent discharges during flight. The sample is deposited on the inner side of the window. The refractory sample can be analyzed before and after exposition via spectroscopic (IR, UV) measurements. After exposure, it can be recovered by dissolving in a solvent for further analyses with gas chromatography coupled to a mass spectrometer (GC-MS) and/or liquid chromatography-mass spectroscopy (LC-MS).
Top a and b: view of the BIOPAN/UVolution open cells (height: \(9.1\pm 0.1~\mbox{mm}\), \(\varPhi\): \(9\pm0.1~\mbox{mm}\)). They are made of a cylindrical aluminum body onto which a 9 mm (diameter) by 1 mm (thickness) MgF2 or quartz window is glued (epoxy glue). Middle c and d: view of the UVolution close cells (height: \(9.1~\mbox{mm}\), \(\varPhi\): \(9~\mbox{mm}\)). They are made of two cylindrical aluminium bodies which can be screwed one into the other. An O-ring prevents leaks between the two parts. Two MgF2 or quartz windows are glued at both ends of the cell Bottom e: schematic view of the BIOPAN/ORGANICS sample cells. Each sample cell contains an MgF2 window that contains the deposited molecules, a Viton O-ring to close the sample compartment, a quartz window as bottom sealing window and a Delrin spacer with O-ring as fixture. (Picture credit: H. Cottin, Ruag, Air Liquide)
A first type of closed cells was used during the ORGANICS and UVolution experiment. ORGANICS cells are made of an MgF2 window that contains the deposited molecules, a Viton O-ring to close the sample compartment, a quartz window as bottom sealing window and a Delrin spacer with O-ring as fixture. The sample cells are contained in aluminum sample containers that are closed by stainless-steel bolts (M3). All aluminum surfaces were treated with Alodine. All samples were closed in a sealed glove-box and stored under 1 atm argon. Another type of closed cell was used for UVolution. They are made of two cylindrical aluminium bodies which can be screwed one into the other. A Viton O-ring prevents leaks between the two parts. The volume inside the cell is approximately 275 \(\mbox{mm}^{3}\). Sealed cells can be used to study the photolysis of a 100% gaseous starting mixture, or for the same kind of solid materials as those deposited in the vented cells. In this case, gaseous fragments resulting from photolytic processes of any solid exposed samples are kept inside the cell and can be analyzed after the recovery of the experiment. The walls of the two parts are treated with Alodine. MgF2 or quartz windows are glued at both ends of the cell to enable the photolysis of the sample from the top window and an in-situ IR or UV analysis. However, infrared analysis of the cell content is limited by the cutoff of the windows at 1000 \(\mbox{cm}^{-1}\) for MgF2 and ∼3000 cm−1 for quartz. The deposition of a solid sample can be made when the two parts are separated using the same procedure as that used for an open cell. If the starting material inside the cell is made only of gaseous compounds, then the preparation of the cell proceeds only through the analytical cell described in Sect. 3.2. In each case, solid or gaseous sample, the total pressure inside the cell before launch was adjusted with Ar to 1.5 bar to avoid contamination from Earth’s atmosphere before launch. The closed cells had a small enough leak rate for short duration experiments such as the Biopan’s ones, but too high for long exposure experiments such as the one conducted on the ISS EXPOSE facilities.
In each Biopan experiment, all exposed samples were accompanied by unexposed counterparts that were positioned just underneath the samples exposed to solar photons as shown in Fig. 20, the so called dark controls. Photons can only reach the upper layer, while energetic particles might also interact with the second layer (which has never been observed in LEO).
Samples holders for the BIOPAN/ORGANICS (top) and BIOPAN/UVolution (bottom) experiments. For each of them, two layers of samples could be accommodated: one facing the solar UVs, and the other one underneath, that could be only reached by energetic particles and experiencing the same thermal history than the upper layer
After their return to Earth, depending on the experiment, samples were analyzed by infrared or UV transmission spectroscopy, HPLC, GC-MS, and electrophoresis technics.
c. EXPOSE (Process/Organic/Amino/PSS)
The EXPOSE facility is presented in detail in Sect. 3.1 and Fig. 9. Sample carriers for experiments related to astrochemistry can contain up to 25 sample cells with MgF2 windows for the transmission of solar UV photons to the samples down to 110 nm (Fig. 21).
Sample carrier for EXPOSE-R2/PSS was designed to receive up to 25 exposure cells. They can accommodate open cells (grey) RUAG closed cells (purple) or CNES closed cells (yellow, more easy to see on the right panel). Two layers of samples are flown at the same time: one layer exposed to space, and one layer right below acting as a flight control layer. Pictures courtesy of RUAG/Kayser-Threde GmbH
Usually, ESA provides sample cells that are open (or vented), towards open space, or semi-tight (presented as RUAG closed cells in this paper). In such a sample accommodation, gaseous compounds produced by the degradation of the exposed refractory molecules are vented out to space. It also makes the irradiation of gaseous mixtures impossible. Therefore the scientific team at LISA, and CNES, have been working on the development of closed cells (called here CNES closed cells) which have been used since 2007 and continuously improved since then.
c1. Open Cells
Open cells used in EXPOSE-E are the same as those used in the Biopan/UVolution experiment presented earlier in this paper. Configuration for EXPOSE-R/-R2 is different. Open cells used in the PSS experiment on EXPOSE-R2 are shown in Fig. 22. Samples are deposited on the inner side of the window (MgF2). Such cells can be used for kinetics studies of solid organic compounds. Although gaseous photoproducts cannot be analyzed because they are vented to space, this is in the end beneficial for accurate kinetic measurements because the gases cannot be photolyzed and interact with the sample as it would be the case in a closed cell.
c2. RUAG Closed Cells
For some of the samples, another configuration is used, called RUAG closed cells (Fig. 23). Two discs separated by a spacer are adjusted inside the sample carrier and kept in place with a bushing. This configuration can be used for semi-volatile samples to prevent their passive outgassing towards space. However, these cells are not tight enough to be considered for use with gaseous samples, or to keep the gaseous photoproducts of solid samples trapped for analysis after return on Earth. Incidentally. this configuration protects samples in case of a contamination event from the inside of the facility. RUAG closed cells can also be used for radiation measurements conducted on samples that are extremely fragile, for example, graphite disks.
EXPOSE-R2/PSS/RUAG closed cells are made of two discs accommodated in a sample carrier with a bushing. The organic sample is deposited onto the inside face of the upper window. A graphite disk can be used in place of the upper window for radiation measurements. Picture courtesy of Kayser-Threde GmbH/RUAG
c3. CNES Closed Cells
CNES closed cells can be used either to study the photolysis of a gaseous mixture (simulation of an atmosphere), to collect the photodissociation products of a solid molecule, in addition to results derived from exposition in open cells, or for the photolysis of a solid molecule during the interaction with a simulated atmosphere (for example a molecule at the surface of Mars).
They have been used in experiments UVolution, PROCESS, AMINO and PSS. The concept has been the same since 2007 but the manufacturing process has been drastically changed since their first use in UVolution and PROCESS. In their first implementation, closed cells were made of two aluminium cylinders screwed one into the other. An MgF2 window was glued at each end of the cell. A Viton O-ring between the two parts was used for tightness (Fig. 19). It has been shown that this first generation of closed cells is not adequate for EXPOSE-like long duration experiments outside the International Space Station (leak level above \(10^{- 8}~\mbox{mb}\,\mbox{L}\,\mbox{s}^{-1}\)), although the level of vacuum-tightness is acceptable for short duration experiments, such as UVolution (14 days in space, and roughly one month between the preparation of the samples, and their analysis after return). Due to time constraints, for PROCESS, first-generation CNES closed cells were used and, unfortunately, some of them were empty when they returned to Earth (from 5 to 100% empty) (Cottin et al. 2012).
For the AMINO experiment, a new kind of closed cell has been designed and manufactured, under the supervision of Air Liquide (DTA Grenoble, France). The windows are now brazed, the main body of the cells is made of stainless steel, and the vacuum-tightness between the two parts is ensuring with laser soldering. Leak level of below \(10^{-10}~\mbox{mb}\,\mbox{L}\,\mbox{s}^{-1}\) can be reached, which is compatible with long duration experiments in orbit (loss \(<10\%\) for 2 years in space) (Fig. 24).
EXPOSE/CNES closed cells. For the first generation of CNES closed cells, two aluminum cylinders are screwed into each other. Two MgF2 were glued at both ends to allow the analysis of molecules inside the cell by spectroscopy. Sealing (relative to lab atmosphere or vacuum in space) was ensured by a Viton O-ring. For the new generation of closed cells used since the AMINO experiment, the body is made of stainless steel, windows are brazed, and sealing is ensured thanks to laser soldering. Picture credit Air Liquide & H. Cottin
Recently, CNES closed cells have been significantly improved with the possibility of including specific optical filters (Fig. 25). This opens up the possibility of turning a cell into a miniature Martian simulator if the samples are deposited below a KBr disk, for instance (Fig. 26a & b). The global architecture of the cell remains the same and does not have to get through a series of space qualification process each time a new filter is chosen. Such a configuration is used in the PSS experiment. In the future, thanks to this configuration, specific studies as a function of the wavelength can be implemented (Fig. 26c).
(a) Irradiance curve in the UV as estimated: 1. at the top of the Martian atmosphere, 2 & 3. at the surface of Mars for various inclination and seasons, 4. at the surface of the Earth (adapted from Cockell et al. 2000). (b) Transmission spectrum of a 2 mm thick KBr window, to be compared with curve (a)-2. (c) Various filters that could be used for wavelength sensitivity studies in future experiments. CaF2 could be used to get rid of the Lyman \(\alpha\) line, as well as different kind of quartz and silica (source: eSource Optics/Whitinsville, MA, USA)
A last configuration of CNES closed cells has been used in order to study the resistance of biochips to space constraints (especially high energy particles and thermal cycling) (Vigier et al. 2013). The biochip detection principle is based on the recognition of a target molecule by specific receptors fixed on a surface. Such sensors are being studied to assess their suitability for planetary exploration since they present great potential for the search for biomarkers thanks to their size (miniaturized devices) and sensitivity. One of the concerns for the development of such an analytical system is the stability of the biological receptors in a space environment. Therefore a series of biochips has been accommodated in CNES closed cells during the PSS experiment. MgF2 windows were replaced by thin stainless steel plates since UV photons are not relevant for those experiments: only energetic particles, thermal cycling and vibrations/chocks at launch and return to Earth are of interest to characterize the stability of the biochips during a journey into space (for instance from Earth to Mars) (Vigier et al. 2013).
c4. Sample Preparation
Solid samples are usually prepared in sublimation chambers under vacuum (Guan et al. 2010; Ten Kate et al. 2005). Their thickness is monitored by interferometry to ensure that they are homogeneously photolyzed in space. Samples which cannot be sublimated (because they are degraded at high temperature) are deposited using an evaporative method. They are first dispersed as a powder in a solvent to prepare a suspension by mechanical agitation and then deposited at the surface of the exposed window. Finally, sample preparation is finalized after the total evaporation of the solvent (Boillot et al. 2002). Additionally, for the recent PSS experiment, some organic solid samples have been prepared by 200 keV He+ irradiation of frozen icy mixtures, directly on the MgF2 windows meant to be included in the open cells (Baratta et al. 2015). These residues simulate organic material in some astrophysical environments as comets.
“Analytical” cells, such as the one shown in Fig. 27, are used to prepare gaseous samples. They are used for filling and sampling the closed cells with gas, and were also meant for infrared analysis of the gaseous content of the cells after opening. This function however has never been proven to be more useful than direct spectroscopic analysis through the closed cells themselves. The analytical cell is made of two main stainless steel parts on the inside of which both parts of a CNES closed cell can be fitted separately. The two analytical cell parts are then adjusted one opposite the other. The gaseous sample can then be introduced inside the analytical cell before the two parts of the closed cell are screwed one into the other, using a manifold connecting one part of the sealed cell to the outside of the analytical cell. The gases are then enclosed inside, and the closed cell be removed from the analytical cell, soldered by laser, and placed onto a sample holder for the experiment in space.
c6. Sample Exposure Principle
For each kind of sample (pure molecule, mixture etc), the following pattern is applied: two cells are exposed (first layer on the left of Fig. 28, exposed to the influence of UV radiation, energetic particles, temperature, vacuum), two cells are placed right below as flight dark control (second layer in the left of Fig. 28, exposed to the influence of transmitted energetic particles, temperature, vacuum). DLR Cologne (Germany), control ground sample cells are stored in vacuum facilities. In a first chamber, they can be either photolyzed by a solar simulator (\(\lambda> 190~\mbox{nm}\) only) or kept in the dark, and have a temperature history similar to the samples in space (transmitted via telemetry for study of the influence of temperature and vacuum). In a second chamber, a last set of two cells are stored at 5°C (to study the influence of vacuum).
Scheme showing the experimental breakdown of EXPOSE samples between the ISS and DLR Cologne, and the environmental conditions to which they are exposed. In the recent case of the PSS experiment a total of 75 samples can be exposed directly in LEO, with 75 dark controls directly below them (A). The same disposition is reproduced in an irradiation chamber at DLR Cologne (B), with an additional series of samples that are kept in vacuum, in the dark, at 5°C (C) (only one sample carrier position shown in the figure)
c7. Sample Analysis
The analysis of the samples can be conducted by VUV, UV-Vis, Raman and IR spectroscopy before launch and after retrieval. From the results, the photochemical reactivity of these compounds is derived and the kinetic details of photochemical degradation can be characterized. Additionally all the gaseous samples can be analyzed by gas chromatography-mass spectroscopy (GC-MS) after retrieval, to compare the UV-exposed and ground control samples in order to identify photo-products and fragments that cannot be unambiguously determined via optical spectroscopy. The study of the samples is completed by additional laboratory experiments including exposure to a UV \(\mathrm{H}_{2}\)-discharge lamp illumination to provide short-wavelength, high-energy UV radiation that simulates interplanetary and interstellar conditions. Finally, it is possible to calculate the photochemical lifetime of the molecules at 1 AU, which can subsequently be extrapolated at other astrophysical environments (such as other heliocentric distances, diffuse interstellar medium, dark clouds).
d. Tanpopo (Sub-theme 3)
Micrometeoroids (also to be known as interplanetary dust particles (IDPs) and micrometeorites) have been proposed to be the major carbon source accumulated on the Earth before the origin of life (e.g. Chyba and Sagan 1992). However, the organic compounds inside micrometeoroids are susceptible to high-energy effects of cosmic rays and solar radiation, because of their small sizes. In order to test the alteration as well as the survivability of pre-biological organic compounds inside micrometeoroids, amino acids and possible amino acid precursors, have been exposed to the space environment on the Kibo Exposure Facility, under strong UV and cosmic ray irradiations.
Organic compounds selected for the exposure experiments for the Tanpopo sub-theme 3 are amino acids (glycine and isovaline), their possible precursors (hydantoin and 5-ethyl-5-methyl hydantoin), and a complex amino acid precursor material synthesized from a mixture of carbon monoxide, ammonia and water by proton irradiation (Takano et al. 2004). All samples were \(^{13}\mathrm{C}\) labeled, in order to differentiate the samples from terrestrial contaminations. The solutions of compounds were \(1.5~\upmu\mbox{L}\) each. They were amino acid water solutions, hydantoin ethanol solutions, and the synthesized complex amino acid precursor. They were added to each well on aluminum sample plates (Fig. 29), and dried in a clean booth. The dried samples ca. 75 nmol each of amino acids and hydantoins, and the synthesized complex amino acid precursor equivalent to 15 nmol glycine, respectively, were embedded in the wells. Then the samples were covered with hexatriacontane (\(\mathrm{C}_{36}\mathrm{H}_{74}\)) in order to prevent sample loss during the exposure experiments and transportations.
Each Exposure Unit had a \(\mathrm{MgF}_{2}\) or \(\mathrm{SiO}_{2}\) (quartz glass) window, and contained two sample plates inside: one for the space exposure and the other for unexposed “dark” control (Fig. 30). Two Exposure Units, each with \(\mathrm{MgF}_{2}\) or quartz glass window, were assembled to the Exposure Panels (Fig. 11b). The Exposure Panels after 1- to 3-year exposure will be returned to the ground laboratories for the analyses of amino acids and other organic compounds using HPLC and GC-MS, one panel per year.
e. O/OREOS-SEVO Payload
The astrochemistry experiment onboard the O/OREOS nanosatellite (see above for details), the SEVO payload, included 22 samples housed in a carousel (Fig. 31), organized in two concentric rings with the one set of optics per ring linked to a common UV-vis spectrometer (Bramall et al. 2012). The carousel brings a given cell into the analysis position, where spectra can be measured using the Sun for illumination. Each cell is sealed and held in an 11-mm-diameter aluminum sleeve, with a wave spring washer to keep it under pressure and maintain the integrity of a pair of indium seals (Fig. 32). Each SEVO sample cell is constructed from a stainless-steel ring (\(9~\mbox{mm}~\mbox{O.D.} \times 4.5~\mbox{mm}~\mbox{I.D.} \times 3~\mbox{mm high}\)), one MgF2 window on the top and one sapphire window at the bottom. Organic samples are deposited in the form of thin films onto the MgF2 windows. The sapphire windows, which are transparent over the 200 to 1000 nm range of the spectrometer, are used on the bottom of the sample cells to block out the VUV component of sunlight that might degrade the optical fibers and optics and because sapphire has superior mechanical strength, which simplifies cell assembly. After film deposition, the windows are cold-welded to the stainless-steel body using indium, hermetically sealing a controlled atmosphere within the cell. Some of the sample cells in the carousel are used as reference blanks, both as spectroscopic references and to monitor any changes in the windows (e.g. the formation of color centers induced by VUV and particle irradiation) or unforeseen chemical reactions with the cell materials.
Diagram showing how staggering of inner/outer sample rings allows the use of a two-fiber bundle to passively interrogate individual cells in O/OREOS-SEVO. When spectra are not being collected, the wheel is parked so that blank locations are over the collection optics, shielding them and the spectrometer from unnecessary UV light. Baffled sunlight sensors are used to trigger the spectrometer so that spectra are only collected if the wheel is turned toward the Sun. These sensors record the solar exposure the samples receive. Radiation-sensing field-effect transistors (RADFETs) measure the total integrated ionizing radiation dosage the samples receive. Figure from Bramall et al. (2012)
(a) View of an assembled reaction cell for O/OREOS nanosatellite. Two optical windows are cold- welded to a stainless steel spacer using indium. An organic film is deposited on the MgF2 window where it is exposed to the full brunt of solar radiation and cosmic rays. Only the center 3 mm of the film are monitored using absorption spectroscopy. (b) The assembled reaction cells are housed inside anodized aluminum sleeves where they are kept compressed by a wave spring washer to prevent the indium from creeping. The total package is only 11 mmi n diameter. Figure from Bramall et al. (2012)
Two measuring positions, one in each ring, contain no sample cell, allowing for the collection of solar reference spectra. The positions of well-known solar features in these spectra are used for wavelength calibration of the spectrometer. The solar reference spectra (in combination with blank sample cells) are also used to identify any color centers that may appear in the window materials. When SEVO is not acquiring data, the sample carousel is parked in a position that exposes all 22 cells to solar radiation while simultaneously blocking solar radiation from unnecessarily reaching the collection optics and spectrometer. Dark spectra are also measured in this position.
SEVO cells are filled with so-called microenvironments that are meant to expose the organic samples to interplanetary or planetary conditions. These microenvironments are not intended to represent actual simulated natural environments and the analogies between them and natural environments are limited. Rather, they are used to establish a set of initial reaction conditions to perform photochemical experiments that may be used to elucidate reaction processes that may occur in natural environments. Four of them were studied during the O/OREOS mission:
-
(1) inert cells, in which organic sample is placed in an inert argon atmosphere;
-
(2) surface cells, where organic molecules are in contact with mineral (\(\mathrm{SiO}_{2}\)) that might have a photocatalytic effect (+argon in the gas phase);
-
(3) atmosphere cells, designed to explore the effects of photochemically induced gas-phase reactions on the stability of organic compounds. These cells include a Lyman-\(\alpha\)-filtering coating on the \(\mathrm{MgF}_{2}\) windows: a 200 nm layer of \(\mathrm{Al}_{2}\mathrm{O} _{3}\) blocks \(\mbox{UV} < 140~\mbox{nm}\). They contain a Mars-analogous atmosphere: \(\mathrm{CO}_{2}\) (10 mbar (1000 Pa)), \(\mathrm{O}_{2}\) (0.01 mbar (1 Pa)), and the balance Ar to a total pressure of 1000 mb;
-
(4) humid cells, to study the impact of water vapor photolysis on the degradation of organics, which may affect the rates and products of reactions in many planetary environments (e.g., an early “wet” Mars). Here again the MgF2 window is coated with a 200-nm layer of \(\mathrm{Al}_{2}\mathrm{O}_{3}\) to act not only as a UV filter, but also to prevent the interaction of the water vapor and its photolysis products with the hygroscopic MgF2 window. Relative humidity in the sample cell filled with Ar is maintained at about 2.1% (at 25°C) using a hydrated salt pair: \(\mathrm{Mg}(\mathrm{NO}_{3})_{2}\cdot 2\mathrm{H}_{2}\mathrm{O}\) and \(\mathrm{Mg}(\mathrm{NO}_{3})_{2} \cdot 6\mathrm{H}_{2}\mathrm{O}\).
The same kind of cells and sample configurations are planned for the OREOCUBE project.
f. MUE: Miller-Urey Experiment
Although this experiment has not yet been transformed into an actual space experiment, the Miller Urey project is discussed in this review as a project related to astrochemistry that reached a rather well advanced stage of development, based on the use of LEO for near-weightlessness rather than for radiation.
Miller-Urey types of experiments have been carried out under a variety of conditions (Cottin et al. 2015a); however, the influence of weightlessness on such chemical reactions as an additional parameter has never been investigated. Comparison between the results from traditional Miller-Urey type experiments and Miller-Urey type experiments performed under weightlessness conditions can confirm proposed scenarios (e.g., the Strecker amino acid synthesis), leads to the identification of new reaction pathways, and provides a better understanding of how organic compounds formed in outer space. Today, the fact that meteorites, and in particular the carbonaceous chondrites, contain organic compounds is very well established (Ehrenfreund and Sephton 2006). The extraction and analysis of organic compounds like amino acids and nucleobases from meteorites has provided important information on processes and conditions prevalent in the solar nebula (Callahan et al. 2011; Cronin and Chang 1993; Martins et al. 2008; Martins and Sephton 2009). Investigating the influence of weightlessness on the Miller-Urey experiment could also simulate conditions in the solar nebula. Under weightlessness conditions thermal convection is suppressed and the formation and disintegration of organic molecules can be tested on the surface of ice. In summary, performing the Miller Urey experiment in space (on the International Space Station) would shed new light on prebiotic synthesis pathways leading to the formation of biologically relevant molecules in the context of the origin of life.
The Miller Urey Experiment (MUE) aimed at investigating prebiotic chemical pathways for the synthesis of organic compounds in the protosolar nebula. This environment could be simulated in vials filled with various gas mixtures and solid particles. A series of 6 vials initially filled either with a mixture of \(\mathrm{H}_{2}\):CH4:NH3 (2:4:4) or \(\mathrm{H}_{2}\):CO:\(\mathrm{N}_{2}\) (6:2:2) would had been used. The particle material (SiO2) and size are those suggested to be present in the solar nebulae, together with other Si-based compounds. The particles could act as surfaces onto which thin water mantles are formed. A high-voltage spark discharge through which particles are repeatedly moved by a shaker (as slowly as possible) injects energy into the system and causes chemical reactions analogous or closely related to the original Miller-Urey experiment. A representative schematic of the proposed experimental set-up is shown in Fig. 33.
Left: picture of a prototype of the MU experiment (Dual Vial Construction). Right: a representative diagram of the proposed MU experiment illustrating the water-ice coated particles, a high voltage spark discharge (energy source), a shaker to slowly move the particles in near-weightlessness and water reservoir system to inject water to coat the particles at the start of the experiment
The three main differences in the proposed MU Experiment in space compared with the original experimental set ups would be:
-
1.
At temperatures below the freezing point of water, water-ice mantles would be formed around dust particles, providing a realistic simulation of the protosolar nebula environment. The lack of thermal convection and its effect on the equilibrium conditions affecting the formation and degradation of complex organic molecules on the surfaces of ice particles (dust grains used for aggregation seeds) cannot be simulated on Earth. On Earth the particles would not remain suspended in the gas mixture.
-
2.
With water only present in the form of ice and vapor at relatively low partial pressures, and with the temperature being much cooler, the hydrolysis of intermediates (e.g. cyanohydrines in the Strecker synthesis) would be far slower than in the Earth-bound system.
-
3.
The reactions in the original Miller-Urey experiment took place at 100°C and in water vapor. In the proposed MU experiment, the temperature will be much lower. The probability is that reaction pathways and mechanisms are significantly different with such a major reduction in temperature and the presence of longer-lived intermediates at the lower temperature and low water vapor pressure may facilitate kinetic control of the product mixture and potentially lead to the identification of new reaction products.
Such an experimental concept could be developed again in the future and pushed toward completion and actual implementation in Earth orbit.
3.2.3 Results
a. EURECA/ERA
Several samples relevant for astrochemistry were exposed during the EURECA/ERA mission. In the experiment “Space Biochemistry”, organic molecules samples such as urea and a mixture of amino acids (glycine:alanine:glutamic acid:aspartic acid—1:1:1:1) were exposed and almost entirely destroyed (\(>95\%\)) during the experiment. It is however not obvious whether the loss of material was due to photolysis or sublimation since it is thought that those samples experienced temperatures well above 50°C (Dose et al. 1996). In the experiment “Photoprocessing of Grain Mantle Analogues”, synthetic organic residues were prepared after the laboratory photolysis of a variety of ice mixtures made of \(\mathrm{H}_{2}\mathrm{O}\), CO, NH3, CH3OH, CH4 and \(\mathrm{C}_{2}\mathrm{H}_{2}\). Those “first generation” residues were subsequently exposed to the actual space radiations on EURECA. Infrared spectra measured before and after the space experiment have shown that the \(3.4~\upmu\mbox{m}\) organic absorption features related to C–H bonds match those of the diffuse interstellar cloud dust better than any other analog to the interstellar organics after the exposure in space. The fact that the final \(3.4~\upmu\mbox{m}\) spectra are quite uniform and not sensitive to the initial ice mixture suggests that the structural characteristics of highly processed organics are only weakly dependent on the initial composition (Greenberg et al. 1995).
b. Salute-6,7, Kosmos 2044, Bion-11 and MIR
Kuzicheva and Gontareva (2003) provide a comprehensive overview about the results derived from the series of experiments by Soviet and then Russian scientist regarding the behavior of nucleoside and phosphate mixtures on the Salute-6,7, Kosmos 2044, Bion-11 and MIR spacecraft. While the experiment conducted outside Salute-6 space station demonstrated the formation of nucleosides from adenine, thymine, ribose and deoxyribose (Khenokh et al. 1979), the next generation of experiment focused on the production of nucleotides from mixtures of nucleoside + phosphates. A comparison between the results obtained during space exposure (Kuzicheva and Gontareva 1999; Kuzicheva and Simakov 1999; Kuzicheva et al. 1989) and ground experiments (Kuzicheva et al. 1996) showed that the most effective process to form nucleotides was the heating of the samples, followed by VUV photons (145 nm) and finally UV light (254 nm).
Regarding the Perseus-Exobiology experiment outside the MIR station, two amino acids (L-leucine (Leu) and \(\alpha\)-methyl L-leucine (MeLeu)), one cyclic dipeptide (L-leucine diketopiperazine (DKP)) and one activated tripeptide (tri-L-leucine thioethylester, (Leu3Set)), all being of one pure enantiomeric form, were exposed to space conditions outside MIR. The aim of the experiment was to study the stability and the resistance to racemization using the two amino acids Leu et MeLeu, detected in the Murchison meteorite, the chemical stability of the peptide bond using the two peptides DKP and Leu3SEt and a possible photochemically-induced polymerization using the activated tripeptide Leu3SEt. The thiol ester activating group was selected since it is considered as important in scenarios related to early metabolism (DeDuve 1998; Huber and Wächtershäuser 1997, 1998).
After 97 days of exposure in Earth orbit, the sample films were recovered and analyzed on Earth. It was shown that the photodegradation of the peptides was more important for the linear molecule than for the cyclic formula, and did not lead to the formation of amino acids since the main photolytic process inferred from mass spectrometry analyses was decarbonylation (loss of CO) and decarboxylation (loss of CO2). The peptide bond destruction seems to have inhibited the oligomerization of Leu3SEt.
c. Biopan (Dust/Organic/Uvolution)
c1. DUST The DUST experiments were conducted on Biopan in 1994 and 1997 (Barbier et al. 1998, 2002). They have been used as precursors to select the best samples for the long duration experiment outside the MIR space station. During DUST 1 experiment, six amino acids detected in the Murchison meteorite (Gly, Ala, Leu, Val, Asp, Glu) were exposed to measure their degradation and racemization kinetics. L-tyrosine was also used to measure any polymerization induced by photolysis. The dipeptide L-alanyl-L-alanine was also selected to test the stability of the peptide bond. After exposure, no detectable trace of D configuration, and hence racemization process could be found after the flight in any of the samples. Aspartic and glutamic acids exposed as free samples were partially decomposed during exposure to solar UV radiation. However, the decomposition was prevented when the amino acids were embedded in minerals such as montmorillonite or kaolinite. The other amino acids samples remained unaffected by the flight. Tyrosine did not polycondense and the dipeptide remained stable (Barbier et al. 1998). During DUST-2 experiment, four amino acids (Gly, Glu, Asp, Tyr), some amino acid esters and two peptides were exposed in LEO. The absence of racemization of the exposed molecules has been confirmed as well as the high sensitivity of acidic amino acids towards UV radiation already observed in the DUST 1 experiment. Important degradation was observed for unprotected exposed samples, and a small protecting effect was observed for samples embedded in thick montmorillonite films (\(\sim5~\upmu\mbox{m}\)). This implies that some kind of protection is needed for the survival of amino acids in space.
c2. ORGANICS
The ORGANICS experiment was flown on Biopan-5 in 2005 (Ehrenfreund et al. 2007). Several polycyclic aromatic hydrocarbons (PAHs) and fullerenes were exposed to a total fluence of \(602.45~\mbox{kJ}\,\mbox{m}^{-2}\) (for photons in the range 170–280 nm). The experiment was a precursor hardware test-flight for the long-term exposure experiment (Survival of organics in space) on the EXPOSE facility on the International Space Station (ISS). For the small fluence that was collected during the BIOPAN V experiment Ehrenfreund et al. (2007) found little evidence of photo-destruction. The results confirmed that PAH molecules are very stable compounds in space.
c3. UVolution
In 2007, during 14 days in orbit, and about 30 hours of direct Sun exposure, the UVolution experiment on Biopan-6 tested with success the exposure of gas mixtures in space in closed cells. Comparisons between the kinetics of photolysis in space and in the laboratory for solid state samples related to comets and Mars have also been achieved. The results show that extrapolation from laboratory simulations to space conditions (typically by taking only the influence of the Lyman \(\alpha\) line at 122 nm into account) are extremely hazardous and can lead to errors by a factor 100 on the kinetics for some molecules (Guan et al. 2010). The case of Martian simulations is more favorable since the lamps simulating the UV flux reaching the surface of Mars (\(\lambda> 190~\mbox{nm}\)) are quite acceptable sources. However, there is still a factor of 2 to 13 difference between space and laboratory results, depending on the molecule remains (Stalport et al. 2010a). Interestingly, UVolution results have shown that, contrary to previous measurements of shielding effects due to organic compounds embedded in minerals, photolysis of organic molecules can be activated by the presence of a mineral analogue of Martian soil. The stability of carbonates has also been studied, but the duration of the exposure was too low to measure significant changes (Stalport et al. 2010b).
d. EXPOSE (Process/Organic/Amino/PSS)
d1. PROCESS PROCESS was part of the payload of EXPOSE-E and exposed a total of 40 solid and gaseous samples during 1.5 year in space from February 2008 until September 2009 (\(\sim1500\) hours of direct Sun exposure). Open and closed cells were used. Measurements regarding the photostability of amino were conducted, and lifetimes calculated (Bertrand et al. 2012; Noblet et al. 2012). In particular, it has been shown that amino acids with a diacid group, such as aspartic acid, were more sensitive to UV radiation than amino acids with alkyl chains. Moreover, the amino acids with a substituted chain, such as valine, were more stable than those with a linear chain, as in the case of aminobutyric acid. As for the DUST experiments, neither racemization nor oligomerization was identified for any of the compounds. Regarding samples related to the Martian environment, molecules selected as interesting targets to be searched for at the surface of the planet (e.g. glycine, serine, phthalic acid, mellitic acid) were totally destroyed after a few tens of hours and proved not to be pertinent molecules to be searched for at the Martian surface (Noblet et al. 2012) The CNES closed cells designed for gaseous samples revealed they were not tight enough for such a long duration to ensure that the mixtures did not leak into space (Cottin et al. 2012). This aspect was significantly improved for the AMINO experiment.
d2. ORGANIC
The objectives of the ORGANIC experiment on EXPOSE-R were (i) to study the photostability of selected PAH and fullerene-type molecules in an interplanetary environment to allow a comparison with space data; and (ii) to allow a quantitative estimation of dissociation regimes for organic molecules that can be extrapolated to different space environments (interstellar medium, interplanetary, Earth atmosphere, etc.) (Bryson et al. 2011). The ORGANIC experiment on EXPOSE-R spent 682 days outside the International Space Station and the fourteen samples (11 polycyclic aromatic hydrocarbons (PAHs) and three fullerenes) received an irradiation dose of the order of 14 000 MJ \(\mathrm{m}^{-2}\) over 2900 h of unshadowed solar illumination (Bryson et al. 2015). Analyses on the returned samples and ground control measurements showed limited spectral changes in most cases pointing to the stability of PAHs and fullerenes under space exposure conditions. However, some molecules have been strongly depleted and the experiments confirm the known trend in the stability of PAH species according to molecular structure: compact PAHs are more stable than non-compact PAHs, and least stable are PAHs containing heteroatoms (Bryson et al. 2015).
d3. AMINO
The AMINO experiment was also part of the payload of EXPOSE-R. As in the ORGANICS experiment, 30 samples were exposed to space. Open and closed cells were used (Cottin et al. 2015b). Samples were made of new amino acids mixtures (Bertrand et al. 2015), and gaseous samples in new cells that have proven to be sufficiently tight. Nevertheless, it appeared that the MgF2 windows VUV transmission of the closed cells was degraded during their industrial production. We have addressed this issue for the new PSS experiment. In addition, the entire EXPOSE-R facility was subjected to significant contamination most probably originating from the facility itself (Demets et al. 2015). This jeopardized the analysis of the results of some of our samples, but not all. Concerning the amino acids, results from PSS were confirmed: resistance to UV radiation depends on the chemical nature of the exposed molecules and species with a diacid group are more sensitive to UV radiation than amino acids with hydrocarbon chain. High resolution mass spectrometry analyses demonstrate for the first time, that some degradation is due to chemical reactions, such as decarbonylation and decarboxylation caused by exposure to UV radiation. The new compounds formed by losing their carboxylic group were then more resistant to UV radiation. The photochemistry of methane was for the first time studied in space in a quantitative manner (Carrasco et al. 2015). The saturated hydrocarbon photoproducts (up to five carbons) could be successfully quantified, but no unsaturated species were detected. As unsaturated molecules are much more reactive, those provide efficient precursors for polymerization and functionalization with nitrogen and oxygen. Their production budget remains a priority to evaluate the capacity of the methane photolytic system to support organic growth in planetary atmospheres.
d4. PSS
PSS is part of the EXPOSE-R2 facility. This is a new use of EXPOSE-R, refurbished with new hardware and samples and, hopefully, in which no contamination event will occur again. The hardware has been upgraded to accommodate more samples (75 exposed samples). Most of them are still related to interstellar medium, meteorites, comets, Mars, Titan, with addition of new subjects such biochips (Vigier et al. 2013).
e. O/OREOS SEVO
The O/OREOS nanosatellite carried two payloads addressing important aspects of astrobiological and astrochemistry research, including organic chemistry in space, adaptation of life to the space environment, planetary protection, and in-situ monitoring technology (Ehrenfreund et al. 2014). The spacecraft and payloads are described briefly above in “O/OREOS Nanosatellite” (Sect. 3.1.g).
The O/OREOS chemical payload, SEVO, supported two dozen samples comprised of thin films of organic molecules, deposited onto UV-transparent MgF2 windows and enclosed in hermetically sealed sample cells (described in detail above in “O/OREOS-SEVO payload sample cells” (Sect. 3.2.2.e)). Changes in the samples were monitored via a miniaturized UV-Vis-NIR spectrometer in order to measure in-situ their photodegradation pathways (Bramall et al. 2012). Because of their astrobiological and astrochemical relevance, films from four different classes of organic compounds were chosen: polycyclic hydrocarbons (PAHs), quinones, porphyrins, and amino acids. Sample cells were filled either with 1 bar of argon or an argon-CO2 mixture (10% CO2). Additional cells also contained a hydrated salt pair providing a 0.3–2.3% relative humidity, dependent on ambient temperature. Results from the SEVO payload showed high photostability of the quinone anthrarufin whereas the molecules of the other three classes degraded, especially in the presence of water molecules, which expectedly accelerated the degradation of the PAH isoviolanthrene, compared to cells with only argon or argon/CO2 (Mattioda et al. 2014). A similar effect was measured for tetraphenylporphyrin chloride (Cook et al. 2014), again with water vapor significantly enhancing the rate of degradation. For both these materials, thin-film reaction rate data and other spectral information measured in situ, in combination with ground-based experiments and modeling, were used to postulate photo-reaction mechanisms pertinent to the SEVO microenvironments and related space environments. Analysis of the spectral data from SEVO demonstrated the power of in-situ, time-resolved measurements to capture degradation kinetics and mechanistic details of sample reactions induced by solar and cosmic radiation.
f. STONE Results
The speeds of atmospheric entry of the Foton capsules were about \(7.6~\mbox{km}/\mbox{s}\). These speeds are the equivalent of a tangential meteor entry, although most meteors enter at much higher speeds, \(\geq 18~\mbox{km}/\mbox{s}\). Mineralogical changes in the samples indicated that the temperature on the ablation surface started, from a few degree Celsius, increased up to 2000°C at the apex and decreased to less than 0°C within a few tens of seconds. Some samples were lost from the Foton capsule during entry (both times the dolerite) but the dolerite on Stone 5 survived and exhibited the fusion crust typical of stony meteorites. Interestingly, no fusion crust was formed on the artificial sediment while the dolostone, sandstone, impact-shocked gneiss and volcanic sandstone produced cream-coloured fusion crusts and the carbonaceous laminite a cream/green-colored crust, very different to the black fusion crusts of the dolerites and stony meteorites.
Per sample, between half and 75% of the rocky material was lost by ablation during entry. The heat of entry also transformed the mineralogy of the sediments (Brack et al. 2002; Brandstaetter et al. 2008; Foucher et al. 2010; Parnell et al. 2011). The carbonate of the dolostone decomposed to CaO and MgO (periclase) with the release of CO2, while CO2 and SO4 were released from the artificial sediment. With respect to the volcanic sandstone, heat metamorphism changed the phyllosilicates into biotite. Thermal dissociation of the carbonate in the carbonaceous laminate resulted in the formation of portlandite (Ca(OH)2).
In both samples containing carbonaceous biosignatures, the kerogen showed signs of thermal maturity resulting in increasing structural maturity (graphitisation) (Foucher et al. 2010; Parnell et al. 2011). While microfossils away from the fusion crust were still identifiable (Foucher et al. 2010; Westall et al. 2011), the biosignatures in the carbonaceous laminite showed preferential loss of thermally unstable compounds and substantial loss of extractable organic matter. Despite the thermal abuse, sufficient biomarker compounds survive to prove the biological origin of the organic matter (Parnell et al. 2011).
3.2.4 Limitations of Current Astrochemistry Facilities
The existing tools for chemistry experiments in Earth orbit are very valuable; however, their capabilities and scientific return are hampered by significant limitations. One of the most critical issues for current studies is the lack of any in-situ analysis of the evolution of the samples as a function of time on almost all the exposure facilities. Only two measurements are available for most of the experiments: one before and one after the exposure. Thus, critical science conclusions from the studies must rely upon assumptions about how the samples behave between the two time points. To date, most of the kinetic modeling of the behavior of samples in space has assumed optically thin samples and first-order decay kinetics (i.e. exponential chemical change) (Guan et al. 2010). Recent in-depth studies of the absorption properties in the VUV range of molecules previously exposed in Earth orbit, such as adenine and guanine, have shown that a first-order kinetics assumption is not valid and that more complex modeling of the evolution of the samples is required to derive the key kinetic constants (Saiagh et al. 2014, 2015). A significant step forward has been achieved with the O/OREOS NASA nanosatellite and the OREOcube ESA project with onboard UV-visible measurements. However, for organics, following the evolution of the samples would in many cases be more informative and provide greater insight with the use of infrared measurements, which convey organic functionality-specific information based on fundamental vibrational mode data in the mid-infrared range (\(4000\mbox{--}1000~\mbox{cm}^{-1}\)).
Another limitation is that the temperature of the samples is not controlled, thus restricting the selection of molecules for the experiments. Compounds that are not sufficiently refractory can sublime if the sample reaches high temperatures (the meaning of “high” depends on the nature of the molecule, but can be as “low” as 40°C for long-duration experiments). Furthermore, to date it has been impossible to study the evolution of samples that must be held at low temperature (pure ice, or ice mixtures, at \(T\) in the 10–100 K range). Such experiments are both important and common in the laboratory, enhancing the understanding of chemical evolution in interstellar and cometary ices, at the surfaces of the icy satellites of the giant planets (Europa for instance), and at the surfaces of Pluto and transneptunian objects.
All space experiments discussed in this paper were conducted in low Earth orbit. In the relatively low-inclination/low-altitude portion of LEO where ISS is located and where most studies have been conducted, as shown in Table 1, samples are not exposed to significant levels of GCRs due to shielding by Earth’s magnetosphere. While total ionizing dose rate can be significant in LEO, particularly with minimal shielding of samples or in “special” LEO regions near the poles or at higher orbital altitudes (Table 1), the exposure experiments were not generally conducted there, with the exception of O/OREOS, whose near-polar, 650-km elevation provided higher dose rates. Yet even in such “special LEO” regions, the high-energy heavy ions (especially iron nuclei) that are an important component of solar and galactic cosmic radiation are very scarce.
Nonetheless, all the orbital environments in Table 1 provide a simultaneous combination of photons and multiple types of high-energy particles that bring unmatched added value to space exposure in comparison to Earth laboratory simulations. For such experiments, a limitation is that accurate, “real-time” dosimetry sensors have not been implemented for both VUV and particles. On current facilities such as EXPOSE, only sensors for UV down to 220 nm are included, while radiation is monitored with both the R3D experiment (Dachev et al. 2015) and passive thermoluminescence dosimeters located beneath the sample carriers to determine the dose levels for maximum shielding (Berger et al. 2015). Due to discontinuous measurements from UV sensors during the EXPOSE campaigns, simulations based on orbits and shadows cast on the samples were necessary to interpret the results (Rabbow et al. 2015b; Rabbow et al. 2012). Appropriate VUV sensors will have to be implemented for future facilities, and strict attitude control and shadowing effect knowledge are necessary to model the VUV-UV dose reaching the samples hosted on satellites such as SORCE (Pankratz et al. 2005; Rottman 2005).
Finally, it is noteworthy that the International Space Station outside environment is a rather polluted one, chemically speaking. The large surface of the station exposed to outer space is continuously outgassing, some experiments inside ISS have to be vented to space now and then, sometimes in close proximity to an EXPOSE facility, and fuel is used near the ISS by the various spacecrafts when docking or undocking, as well as by the ISS itself to maintain its altitude and perform various maneuvers. Combustion products resulting from the use of the various thrusters can create contamination. Therefore, exposed samples must be protected and MgF2 windows have been generally used on most exposure experiments. MgF2 absorbs VUV photons increasingly as wavelengths fall below 120 nm (Fig. 17), and therefore the photochemistry induced by photons shorter than this wavelength cannot be studied with the current facilities. Moreover, samples must be protected not only from chemical pollutants, but also from the strong oxidizing effect of atomic oxygen, which is the dominant species of the Earth residual atmosphere in LEO (\(\sim 10^{8}~\mbox{atoms}/\mbox{cm}^{3}\)) (Fig. 1); MgF2 windows also provide protection. If samples are to be directly exposed to space without any protection, then they should be exposed in an environment where neither propellant and its products, nor residual Earth atmospheric species, alter the results.
3.3 Space Experiments for Biology
3.3.1 Diversity of Samples for Biology Experiments in Space
a. Extremophiles
The study of life on Earth has shown the astounding ability of living systems, the so called extremophiles, to adapt to a vast variety of extreme environments (with regard to temperature, pressure, pH, humidity, salinity, radiation dose, etc.). Such a tremendous capacity provides the scientific perspective for searching life elsewhere (Rothschild and Mancinelli 2001). In order to support space exploration it is necessary to identify which physical extremes that are not found on Earth, constrain life elsewhere. Thus it is imperative to test the endurance of extremophiles under extraterrestrial conditions specific to space and/or planetary or planetary satellite environments. Low pressure down to space vacuum, exceptionally low relative humidity, and in particular highly ionizing and short-wavelength solar UV radiation are quite common in space and on the surfaces of Solar System bodies, and can only partly be simulated due to their complexity (Horneck et al. 2010). In particular it is important to study the response of extremophiles to multiple extremes (Harrison et al. 2013).
In astrobiological space experiments to date, a selection of extremophiles able of drying without dying, the so called anhydrobiotes (Crowe and Crowe 1992), has been exposed to the environmental conditions of space or planetary conditions simulated in space, e.g. to simulated Martian conditions in low Earth orbit. Analysis of the exposed samples was performed after flight, requiring the download of the samples, and in ground-based simulations as well (Rabbow et al. 2012, 2015b).
Upon drying, anhydrobiotes enter metabolic dormancy and resume active metabolism when water becomes available: This is a prerequisite to cope with space vacuum. In addition the desiccation-tolerant organisms tested so far exhibited an extraordinary resistance to radiation, both UV and ionizing radiation: This is a prerequisite to cope with non-Earth radiation environments.
A list of anhydrobiotes exposed to space and/or Martian simulated conditions includes tardigrades, lichens, fungi, yeasts, plant seeds, bacterial spores, non-sporulating bacteria and cyanobacteria.
A common feature for anhydrobiotes is that they accumulate upon desiccation large amounts of the disaccharides, trehalose or sucrose, that stabilize their membranes and macromolecules in the dried status.
Anydrobiotes are considered good models for searching for life on Mars, partly because the evolution and adaptation of microbial life on Earth during the formation of desert areas, it could be similar to what happened on Mars (Davila and Schulze-Makuch 2016).
Anydrobiotes exposed to space and Martian simulations can be divided into two different categories:
(i) Organisms entering the dried, ametabolic state in the vegetative status; while exposed to space and Martian simulated conditions they accumulate damage at every level of the cellular organization. Their survival upon rewetting depends on the capability to restore an active metabolism and repair the accumulated damage.
(ii) Organisms in specialized dormant forms, which enable survival under unfavorable conditions as in the case of bacterial spores and cyanobaterial akinetes, or which are reproductive stages, as in plant seeds, where embryonic plants are enclosed in a protective coat.
Examples of organisms exposed in space (Table 6) are (i) bacteria—Bacillus subtilis, Bacillus pumilus, Bacillus licheniformis, Deinococcus radiodurans, Synechococcus sp., Chroococcidiopsis sp., Anabaena cylindrica, Nostoc commune, Gloeocapsa sp., Bacillus amyloliquefaciens; (ii) archaea—Haloarcula sp., Halococcus dombrowskii, Halorubrum chaoviatoris; (iii) fungi—Penicillium sp., Aspergillus ochraceus, Aspergillus niger, Ulocladium atrum, Cryomyces minteri, Cryomyces antarcticus, Penicillium italicum, Penicillium expansum, Penicillium aurantiogresium, Aspergillus sydowi, Aspergillus versicolor, Geomyces pannorum, Trichoderma koningii; (iv) algae—Chlorella sp., Rosenvingiella sp.; (v) lichens—Rhizocarpon geographicum, Xanthoria elegans, Circinaria gyrosa (renamed form Aspicilia fruticulosa); (vi) animals—tardigrades (Cockell et al. 2011; Horneck et al. 2010; Jönsson et al. 2008). In addition, the bacteriophages T1 and T7, the tobacco mosaic virus, and plasmid DNA (pBR322, pUC19) also have been investigated (Horneck et al. 2010).
b. Microbial communities and symbiosis
On Earth the establishment of microbial communities lead to appearance of the first microbial biofilms, the stromatolites, whereas biological interactions, namely the symbioses, such as the endosymbiotic origin of mitochondria and chloroplasts, or those forming the lichens, have played a key role in the evolution of life (Lyons and Kolter 2015). Interactions are thought to facilitate the endurance of life within extreme environments. Hence, considering that one specific organism may not be able to cope with extreme parameters, various natural microbial communities were selected for space experiments in low Earth conditions. The final aim was to investigate their capability to survive drastic environmental changes (such as the ones that occurred on Mars) in association with others of its own kind (biofilms) or other kinds (ecologies). Space experiments carried out on microbial communities and symbiotic organisms are also relevant to investigate if outer space can act as a selection pressure on the composition of the microbial communities and to discern the limits of symbiosis’ resistance.
Microbial communities collected in extreme terrestrial environments, such as permafrost communities, desert endolithic cyanobacteria, endoevaporites (de la Torre et al. 2010), antarctic cryptoendolithic communities (Onofri et al. 2012, 2015; Scalzi et al. 2012) and epilithic phototrophic biofilms (Cockell et al. 2011; Horneck et al. 2010) as well as a wide selection of lichens (de Vera et al. 2012) collected from extreme environments were exposed to space conditions in low Earth orbit and upon retrieval examined on the ground (Brandt et al. 2015, 2016; de la Torre et al. 2010).
The outcomes of the experiments on the resistance of extremophiles exposed to space and planetary simulation are relevant in the context of Lithopanspermia as well as of Planetary Protection (Horneck et al. 2010). The possibility that organisms could travel and survive on meteorites ejected from Mars to Earth needs to be investigated, as well as the survival strategies of organisms exposed to new environmental parameters not found on Earth (Nicholson 2009). In addition, since it must be assumed that space exploration could mean contamination this is a key point in view of Planetary Protection. Increased knowledge of the extremophile survival strategies and of their limits will consequently lead to the development of improved decontamination procedures.
c. Biosignatures
The biosignatures are related, for instance, to gases of possible biological origin; spectral evidence of biology; complex biomolecules such as DNA, RNA, lipids, proteins; chiral organic molecules; biominerals; morphological features produced by microorganisms and their activities and more (Westall and Cavalazzi 2011; Westall and Cockell 2015).
The search for life or fossil remnants of life on other planets is a prime goal of future missions in the Solar System; it is thus necessary to understand the stability of biomolecules associated to life as we know it. To date, there is no definitive answer to whether biomolecules are resistant to conditions encountered on Mars or on the Jovian and Saturnian icy moons, which are classified as potential habitable places in the Solar System (de Vera and Ott 2010). Studies on the structure and stability of biological components, such as membranes, proteins, pigments, DNA, etc, after their exposure to space and planetary simulations, are thus highly relevant.
On Mars the surface today is more likely to harbor signs of extinct life compared to extant life (Westall and Cavalazzi 2011; Westall et al. 2015). Because nowadays atmosphere is very thin and there is no magnetic field on Mars, the surface is exposed to by different kinds of radiation (both UV irradiation and ionizing radiation) that is considered the main driver for the degradation of organics. For instance DNA quickly degrades under UV radiation and the chances to detect it on radiation exposed surfaces is considered to be very low. However, the results of BIOPAN-campaigns, EXPOSE-E, EXPOSE-R space missions and ground-based simulations pointed out that DNA within a cell or even better in a biofilm, takes advantage of enhanced protection (Baqué et al. 2013). The use of different laser induced spectroscopy methods (Fluorescence-, IR-, LIBS-, mass-, Raman-, UV-VIS-spectroscopy, etc.) was identified as useful to investigate biomolecules that might serve as biosignature references, after being exposed in space and Mars-like conditions (Baqué et al. 2015; de Vera et al. 2012). A key point to be addressed by performing experiments in Mars-like conditions in space is to help avoiding possible pitfalls which might occur due to possible overlapping of abiotic and biotic spectral signatures during spectroscopic detection procedures. In this context it is crucial to study the exposure of macromolecules in contact with Mars regolith analog mixtures reflecting alteration minerals formed during different Martian conditions (de Vera et al. 2012). Hence, the characterization of biomolecules under space conditions is important since it will also contribute the development of life detection instruments.
3.3.2 Hardware for Biology
The environmental parameters and conditions investigated in astrobiological space experiments encompass space vacuum, inert gas atmospheres (N2, Ar), simulated Martian atmosphere and pressure, different temperatures and temperature cycles, extraterrestrial UV radiation or selected UV wavelength bands, simulated Martian UV radiation, cosmic radiation and trapped radiation (in the Van Allen Belts), direct exposure or shielding by meteorite material or Mars regolith simulants, and the presence of cryo-protectants. As described in the chapters for chemical space experiments, it was not possible to expose biological samples to the whole combination of space factors simultaneously in ground simulation or control experiments. This could only be realized by dedicated experiments conducted in space.
Multiple exposure facilities have been used (Table 6) in space for short-term experiments up to two weeks and for long-term experiments up to nearly six years. Common to these facilities was the possibility to expose several dry biological samples simultaneously in small sample carriers beneath optical filters in addition to one or two additional layers of identical samples without exposure to solar electromagnetic radiation including UV. With this arrangement, the wavelength band of extraterrestrial solar radiation was chosen individually for each experiment depending on the scientific question. Space-flight dark controls were investigated under the same conditions. The sample sizes ranged from 7 to 11 mm in diameter and 4 to 10 mm in height. Small stacks of samples were also studied. The key environmental parameters included temperature, UV/solar and ionizing radiation, which were measured during the mission and used to set the levels for ground simulation experiments, which were performed in parallel.
Some examples of the experiments performed are described in the following sections. For the short-term facilities, the hardware of the experiments LICHENS (Lichens as extremophile organisms in space) (Sancho et al. 2007) and LITHOPANSPERMIA (studies of the interplanetary transfer and entry processes of epi- and endolithic microbial communities) are presented (de la Torre et al. 2010). These experiments were conducted on Biopan 5 & 6. For the long-term experiments, the EXPOSE-E and EXPOSE-R/R2 facilities were used as described in Sect. 3.1.e.
LDEF/EURECA/MIR
An early series of experiments in space on microorganisms were performed using the LDEF and EURECA platforms and the space station MIR, while only twice, during translunar trips of Apollo 16 and 17, microorganisms were exposed to space conditions beyond Earth’s magnetic shield, in the MEED (microbial ecology equipment device) facility and in the Biostack experiments (see Table 6).
b. Biopan (SURVIVAL/LICHENS/LITHOPANSPERMIA)
The Biopan facility has been presented in Sect. 3.1.d and Fig. 8. Various kinds of exposure designs have been used over the years to accommodate a large number of experiments related to biology. Only some of them are presented in this section.
b1. SURVIVAL I–III Experiments
The exposure experiments Survival I to III were flown on the 3 successive Biopan missions Biopan 1, a 15 day mission from 29.07. to 17.08.1994, Biopan 2, a 10 day mission from 09.10. to 23.10.1995 and Biopan 3, a 12 day mission from 09.09. to 24.09.1999.
In all 3 missions, the hardware, developed by DLR, Germany, was the same: a total of 61 hexagonally shaped quartz covered sample carriers (Fig. 34) were accommodated in 2 layers in roughly one quarter of the lid of the respective Biopan. Total surface was \(165~\mbox{cm}^{2}\). Each sample carrier had up to 4 horizontal holes for gas escape followed by vacuum exposure. Cylindrical membranes prevented the biological material from a similar escape to space. The sample carriers were 13 mm in diameter, 6.6 mm high with an inner area of 11 mm in diameter and made of aluminum alloy AlMgSi 1 with material number 6082 of the 6000 series according to DIN 1747. The upper layer was exposed to the full space environment of vacuum, temperature cycles, ionizing radiation and extraterrestrial short wavelength UV. The bottom layer was exposed to the same space conditions except the optical radiation (Delonge 1995; Horneck et al. 2001).
The first space experiment with cyanobacteria was performed in 1994 aboard ESA’s BIOPAN-I facility: The halophilic cyanobacterium Synechococcus (Nägeli), inhabiting evaporitic gypsum-halite crusts from a marine intertidal area along the coast of Baja California in Mexico, was exposed for two weeks to space in order to test the hypothesis that gypsum-halite by attenuating UV-radiation enhances survival (Mancinelli et al. 1998). Later, during the BIOPAN-VI mission in 2007 (see experiment LITHOPANSPERMIA), endoevaporitic microbial communities dominated by photoautotrophs obtained from Salar Grande in the Atacama Desert, in Chile and epilithic cyanobacterial communities from coastal lime- stone/sandstone cliffs in Beer, Devon, UK were exposed to the space (de la Torre et al. 2010; Olsson-Francis et al. 2010). During the same space mission the endurance of cyanobacterial resting-state cells, the akinetes, dried onto limestone rocks was tested as well, indeed although akinetes are similar to bacterial spores, until then, there have been no investigations on their survival in space (Olsson-Francis et al. 2009). During the BIOPAN I–II–III missions bacterial spores were exposed to space with the aim of verifying their survivability when covered by thin layers of clay, rock or meteorite material (Rettberg et al. 2004). A follow up of the SURVIVAL experiments, was the MARSTOX experiment designed to investigate the influence of the Martian UV radiation climate on the survivability of bacterial spores in the presence of different types of Martian soil analogues (Rettberg et al. 2008).
b2. LICHENS Experiment
The hardware for the Lichens experiment (Fig. 35) was designed to discriminate, with the greatest possible degree of reliability, the effect of UV radiation from other harmful factors such as high vacuum, rapid temperature changes.
The hardware to which the lichen samples were fixed was designed and constructed by INTA (Spanish Aerospace Establishment, Madrid, Spain). The sample carrier consisted of two sample plates (\(78 \times 50 \times 23~\mbox{mm}\)) for the accommodation of lichen samples (n°2 and 3 in Fig. 36) and a cover plate (\(80 \times 50 \times 3~\mbox{mm}\)), which gave support to screw heads and the optical filters below its circular holes (n°1 in Fig. 36). Each sample plate consisted of 12 cells that contained the lichen samples; vent holes made for a direct interaction with the space vacuum. Ventilation holes passed through all three plates and were left open during the entire space flight. The top layer (n°2 in Fig. 36) allowed for exposure of the lichen samples to the full space environment, which included selected wavelength ranges of extraterrestrial solar UV and VIS radiation. To expose the samples to selected wavelength ranges, each sample cell was covered with an optical filter (13 mm diameter and 2 mm thick). Assembly was as follows: 3 cells with a Suprasil SQ0 quartz window allowing transmission of wavelengths >170 nm; 3 cells with a long pass filter allowed transmission of UV with \(\lambda> 280~\mbox{nm}\); 3 cells with a long pass filter allowed transmission of UV with \(\lambda> 320~\mbox{nm}\); and 3 cells exposed the samples to VIS (\(\lambda> 400~\mbox{nm}\)). The bottom layer (n°3 in Fig. 36) accommodated an identical set of lichen samples, which experienced the same space exposure conditions as those in layer 2 except for exposure to solar UV and VIS radiation. They served as inflight dark controls. For thermal control, the cover plate (plate n°1 in Fig. 36) was painted white (SG-121 FD®) to reflect radiation. For fabrication of the hardware standard space materials were used, ISO AlMgSi, an aluminum alloy, which had a final area of \(78 \times 50~\mbox{mm}\) and a total mass of 250 g.
LICHENS experimental hardware: The lichen samples were accommodated in the 12 cells of plates n°2 and 3. The top plate (n°1) gave support to screw heads. Plate n°2 contained the cells for the flight samples exposed to solar UV and VIS radiation and plate n°3 the cells for the in-flight dark controls. Ventilation holes pass
Cylindrical biological membranes of polysulfon (Berghof und Anlagetechnik GmbH), with a pore size of 100,000 daltons, were positioned inside the sample cells to avoid contamination between the experiments integrated in Biopan. For fixation of the lichen samples in the hardware cells, Scotchweld® and Silicone RTV566® glue mixed with Primer SS41565® (General Electric Silicones Europe) were used. The lichens were exposed to the space environment for two weeks while in Earth orbit aboard the Biopan facility. An identical set of hardware with lichens was built and kept for the same period in the laboratory at ambient conditions (air, 20°C) and in darkness to serve as laboratory ground control (Earth control). During the mission all samples were in an anhydrobiosis state caused by natural dehydration (Sancho et al. 2007).
b3. LITHOPANSPERMIA Experiment
One unit of the Lithopanspermia experiment was part of the payload of the Biopan-6 space mission of ESA. The hardware of Lithopanspermia (Fig. 37) was constructed of aluminum–silica alloy-ISO Al Mg Si (Al6082 T6) and consisted of a top (level-1) and a bottom (level-2) plate, each dimensioned \(146~\mbox{mm} \times 129~\mbox{mm} \times 23~\mbox{mm}\), and each accommodating 36 cylindrical sample cells of 13 mm in diameter and 9 mm in height. The cells of the top plate were covered by optical longpass filters with the following characteristics: (i) MgF2, which is transparent for the complete spectrum of extraterrestrial solar electromagnetic radiation of \(\lambda> 110~\mbox{nm}\); (ii) SQ0 synthetic quartz and appropriate filter transmitting solar electromagnetic radiation of \(\lambda> 200~\mbox{nm}\), thereby simulating the UV and VIS radiation climate on the surface of Mars; (iii) long-pass filter for \(\lambda> 290~\mbox{nm}\) to simulate the terrestrial UV and VIS radiation climate (as a control) and (iv) for \(\lambda> 400~\mbox{nm}\) thereby cutting off all solar UV radiation. Reference samples in the bottom plate were kept in the dark during the whole mission. A channel system was drilled in and between the cells for allowing access of space vacuum to the sample. A tubular membrane (polyethersulfon PES (371WPET12, Berghof Filtrations- und Anlagetechnik GmbH&Co KG, Eningen, Germany)) with a pore size of 100 kD and a diameter of 12 mm was inserted in each cell to prevent possible contamination between adjacent cells through the channels. The samples inside the cells, and the optical filters on top of the cells, were fixed by a mixture of silicone and primer (RTV-576 with primer SS41555).
The hardware (except the optical filters) was covered by a white coated (paint SG121FD) thin plate (\(146~\mbox{mm} \times 129~\mbox{mm} \times 2~\mbox{mm}\)) to reduce temperature excursions and to limit the temperature gradient in relation with the Biopan structure. The mechanical function of this white plate was to clamp the optical filters and the experimental package of Lithopanspermia to Biopan’s mounting plate. Steel bolts (14 M5 bolts) were used to secure Lithopanspermia onto the lid plate of Biopan and to hide the bolts, which fixed the top and bottom plates (6 M5 bolts). Total mass of the experimental package was 636.7 g (de la Torre et al. 2010).
In addition to the Biopan-provided sensors, a thermoluminescence dosimeter (TLD) assembly Litho-Dose (Fig. 37-D) was integrated adjacent to the sample cells, to register the depth dose distribution of cosmic radiation. On the opposite side, one of the AD590 temperature sensors from Biopan was located. The time profile of the radiation dose during the mission was recorded by another experiment on Biopan-6, called R3D (Dachev et al. 2009).
c. STONE
One of the objectives of the STONE experiments was to test the “lithopanspermia” hypothesis, i.e. the transport of living organisms in a rocky habitat between planets (see for instance Horneck et al. 2008). Therefore, in addition to the accommodation described above, two of the STONE experiments also carried “piggy-back” living microorganisms. Dried colonies of the endolithic cyanobacterium Chroococcidiopsis were placed behind the rocks (either 1 cm thick or 2 cm thick for experiments STONE 5 and STONE 6, respectively) as shown in Fig. 38.
d. EXPOSE
The EXPOSE facilities have been presented in Sect. 3.1.e. and Fig. 9. As for Biopan, various kinds of exposure designs have been used over the year to accommodate a large number of experiments related to biology. Only some of them are presented in this section.
d1. LIFE on EXPOSE-E
LIFE experiment (Onofri et al. 2012, 2015; Scalzi et al. 2012) aimed to investigate the fate of lithic organisms and communities during long-term travel in space on the EXPOSE-E facility. Biological test systems of the experiment were rock-dwelling organisms from hostile regions: Antarctic cryptoendolithic (dwelling inside rocks) communities in their natural sandstone, microcolonial black cryptoendolithic fungi (Cryomyces antarcticus and Cryomyces minteri) isolated from Antarctic sandstone (Selbmann et al. 2005), and high mountain epilithic lichens (Rhizocarpon geographicum and Xanthoria elegans). The biological samples were accommodated in wells (1.4 cm in diameter) as shown in Fig. 39. During the space mission they were exposed either to the full space environment (vacuum from \(10^{-7}\) to \(10^{-4}~\mbox{Pa}\), fluctuations of temperature between −21.5 and +59.6°C, cosmic ionizing radiation up to 190 mGy, and solar extraterrestrial electromagnetic radiation up to \(6.34 \times 10^{8}~\mbox{J}\,\mbox{m}^{-2}\)) or they were shielded from insolation. After 1.5 years in space, the samples were retrieved and their viability was investigated. During the mission the sun-exposed LIFE samples had been exposed to 1,879 eSCh (estimated Solar Constant hours) (Rabbow et al. 2012). The samples were kept also in simulated Mars atmosphere and pressure (1.6% Ar, 0.15% \(\mathrm{O}_{2}\), 2.7% \(\mathrm{N}_{2}\), 370 ppm \(\mathrm{H}_{2}\mathrm{O}\), in CO2 at a pressure of \(10^{3}~\mbox{Pa}\)), exposed to simulated Mars UV radiation, cutting-off the spectrum of solar extra-terrestrial electromagnetic radiation at a wavelength of \(\lambda= 200~\mbox{nm}\) by use of optical filters. Some samples were insolated with a reduced irradiance by three orders of magnitude by using neutral density filters, with fluences of \(9.19 \times105~\mbox{J}\,\mbox{m}^{-2}\), below a 0.1% transmission neutral density filter. In addition, dark flight samples were located beneath the insolated ones.
Different viability assays were applied: (a) photosynthetic activity of the lichenized alga (photobiont) of the lichens, (b) colony forming ability of C. antarcticus and C. minteri and isolation of microorganisms from rock fragments (c) fraction of DNA amplified from cells with undamaged membrane (PMA, Propidium-monoazide assay) of C. antarcticus and C. minteri and of cryptoendolithic communities inside sandstone fragments and (d) viability of X. elegans and the fungus of the lichen (mycobiont, cultured without the algal symbiont and dried) by means of vital staining.
d2. ADAPT on EXPOSE-E
ADAPT (Wassmann et al. 2012) was an astrobiological experiment that investigated the capability of microorganisms to adapt to environmental conditions qualitatively and quantitatively different from those of their natural habitat on Earth, as e.g. UV radiation on Mars. The Mars atmosphere determines the Martian UV radiation climate, allowing the energy-rich and biologically harmful UVB and UVC radiation with wavelengths \(>200~\mbox{nm}\) to reach the surface—different from the current terrestrial UV radiation flux. ADAPT experimentally investigated the hypothesis that longer-lasting selective pressure by natural stressors or artificial stressors as e.g. UV radiation of a quality different from Earth´s natural UV radiation and applied in the laboratory, results in evolved microorganisms that are more resistant to the action of further ‘extreme’ environmental factors like e.g. vacuum or cosmic radiation.
Highly resistant microorganisms from very distinct terrestrial habitats were exposed to space (EXPOSE-E tray 1) and Mars (EXPOSE-E tray 2) environmental conditions with respect to pressure and atmosphere, with and without the respective UV radiation exposure. They were accommodated in stacked 2-layer 16 well sample carriers in compartment 1 and in one half of the compartment 3 of each tray 1 and 2, sharing this compartment and consequently the carrier with the experiment PROTECT, as shown in Fig. 40. Selected microorganisms consisted in: i) a natural phototroph biofilm consisting of—amongst others—the two algae Chlorella and Rosenvingiella spp. and the cyanobacterium Gloeocapsa sp., augmented with akinetes of Anabaena cylindrica, vegetative cells of Nostoc commune and Chroococcidiopsis, an epilithic and endolithic photosynthetic cyanobacterium often naturally exposed to high levels of solar UV radiation, ii) Halococcus dombrowskii, an Archaeal isolate from a permo-triassic Alpine salt deposit, and iii) spores from Bacillus subtilis strain MW01, a ubiquitous soil bacterium artificially evolved from an ancestor (strain 168) by repeated exposure to short wavelength Mars-like UV radiation. ADAPT samples MW01 were prepared as organisms dried onto UV-transparent MgF2 (tray 1) or quartz (tray 2) discs. In each sample well, 3 of these discs were stacked on top of each other, separated by Viton rings. To allow depressurization during vacuum (tray 1) or Mars pressure (tray 2) exposure, the rings were opened on one side. Inn this arrangement, the UV shading effect of the upper samples to the lower samples in the stack was investigated. Other Adapt samples were exposed on quarz discs ((ii Halococcus dombrowskii) or sandstone material (I communities) in one layer. Each sample well was closed on top with a MgF2 or quartz window. Additional neutral density filters attenuated the optic radiation for a part of the samples, while cut off filters selected to transmit wavelength >200 nm provided a Mars UV spectrum for the samples in tray 2. The tray was in addition filled with a gas similar to the Mars atmosphere at a Mars similar pressure.
EXPOSE-E integrated on the EuTEF platform before launch (Credit: ESA). The bottom two compartments and the lower half of next compartments contained the sample carriers (explosion drawing on the right, adapted, credit: Kayser-Threde) with the sample from ADAPT. Left vertical row is tray 1, open to space; middle vertical row is tray 2 with Mars conditions
d3. PROTECT on EXPOSE-E
PROTECT (Horneck et al. 2012) investigated the resistance of spores from different bacillus strains collected in space craft assembly clean rooms, exposed on space hardware material to space conditions.
Samples of the experiment PROTECT were arranged in a similar way in compartments 2 and the second half of compartments 3 of both trays. PROTECT samples of spores of cleanroom isolates dried onto discs made of aluminum as space hardware representative material were stacked in the same way as the ADAPT samples. Because Aluminum is not UV transparent, only the top layers of the stacks in the upper carriers were irradiated
d4. BOSS on EXPOSE-R2
The experiment BOSS (Biofilm Organisms Surfing Space) on EXPOSE-R2 (still ongoing at the time of the preparation of this manuscript) investigates the hypothesis that the biofilm form of life supports long-term survival of microorganisms under the harsh environmental conditions as they prevail in space or on Mars and is superior to the planktonic life form. An international consortium of 8 investigators provided the microorganisms Deinococcus geothermalis, Halomonas muralis, Halococcus morrhuae, Gloeocapsa sp. Chroococcidiopsis and Bacillus horneckiae either grown as biofilms or in their planktonic life form for the experiment ADAPT during the ca. 1.5 year exposure mission in LEO. Subsets of the organisms were exposed i) to space conditions, i.e. vacuum and short wavelength UV of \(\lambda> 110~\mbox{nm}\) in tray 1 of EXPOSE-R2, ii) to Mars conditions in a Martian atmosphere, pressure and UV climate with wavelengths \(>200~\mbox{nm}\) in tray 2, or iii) to the respective dark conditions.
The overall EXPOSE-R2 hardware used for the experiment BOSS was similar to the hardware already used in EXPOSE-E for ADAPT and PROTECT. Again tray 1 was dedicated to space conditions, tray 2 provided Mars atmospheric composition and pressure. In both experiment trays, a total of 128 BOSS samples were integrated into sample carriers that partly were stacked in 3 layers providing 16 sample wells each in each of the 3 layers: one top UV irradiation layer and 2 dark layers below, and in similar sample carriers with two layers, shared with a Russian experiment. Filter frames with selected optical filter combinations attenuating the extraterrestrial UV radiation and providing the Martian UV spectrum where appropriate covered the top carriers. The position of the BOSS experimental samples in the EXPOSE-R2 trays and sketches and pictures of the sample carriers are shown in Fig. 41.
Left: experiment distribution throughout trays 1 and 2 of EXPOSE-R2. Top: tray 1 (space exposure), bottom: tray 2 (Mars conditions exposure) (adapted, Credit: ESA). The \(1 \tfrac{1}{2}\) sample carriers for the BOSS experiment are circled in red. Middle: pictures of the two-layer sample carrier with 16 sample wells (top) and the three layer sample carrier (bottom) for BOSS taken during flight sample integration (Credit: DLR). Right: sketches of the corresponding sample carriers including the top filter frames and optical filters (Credit: Kayser-Threde)
d5. BIOMEX on EXPOSE-R2
BIOMEX is a space exposure experiment on the exposure facility EXPOSE-R2 mounted on the Svezda module of the ISS using in parallel simulated space conditions in the ground base facility for reference studies. The experiment was launched on the 24th of August 2014 and ended 18th of June 2016. By this experiment it is planned to obtain information on the stability and degradation levels of space exposed pigments, secondary metabolites, membranes and cell surfaces in contact to a terrestrial, Lunar and Martian analogue mineral environments. In parallel, analysis on viability of the investigated organisms will give relevant data for evaluation of the likelihood of interplanetary transfer of life (theory of Lithopanspermia) and may serve as replicate for so far existing exposure experiments on the ISS but during a different solar activity as it has been tested before.
In this project a mixture of Martian and Lunar analogues as well as terrestrial minerals with lichens, archaea, bacterial biofilms and cyanobacteria, snow alga, meristematic black fungi and bryophytes from alpine and polar habitats is exposed to space and to simulated Mars-like conditions on the ISS (de Vera 2012). This concept is planned to evaluate the degree of degradation of the organisms and their secondary metabolites which might be caused by the investigated space parameters (radiation, vacuum, Mars-CO2-gas). Additionally, expected secondary effects or interactions between life forms and minerals are foreseen to be tested. This concept could be developed in future exposure experiments on the Moon and will serve as pre-tests in low Earth Orbit (de Vera 2012). Samples preparation and integration of the BIOMEX experiment into EXPOSE-R2 are shown in Fig. 42 and Fig. 43.
Sample preparation and analysis procedure for the project BIOMEX: (A) Formation of Mars-analog mineral pellets by pressing the Mars-analog mineral powder which was mixed up with bio-molecules/pigments. After Space- and Mars-exposure on the ISS analysis will be performed by spectroscopy (e.g. Raman, IR, UV-VIS) to check the stability of the bio-molecules characterized as specific biosignatures. (B) Overview on the bio-sample preparation lab-table showing cyanobacteria and snow alga placed and grown on Mars-analog mineral pellets. (C) Sample-holder hardware. The samples were placed within this compartments device before flight
Schematic illustration of the EXPOSE-R2/BIOMEX hardware: (A) overview on the compartments and the different sample holder levels (top layer: exposed directly to solar radiation, bottom layer space-dark control areas). (B) Compartment with place for 32 (\(2 \times 16\)) samples. It is divided into two parts with top (radiation exposure) and bottom layer (space-dark control). In the BIOMEX experiment one of this compartment type is exposed in addition to space/solar radiation to space vacuum and another compartment of this size is exposed to a Martian atmosphere. (C) Passive radiation detectors placed on the bottom of the samples. (D) Compartment with place for 192 (\(3 \times64\)) samples. It is divided into three parts with top (radiation exposure) and two bottom layer (space-dark control). In the BIOMEX experiment this compartment is exposed in addition to space/solar radiation to the Martian atmosphere
Data we will get by these results may serve as efficient steps for characterization of stable biosignatures—an essential step for the future search for life in the universe. This means that BIOMEX will directly support future space missions to Mars such as ExoMars and Mars 2020. By installing a BIOMEX data base with a collection of potential Mars- and space-resistant biosignatures, this could significantly serve as backup for the previously mentioned Mars missions. In parallel, the resistance and survival of microorganisms before and after space exposure is checked and will lead to results which might have relevance to evaluate the likelihood of the theory of Lithopanspermia using the Earth-Mars system as a model scenario. The analytic used methods are done after ground simulation tests and will be done after the space exposure by modern microscopic and spectroscopic measurements (RAMAN-, IR-, UV/VIS-spectroscopy, CLSM, SEM), thermo gravimetric analysis and LIVE/DEAD-tests like germination and growth capacity tests, physiological activity check and fluorescence detection.
e. TANPOPO (Sub-Theme 2)
Kawaguchi et al. (2013) have suggested the possible importance of cell-aggregate as an ark for terrestrial microbes to enable interplanetary transfer. The concept was named “massapanspermia hypothesis”. To investigate the massapanspermia hypothesis, the Tanpopo mission exposes the dried deinococcal cells with various depths in the Exposure Panels for the sub-theme (2) at Exposure Facility of Japanese Experimental Module (Kibo) on ISS. The microbe species Deinococcus radiodurans, D. aerius and D. aetherius have been exposed on the ExHAM-1. Wells of 2 mm diameter on the aluminum sample plates were filled with different amount of deinococcal cells (Fig. 44). The sample depths were about 1, 100, 500, 1000 and \(1500~\upmu\mbox{m}\), respectively. The upper aluminum plates are irradiated by UV and the bottom aluminum plates are dark control. The aluminum plates were placed in the Exposure Unit as shown in Fig. 30 in Sect. 3.2.2.d. The Unit was covered either by a MgF2 or a quartz glass window, which shields UV wavelength less than 150 nm or 200 nm, respectively. The Exposure Units were assembled on an Exposure Panel (Fig. 11a). The Exposure Panels harboring the organic compound samples and the microbial samples will be returned to the ground, one panel per year, after about one, two and three years. After the sample return to the ground, the Tanpopo team will test the survivability of the exposed deinococcal cells by colony formation assay. We will also investigate DNA damage under LEO environment using D. radiodurans DNA repair-deficient mutant strains. In addition to the wild type, mutant strains of D. radiodurans have been exposed: D. radiodurans KH311 (carrying a mutation in the pprA gene: Kitayama et al. 1983; Narumi et al. 2004), D. radiodurans UVS78 (carrying a mutation in the uvrA and the uvdE genes (Moseley and Evans 1983)) and D. radiodurans rec30 (carrying a mutation in the recA gene: Moseley and Copland 1975). After the exposure experiment of dried deinoccocal cells, the Tanpopo team will investigate DNA damage such as DNA double- and single-strand breaks by survivability test, pulsed-field gel electrophoresis, and by quantitative Polymerase Chain Reaction (PCR). The frequencies and spectra of mutation in the rpoB gene and the proteotranscriptomic response of deinococcal cells will be tested.
In addition to Deinococcus spp., survivability of the other two microbes, namely dried filaments of Nostoc sp. KH-01, which show high resistance against heat (Kimura et al. 2015), and the spores of Schizosaccharomyces pombe JY3, will be also investigated.
f. O/OREOS-SESLO Payload
The Space Environment Survivability of Living Organisms (SESLO) experiment collected data on the survival and metabolic activity of microbes at 3 times during the 6-month mission (Nicholson et al. 2011): \(t = 2\) weeks, 3 months, and 6 months after orbital deployment. The biological payload includes three “bioblock” modules, one per measurement time point. Each bioblock has twelve \(75\mbox{-}\upmu\mbox{L}\) bioculture sample microwells connected by microfluidic channels and solenoid-operated valves (The Lee Co., Westbrook, CT, USA) allowing the introduction of germination-and-growth medium; a cross-sectional view of a single microwell is shown in Fig. 45 at left. Four views (two solid models and two photographs) of one integrated 12-well bioblock are also shown at right in Fig. 45. Growth medium was introduced from the integrated storage reservoirs at either end of the bioblock with the assistance of a miniature air pump (KNF Neuberger, Trenton, NJ, USA) that supplied pressure to an elastomeric membrane separating the liquid medium from the pressurized air. Over the multi-hour growth period of B. subtilis, the pump was activated periodically for short durations to maintain a slight positive pressure within the fluidic wells, thereby replacing small amounts of water lost via evaporation through the hydrophobic membrane.
Left: Cross-sectional view of a single \(75\mbox{-}\upmu\mbox{L}\) microwell and the associated fluid delivery, thermal control, and optical measurement components. Right: Four different views (solid model at left, photographs at right) of one of three bioblocks included in the O/OREOS-SESLO payload. The two solenoid valves protrude down from the block (upper left); reservoirs are the elliptical structures at both ends of the block; heater is orange; radiation-sensitive FET (RadFET), which measured integrated radiation dose at two locations in the SESLO payload cubes, is a black dot near the center of the bottom left
Using 3-color LED illumination (470, 525, and 615 nm) together with an intensity-to-frequency optical sensor (ams-TAOS USA, Austin, TX, USA) for each microwell, the growth and metabolism details of Bacillus subtilis microbial spores, exposed to the microgravity and ionizing radiation environment of the high-inclination orbit, were determined. Because the growth medium contained 0.1x alamarBlue (ThermoFisher Scientific, Waltham, MA, USA), a metabolic indicator dye, its blue-to-pink-to-colorless transitions were recorded at the three LED wavelengths, providing a quantitative measure of the metabolic kinetics of the microbial culture in each microwell.
3.3.3 Results
a. LDEF/EURECA/MIR
Spores of Bacillus subtilis 168 in the biological dosimeter “biofilm” on board the MIR station were used to quantify the exposure of cosmonauts to extraterrestrial UV radiation during “sunbathing” at a quartz window (Rettberg et al. 1998). It was found that the solar UV radiation penetrating through a quartz window of the MIR station was a health hazard to the cosmonauts because it contained too much biologically harmful UVC and UVB radiation. EURECA allowed the exposure of microrganisms for a nine-month sun-pointing mission and provided exposure to solar UV radiation for six months. Cells of the desiccation-resistant bacterium Deinococcus radiodurans were killed after a nine-month exposure to space vacuum (Dose et al. 1995). On the contrary spores of the B. subtilis survived, especially when exposed to space as thick layers (Horneck et al. 1994). The longest exposure of microorganisms to space was achieved during the LDEF NASA mission (1984–1990). After nearly six years in space vacuum—the record of space exposure reached so far—1 to 2% of B. subtilis spores dried as a monolayer survived (Horneck et al. 1994), while their survival was significantly increased if protecting substances, such as sugars, were added and when they were dried as multilayers in the presence of 5% glucose (Horneck et al. 1994).
b. SURVIVAL I–III, LICHENS and LITHOPANSPERMIA on Biopan
Different methods applied to detect the vitality of the investigated organisms have shown a wide and significant level of survival after being exposed to space conditions (Meessen et al. 2013; Olsson-Francis and Cockell 2010).
b1. SURVIVAL I–III Experiments
The deleterious effects of extraterrestrial solar UV radiation on the survivability of bacterial spores of Bacillus subtilis was confirmed during the BIOPAN I–III missions (Rettberg et al. 2004). The results expanded earlier findings shown by previous exposure experiments carried out on thin and thick layers of bacterial spores by using the ES029 facility on-board the Spacelab 1 (Horneck et al. 1984a, 1984b).
The BIOPAN I–III missions pointed out the protective role of thin layers of clay, rock or meteorite material when they are in direct contact with the spores (see for a review Rettberg et al. 2004). It was shown that bacterial spores were killed within seconds when exposed to space without any protection against solar UV. Whereas, if embedded in artificial meteorites they were efficiently protected, thus supporting the Lithopanspermia hypothesis. Likewise, crystalline salt provided sufficient protection to osmophilic cyanobacteria during two weeks of space exposure (Mancinelli et al. 1998). After the BIOPAN-VI mission only a single Gloeocapsa-like cyanobacterium was isolated out of the microbial communities from coastal limestone cliff in Beer (Olsson-Francis et al. 2010). Whereas a proportion of cyanobacterial akinetes survived Mars simulated conditions in LEO, only if not subjected to UV radiation (Olsson-Francis et al. 2009).
In the SURVIVAL I–III experiments, spores of three Bacillus subtilis strains that differ in their DNA repair capacities were exposed to the harsh space environment in the lid of the Biopan facilities of 3 successive Foton missions. Strain HA101 was the wildtype strain with respect to DNA repair, while strains HA F was defect in DNA polymerase required for DNA repair and TKJ 6312 was deficient in excision repair and spore photoproduct repair. The spores were exposed to space either unprotected or protected by clay from Adendorf, Germany, red sandstone from Heidelberg, Germany, material from the meteorite Millbillillie, probably derived from the asteroid Vesta, Mars regolith simulant MRTE and material from the Martian meteorite Zagami. Previous vacuum experiments had shown that glucose protects spores; hence glucose was also added as protectant to the space experiment. \(5\times 10^{7}\) spores per sample were either mixed with loose powder of the above materials, beneath a layer of powdered rock or soil, as dry layers of spores on the inner side of the carrier covering quartz plates or on their outside, or prepared as dry layers of mixtures of spores and powder. After launch, BIOPAN was opened and all samples were exposed to space vacuum and the temperature fluctuations of the BIOPAN lid. The samples in the upper sample carrier layer were additionally exposed to extraterrestrial solar UV radiation. After return, the samples were analyzed with respect to their colony forming ability (survival). Unprotected spores exposed to extraterrestrial UV were inactivated thoroughly, irrespective if they were on the outside or inner side of the quartz discs. Thin layers of clay shadowing the samples could not prevent the inactivation. Similar samples in the dark layer survived up to nearly 100%, similar to the laboratory controls not exposed to space at all. This result excluded any toxic effect in the dark. When spores were exposed to UV in dry mixed layers with the materials, survival was much better by more than 5 orders of magnitude. UV irradiated spores mixed with powders of the rocky materials survived better than those mixed with glucose, except for those mixed with Millbillillie. Spores that were exposed to UV in similar rates as spores in terrestrial soil were protected best, with survival rates similar to their dark controls. Results of the 3 Missions are presented and discussed in detail in Horneck et al. (2001); Rettberg et al. (2004).
In the MARSTOX II experiment by using exterrestrial UV radiation and cut-off filters the photoprotection and potential UV-phototoxicity of different minerals of the Martian soil were investigated. Results suggested that bacterial spores are protected by the investigated martian soil analogues that do not produce phototoxic effect (Rettberg et al. 2008).
b2. LICHENS Experiment
In the experiment LICHENS, analysis by LIVE/DEAD staining investigations were performed with the Confocal Laser Scanning microscope (CLSM, de Vera 2012; Onofri et al. 2012), by checking the germination and growth capacity (e.g. colony forming unit counts, development of hyphae, sporulation, de la Torre et al. 2010) and by checking the metabolic activity such as tests on photosynthetic activity and gas exchange (de la Torre et al. 2010; Sancho et al. 2007, 2008). Measurements have shown that the tested microorganisms in their desiccated state survived in many cases much more than 50% after exposure to space conditions. The results have relevance for the lithopanspermia hypothesis. The experiments shown that transit of microorganisms through space is possible in a time-frame between 10 days and in maximum 1.5 years. The question remains still open, if longer time scales up to millions of years with space exposure can still be survived so that a successful transfer can be expected.
The experiment LICHENS was aimed at establishing, for the first time, the survival capability of lichens exposed to space conditions (Sancho et al. 2007). In particular, the damaging effect of various wavelengths of extraterrestrial solar UV radiation was studied. The lichen samples belonged to the bipolar and cosmopolitan species of Rhizocarpon geographicum and Xanthoria elegans, which were collected above 2000 m in the mountains of central Spain as well as endolithic communities inhabiting granites in the Antarctic Dry Valleys. Lichens were exposed to space in the Biopan-5 facility of the European Space Agency. The lichen samples were launched from Baikonur by a Soyuz rocket on May 31, 2005, and were returned to Earth after 16 days in space, at which time they were tested for survival. Chlorophyll fluorescence was used for the measurement of photosynthetic parameters. Scanning electron microscopy in back-scattered mode, low temperature scanning electron microscopy, and transmission electron microscopy were used to study the organization and composition of both symbionts. Confocal laser scanning microscopy, in combination with the use of specific fluorescent probes, allowed for the assessment of the physiological state of the cells. All exposed lichens, regardless of the optical filters used, showed nearly the same photosynthetic activity after the flight as measured before the flight. Likewise, the multimicroscopy approach revealed no detectable ultrastructural changes in most of the algal and fungal cells of the lichen thalli, though a greater proportion of cells in the flight samples had compromised membranes, as revealed by the LIVE/DEAD BacLight Bacterial Viability Kit. These findings indicate that most lichenized fungal and algal cells can survive in space after full exposure to massive UV and cosmic radiation, conditions proven to be lethal to bacteria and other microorganisms. The lichen upper cortex seems to provide adequate protection against solar radiation. Moreover, after extreme dehydration induced by high vacuum, the lichens proved to be able to recover, in full, their metabolic activity within 24 hours.
b3. LITHOPANSPERMIA Experiment
The LITHOPANSPERMIA experiment (de la Torre et al. 2010), which was launched in 2007 with the European Biopan-6 facility for a 10-day spaceflight on board a Russian Foton retrievable satellite (Foton M3). LITHOPANSPERMIA included for the first time a vagrant lichen species Circinaria gyrosa (renamed from Aspicilia fruticulosa) from Guadalajara steppic highlands (Central Spain), as well as other lichen species, exposed previously at the experiment LICHENS, but collected from different habitat locations as there are the high central Alps in Switzerland at altitudes between 2000 and 3000 m, where a higher UV fluence is expected compared to lower altitudes. During spaceflight, the samples were exposed to selected space conditions, that is, the space vacuum, cosmic radiation, and different spectral ranges of solar radiation (\(>110\), \(>200\), \(>290\), or \(>400~\mbox{nm}\), respectively). After retrieval, the algal and fungal metabolic integrity of the samples were evaluated in terms of chlorophyll a fluorescence, ultrastructure, germination and growth capacity of ascospores, physiological activity by CLSM analysis and the use of LIVE/DEAD staining dye and CO2 exchange rates (de la Torre et al. 2010). Whereas the space vacuum and cosmic radiation did not impair the metabolic activity of the lichens, solar electromagnetic radiation, especially in the wavelength range between 100 and 200 nm, caused reduction ofchlorophyll a yield fluorescence; however, there was a complete recovery after 72 h of reactivation. All samples showed positive rates of net photosynthesis and dark respiration in the gas exchange experiment. Although the ultrastructure of all flight samples showed some probable stress-induced changes (such as the presence of electron-dense bodies in cytoplasmic vacuoles and between the chloroplast thylakoids in photobiont cells as well as in cytoplasmic vacuoles of the mycobiont cells), the conclusion was that C. gyrosa, R. geographicum and X. elegans were capable of repairing all space-induced damage. Due to size limitations within the LITHOPANSPERMIA hardware, the possibility for replication on the sun-exposed samples was limited. These first results on the resistance of the lichen symbiosis C. gyrosa to space conditions and, in particular, on the spectral effectiveness of solar extraterrestrial radiation were considered preliminary in contrast to the results obtained for the lichens R. geographicum and X. elegans, which were replicates to the previous experiment LICHENS but much more precisely analyzed using other life activity parameters such as germination and growth capacity of the ascospores and LIVE/DEAD analysis by CLSM using different fluorescence dyes. Further testing in space and under space-simulated conditions was performed (de Vera 2012; de Vera and Ott 2010; Sanchez et al. 2012). Results of these studies indicate that the quest to discern the limits of lichen symbiosis’ resistance to extreme environmental conditions remains open.
c. STONE
Regarding the STONE 6 panspermia survival test of cells of the cyanobacterium Chroococcidiopsis, inoculated into a rock at a depth of 5 mm in order to mimic an endolithic community, the heat of entry was too high for their survival (Cockell et al. 2007; Foucher et al. 2010) calculated that at least 5 cm of rocky protection would be necessary to shield living organisms during entry. It was thus concluded, that in the context of lithopanspermia experiment, epilithic and endolithic photosynthetic organisms would be destroyed by atmospheric transit, whereas chasmoendolithic organisms, inhabiting deep fractures might escape ablation, though the fracture might allow the heat propagation to the cyanobacteria hidden within. However, motile photosynthetic organisms, temporarily situated deep within a rock, might escape ablation of the surface and move back into a more favorable light regimen following interplanetary transfer (Cockell et al. 2007).
d. EXPOSE
d1. LIFE on EXPOSE-E After exposure to space conditions, the lichen X. elegans showed by photosynthetic activity with a PSII activity of 45 (\(\pm2.50\%\)). Resistance of X. elegans and its mycobiont and photobiont was confirmed by vital staining and Confocal Laser Scanning Microscopy, and growth tests (Brandt et al. 2015, 2016). R. geographicum showed lower values. Black Antarctic cryptoendolithic fungi lost colony-forming ability after exposure to full insolation (\(\lambda>110~\mbox{nm}\); 100% insolated samples), but the viability of culturable cells was 12.5 (\(\pm4.11\%\)) for C. antarcticus and 0.46 (\(\pm0.24\%\)) for C. minteri, when 0.1% insolated.
The PMA assay showed about 80% of DNA amplified from cells with undamaged membranes of C. antarcticus (100% insolated), in comparison to total extracted DNA. Thirty-five (\(\pm0.15\%\)) of fungal cells with undamaged membranes (PMA assay) was accomplished by an Antarctic sandstone sample that had received the full influx of solar electromagnetic radiation. From exposed sandstone, fungal colonies with a pale pink mycelium (Acarospora sp.) and one colony of a green unicellular alga (Stichococcus sp.), have been isolated.
The survival of cryptoendolithic organisms is of special interest in terms of Lithopanspermia, because rocks may supply an additional external protection to face the impact-driven ejection into space (Horneck et al. 2008) and transfer from one planet to another.
In Mars conditions simulated in space (100% solar electromagnetic radiation at \(\lambda> 200~\mbox{nm}\)), the viability of black Antarctic fungi cells was 0.8 (\(\pm 0.18\%\)) for C. antarcticus and 0.30 (\(\pm0.02\)) for C. minteri. In the PMA assay, amplified DNA from cells with undamaged membranes was 66.32 (\(\pm6.75\%\)) and 45.66 (\(\pm 1.07\%\)), respectively (Onofri et al. 2012, 2015; Scalzi et al. 2012).
d2. ADAPT on EXPOSE-E
After return of the experiment samples from the 1.5 year space mission to the laboratories of the international ADAPT investigators, the different adaptation and survival strategies were analyzed.
In laboratory experiments, the Bacillus subtilis strain MW01 vegetative cells showed an increased UV resistance to short wavelength UV radiation as e.g. on Mars compared to the ancestor strain 168. Therefore, they are an interesting candidate organism for future astrobiological mission focusing on adapted active life forms.
However, spores derived from the novel B. subtilis MW01 strain and exposed to space and Mars conditions in LEO on EXPOSE-E or to similar conditions in the parallel Mission Ground Reference experiment showed no clear advantage or disadvantage compared to the ancestor strain 168 with respect to UV, vacuum or Mars atmosphere and pressure. Exposure of the respective spores to on-ground x-ray or heavy ion radiation fitted to these results.
Complementing the data derived from other space exposed microorganisms, the spores of B. subtilis MW01 were not highly susceptible to space vacuum resulting in an inactivation of not more than 1 order of magnitude. Martian atmosphere and pressure even led to no detectable effect. When exposed to space or Mars UV, the spores were inactivated by 4.5 and 1–2 orders of magnitude respectively, showing that Martian environmental conditions are less harmful for these spores than space conditions (Wassmann et al. 2010, 2011, 2012).
From the augmented natural phototroph biofilm, the two algae (Chlorella and Rosenvingiella spp.), the cyanobacterium Gloeocapsa sp. and two bacteria associated with the natural community survived after exposure to space vacuum only, meaning cynaobacteria and algae multiplied in liquid medium when brought back to Earth. Of the augmented organisms, cells of A. cylindrical and Chroococcidiopsis survived, but no cells of N. commune.
The effect of space or Mars UV on the augmented biofilm samples was even more severe: only cells of Chroococcidiopsis were cultured from these samples after return to ground. Surface cells were bleached and their carotenoids destroyed (Cockell et al. 2011).
The Archaeal Alpine H. dombrowskii isolate seems to be viable, but investigations on culturability are still ongoing due to extremely increased incubation times (personal communication).
d3. PROTECT on EXPOSE-E
Survival of spores of Bacillus subtilis 168 and Bacillus pumilus SAFR-032 was investigated after the mission by their colony forming ability. The spores exposed to space conditions in tray 1 of EXPOSE-E resembled spores on the “trip to Mars”, while the spores exposed to Martian conditions experienced a simulated “Stay on Mars”. For both groups, the extraterrestrial space UV radiation and the Mars simulated UV radiation were the most deleterious space parameter. Spores exposed in multilayers survived better than their counterparts exposed in monolayers (Horneck et al. 2012).
Bacillus subtilis 168 spores were analyzed after return with respect to the mutagenic efficiency of space using the rifampicin resistance (RifR) and sporulation deficiency (Spo−). Samples exposed to both, space (tray 1) and Mars (tray 2) conditions showed highest increase of mutations up to nearly 4 orders of magnitude when they were irradiated by the respective UV regime, but also when exposed to all space parameters but in the dark. While for the flight induced RifR mutants C to T transitions were localized in one hotspot. Data are published in Moeller et al. (2012).
e. O/OREOS SESLO Results
The biological payload, SESLO, monitored the metabolism, growth and survival of Bacillus subtilis spore-forming bacteria under the influence of near-weightlessness and cosmic radiation. Two B. subtilis strains were studied: a wild-type form of strain 168 and an ionizing-radiation-sensitive strain (WN1087) with a mutation that effects its DNA repair system; it is otherwise identical to the wild-type form (Nicholson et al. 2011). Both strains were deposited as spores in microwells of the SELSO payload. Growth medium supplemented with Alamar blue viability dye was pumped into those microwells at three time points (14 days, 97 days and approximately 180 days after launch) in order to stimulate germination and growth. Colorimetric changes of the Alamar blue dye was measured via 3-color LED illumination and a light sensor, thereby tracking bacterial metabolism, growth, and viability. Non-motile cells in near-weightlessness, in unagitated culture, generally grow or metabolize more slowly than those subjected to Earth gravity due to the absence of gravitationally driven thermal convection and, indeed, the SESLO experiments showed that B. subtilis had slower metabolism and growth in near-weightlessness than those in an identical control experiment on Earth. Surprisingly, the mutant strains metabolized more rapidly than the wild-type in near-weightlessness (Nicholson et al. 2011).
3.3.4 Limitations of Current Biology Facilities
The exposure experiments performed so far in space have given new insights into the limits of survival for terrestrial organisms. However, as mentioned above, the investigations were restricted to dry biological samples, which were analyzed after flight on ground. In these samples active metabolism during exposure is not possible. Therefore, the metabolic processes of adaptation to the space environment cannot be investigated in situ. Especially near-weightlessness effects and their interaction with other space effects cannot be approached. The option to investigate actively growing organisms or to take aliquots of a sample and to analyze it in orbit after different periods of time is necessary. This requires the development of new hardware with different sensors for on-line monitoring. Moreover only small samples could be accommodated in the previously used hardware. For some scientific questions, e.g. for the investigation of the response of whole communities, larger sample sizes will be helpful.
Usually, space experiments are proposed by a group of scientists with the same scientific questions and different model systems. The available space in the hardware and subsequently the number, size and biomass of the samples is very limited. For obtaining meaningful results in biological experiments it is absolutely necessary to have at least three identical replicates for robust statistical analysis. The number of identical samples should be larger and/or the exposure experiment should be repeated independently in space.
The time period from handover of the biological samples for integration in the experimental facility and the actual start of the experiment in space as well as from the end of the experiment in space to handover to the scientist after the mission is quite long and can be several months. This is restraining the choice of the sample. If actively metabolizing organisms will be investigated in the future this time period has to be shortened significantly.
The temperature for the exposure experiments cannot be controlled to a degree required for an increased science return by cooling and heating devices. For systematic investigation of cellular repair and adaption processes the temperature has to be controlled strictly to maintain a constant predefined value. Moreover, studies relevant to the Martian surface, or even further in the Solar System, the environment of Europa, would require low temperature for long duration. This will have to be implemented in the future exposure facilities.
The previously described sample type and size requirements show the necessity of the development of new hardware with different sensors for on-line monitoring. In the case of monitoring metabolic activity of microorganisms the new hardware has to implement a set of sensors measuring besides the metabolic activity, gene expression and growth capacity also temperature, humidity, radiation, pressure, gas composition and pH values in parallel. These sensors have to resist both short duration and long duration space experiments. Besides the mentioned sensors it is also necessary to use a set of spectroscopic devices for the biomolecule detection in space. Space influenced changes of biomolecules defined as biosignatures have to be also monitored in situ in space on the organisms themselves and as isolates. Possible instruments besides the mentioned IR-/UV-VIS spectroscopic methods in the previous chapters might be the Raman spectroscopy and new generation of Fluorescence spectroscopy (LIF). Measuring and monitoring over longer time scales might also give information on transformation or degradation of bio-molecules/biosignatures due to the different stage of the life cycle of the observed and monitored organisms (see Serrano et al. 2014). Data obtained by these methods of a new generation of instrument payloads in LEO or the new proposed orbits around the Earth and the Moon will be an important backup for future space missions to Mars and the icy satellites of Jupiter and Saturn where these instruments will be used for searching extinct and extant life. A welcome side effect is also the fact that the new proposed space mission instruments can be tested directly in the space environment.
Finally, photobiological investigations as those in the space exposure facilities are based on the exact knowledge of the applied radiation fluence. In some of the previous exposure experiments, such as EXPOSE-E and -R, no continuous data about the UV radiation are available due to technical reasons. To compensate, model calculations have been performed in some cases. However, real time measurements are absolutely necessary for data analysis and interpretation.
4 Future Astrobiology Facilities
Astrobiology experiments using space as a tool to carry out studies that cannot be performed in Earth laboratories have been conducted since shortly after the start of human spaceflight. The experiments and results presented in this paper show why such facilities are important and what we can learn from them.
Currently, most astrobiology experiments are conducted on the EXPOSE facility on the ISS. Since 2007, the Biopan program has no longer been supported by ESA. However, the two facilities are not strictly equivalent: Biopan enables short-duration exposure periods with quick access to the facility before the launch (one to two weeks) and rapid recovery after landing (a few days) for analysis. In contrast, access to the EXPOSE/ISS requires delivery of the experiments a few months before launch and hence even longer until the actual exposure starts. For some experiments or samples, this is too long (especially for fragile microorganisms). The duration of sample exposure on the EXPOSE facility generally does not exceed 1.5 years, with no possibility to monitor the samples during the experiment; these limitations hamper the study of some sample types. For instance, while it is possible to conduct an experiment with glycine on Biopan in order to derive a lifetime for this molecule, it is not possible on EXPOSE because the entire sample is destroyed after an unknown time of exposure that is less than the shortest available time on the ISS (Cottin et al. 2012). With the end of the ESA support of Biopan, STONE experiments were also discontinued, with no equivalent replacement.
For a number of reasons outlined in this article, “classical” passive exposure facilities are now reaching crucial limitations that can and should be removed by new technological advances. The scope of this section is to suggest technologies and facilities that can be realized, without any major new inventions, within 10 years. These improved facilities should lead to answers to key questions and hypotheses in astrobiology. Focus is particularly on small-satellite concepts that can provide access to various space environments not available for ISS-based experimentation.
4.1 Astrobiological Science Drivers
Key astrobiological science drivers and hypotheses partially or entirely unanswered should drive the development of new instrumentation.
-
Combine photolysis and particle irradiation on appropriate orbits
Despite years of studies on Earth or in space, our understanding of the chemical evolution from the simplest building blocks in the Solar Nebula or in planetary atmospheres (CO, CH4, \(\mathrm{N}_{2}\), NH3, etc.) toward complex molecular structure has to be improved, due to the lack of facilities simulating both photolysis and particle irradiation. Experiments conducted in space should therefore be implemented on orbits favorable for this dual-category energy input; they would require in-situ monitoring tools for a proper investigation of the evolutionary mechanisms leading to complex end-chain products.
-
Monitor the evolution of samples in situ
Proper assessment of the abundance of complex organic molecules (ranging from a few carbon atoms such as in glycine or adenine, to several tens or hundreds as in PAHs or kerogens) in various astrophysical environments requires knowledge of their stability and destruction kinetics. Such studies cannot be done correctly on the basis of only two measurements (before and after). Therefore experimental facilities similar to O/OREOS and OREOcube, enabling in-situ kinetics studies, should be developed for the ISS and for free flying spacecraft, providing measurement systems that extend analysis to the mid-infrared range, which is more suitable to decipher organic complexity. The mid-IR spectral range is accessible using multiple approaches to IR spectroscopy, or via Raman spectroscopy, which utilizes visible light to obtain vibrational spectra over the same energy range.
-
Simulate icy environments
Most of the organic chemistry beyond the orbit of Jupiter and in the interstellar medium occurs in the ice phase (temperature at the surface of Europa for instance is about 120 K). Moreover, studies relevant to the survivability of microorganisms in the Martian environment or in the ices of Europa also have requirements for low temperatures. Therefore, the capacity to conduct experiments at low temperature is critical for future chemical and biological experiments.
-
Simulate the evolution of active organisms in space
From a planetary protection perspective, studies involving microorganisms and dealing with the evolution, distribution, and future of life are based on knowledge of the capacity of terrestrial organisms in multiple extra-terrestrial environments to adapt and survive over the long term. Experiments investigating cellular mechanisms of adaptation and evolution should be performed under conditions of active metabolism, which means that samples must be exposed in a sustainable environment that supports activity, growth, and evolution (with appropriate sets of nutrients, liquid water, and dissolved gases); in-situ measurement of the cellular processes are necessary as well. As for astrochemistry experiments, the combination of photon and particle radiation is critical for improving science return compared to ground experiments: orbits other than LEO must be available.
-
Simulate the atmospheric entry of meteorites
Meteorites are important for the study of the origin of life. Knowing how atmospheric entry can alter them is of utmost interest, especially in the case of sedimentary rock samples from Mars, with their potential to host embedded biosignatures. The capacity to resume experiments like STONE is therefore a priority, either as a piggyback on FOTON satellites as in the past, or with dedicated facilities.
-
Increase the number of samples available on space facilities
Replicates are important for any science experiment. Access to space being expensive and rare, most of the results and conclusions are extrapolated form a very restricted number of replicates for each set of test samples (most of the time as few as 2 or 3). Future exposure facilities should therefore be larger and experiments should be repeated with the support of easier and more regular access to space.
-
Increase access to space opportunities
Usually, the period of time comprising the publication of an announcement of opportunity, submission of a proposal, acceptance by ESA, financing by the national space agencies, and the actual start of the space experiment is very long (e.g., about 10 years for the EXPOSE-R program). This discourages many research projects. Regular and routine access to astrobiological facilities in space is desirable to spur progress.
4.2 Relevant and Accessible Space Environments for Astrobiology Experiments
The vast majority of astrobiology experiments to date have been conducted in low Earth orbit, primarily for reasons of access to space at reasonable cost. The presence there of the ISS as well as a large number of commercial, military, and government launches to deploy communications and observations satellites offer many opportunities for small satellites to be delivered to LEO as secondary payloads (Swartwout 2013).
Space conditions, notably in terms of radiation environment, were discussed in Sect. 2 of this paper and summarized in Table 1. The geometry and altitude of the most common orbits for artificial satellites are shown in Fig. 46. Looking ahead to the next decade from an astrobiological perspective, there is reason for optimism regarding extension of the reach of orbits beyond LEO, especially for small spacecraft. Orbits and trajectories such as Sun-synchronous orbit (SSO), including polar orbit, or geostationary transfer orbit (GTO) provide access to additional, relevant space environments with very different ambient radiation, as described in Sect. 2 and in Table 1. SSO orbits, even if they do not reach particularly high altitudes, have a sufficiently high inclination to escape the Earth’s radiation belts above the poles, as shown in Fig. 5, where samples can interact directly with GCRs and SPEs. GTOs can opportunistically be used for piggyback (secondary) payloads due to the large number of launches of conventional satellites to GEO. Although not maintaining altitudes where GCRs and SEPs are entirely unfiltered, GTOs and other highly elliptical orbits offer high doses of radiation from energetic particles (electrons in the Inner Van Allen belt, protons and electrons in the Outer Van Allen belt) that can interact with exposed samples. New flight opportunities also include adapting small spacecraft and payloads to become “planetary hitchhikers” on larger science and exploration missions destined for such interesting locations as the moons of Mars, Jupiter, and Saturn, lunar orbit, or near-Earth asteroids.
All stable orbits provide a condition of “free fall” as discussed in Chap. 2. Nevertheless, if an experiment requires the absence of all apparent forces exceeding \(10^{-6}~\mbox{g}\), vibrational forces generated by everything from pumps and motors to exercising crew members onboard spacecraft must be considered; eliminating such perturbations can be a reason to choose a specific spacecraft, such as a free-flying small satellite with no moving parts, rather than a reason to seek a different orbit (Beysens and van Loon 2015).
Cubesat concept nanosatellites for astrobiology are discussed in the next section since they offer promising capabilities for dedicated experimentation in relevant environments.
4.3 Cubesats for Astrobiology/Astrochemistry
A major hurdle for performing experiments beyond Earth is the effort and cost required to launch suitable hardware into space. With ongoing efforts at miniaturization in practically every industrial and scientific sector, small-scale and light-weight space hardware is becoming increasingly available with expanded technical and analytical capabilities. This trend also affects the design and development of small satellites and nanosatellites. The “cubesat” format (one or multiple conjoined cubes of 10 cm length, each cube also being referred to as one “U”) is by now an internationally accepted platform for small satellites and offers increasingly sophisticated analytical measurement capabilities in small, lightweight, low-power, inexpensive packages adaptable to many spaceflight and planetary applications (Rose et al. 2012; Woellert et al. 2011). The rapid advances in miniature, micro, and integrated technologies support the development of innovative small payload systems that can be accommodated on small satellites, returning exciting science results at a fraction of the cost of large missions. Due to their low cost and short development periods, cubesats are seen as powerful tools for not only educational and industry projects but also for scientific experiments in space (Swartwout 2013). Cubesats can spur technology advancement in developing and emerging nations as well (Ansdell et al. 2011; Woellert et al. 2011). Within the framework of the United Nations (UN), the UN Basic Space Technology Initiative (UNBSTI) supports cubesat science and technology applications worldwide and organizes workshops and student training (Balogh et al. 2010).
With a compact and light-weight design, cubesats offer favorable cost-benefit ratios, even as the availability of affordable launch opportunities remains a schedule bottleneck. NASA’s CubeSat Launch Initiative, which provides space launches to successful proposers for 1U, 2U, 3U, and 6U spacecraft as auxiliary payloads aboard a range of spacecraft, is one of the programs that help address the challenge, for US entities, of getting cubesats into orbit.
In the realm of fundamental biological and astrobiological studies, the NASA Ames Research Center (ARC) and collaborators have demonstrated successful spaceflight missions utilizing 2U payload systems such as GeneSat and PharmaSat. GeneSat monitored bacterial gene expression by measuring protein fluorescence and light scattering (Ricco et al. 2007). PharmaSat investigated the effect of antifungal drugs on yeast cells via 3-color measurements of microbe population and metabolic activity (Ricco et al. 2011). The latest cubesat mission with an astrobiology focus that has flown in space, already presented in this paper, is O/OREOS (Organism/Organic Exposure to Orbital Stresses), the 1U Space Environment Survivability of Living Organisms (SESLO) payload of which monitored germination and outgrowth of bacterial spores in space by measuring the changes of absorbance of colorimetric dyes (Nicholson et al. 2011).
Spectroscopy applications that consume 0.2–1.5 watts have been demonstrated in 0.3 to 1.5-U payloads. Miniaturized spectrometers have been developed and flown for in-situ UV, visible, and near-IR spectral measurements (Bramall et al. 2012; Colaprete et al. 2010; Jagpal et al. 2010; Sarda et al. 2006). Spectrometers to cover additional spectral regions (vacuum UV, mid-IR) are now in the planning stages or under development. Cubesat missions and potential technologies for Earth observation have recently been surveyed (Selva and Krejci 2012); the now-emerging trend towards the development and flight of 6U cubesats is a boon to this application, as it provides adequate volume for modest but capable optical systems, including deployable telescopes, to enable remote observations of Earth or the cosmos (Tsitas and Kingston 2010).
Miniaturized instruments and microanalytical systems are driving the capability of small satellite science missions forward by harnessing recent advances in microfluidics, microelectromechanical systems (MEMS) including sensors and actuators, polymer microfabrication technologies, low-power microelectronics, miniature high-efficiency motors, advanced materials, and integrated/fiber optics including micro/miniature light sources, cameras, and spectrometers. In addition to suitability for applications on landers, the ruggedness and minimal mass of some integrated technologies enables them to survive shocks associated with penetrator technology. Cubesat payload technology further provides an outstanding opportunity to enable innovative technological advances in sensor and miniaturized instrument design for astrobiology applications in terrestrial field research and space environments inside (or on the outside of) the ISS, planetary orbiters and landers, and lunar platforms (Yost et al. 2007). In the next section, future experiments, not yet funded beyond concept stage, but which could be implemented on small satellites, will be presented and discussed.
4.4 The Gaps: Key Future Facilities
As already mentioned in this paper, with the current space-exposure facilities such as Biopan and EXPOSE, only passive-exposure experiments are available, with only two measurement time points from which to attempt to derive the kinetics: one before flight and one after. NASA’s O/OREOS nanosatellite demonstrated the measurement of the evolution of organic samples with an onboard UV-Vis spectrometer (Mattioda et al. 2012) (see Sect. 3.1.f). The follow-up to this project, currently under development by ESA, is called OREOcube (Elsaesser et al. 2014) (see Sect. 3.1.g). This new ISS facility will monitor samples with a UV-visible-NIR spectrometer.
The logical next step is to implement a mid-infrared in-situ diagnostic capability in order to follow the structural evolution (via vibrational spectra) of samples during exposure as a function of time. Infrared (IR) spectroscopy has the advantage (over UV-visible) of monitoring, via their bonding, the molecular groups of organic molecules using the \(4000 \mbox{ to } 1000~\mbox{cm}^{-1}\) energy range; IR can therefore identify new molecules or molecular fragments produced during photolysis. Spectrometric measurements of the samples when combined with artificial well-defined light sources, e.g. LEDs, allow also the application of a wide range of analytical methods for astrobiological analysis and kinetic measurements of active cultures. Acousto-optic tunable filters (AOTFs) (Pilorget and Bibring 2014) and/or IR telescope technology (to provide solar illumination for IR spectroscopy) could be adapted to exposure facilities in the near future. A compact design inspired by cubesat technology would enable the use of this tool either outside the ISS, within larger cubesats, or as a “hitchhiker” on planetary missions. A concept diagram for such an analytical tool is shown in Fig. 47.
A major breakthrough would be to enable the exposure of synthetic icy mixtures to space conditions. Such a study is currently in the research-and-development phase at CNES, with the goal of depositing an icy mixture on a “cold spot” in a space exposure facility. Frozen samples could be prepared from gaseous mixtures made on Earth and kept in leak-free vessels as shown in Fig. 48. Volatile molecules would be condensed on a cold location or spot. Reaching a range as low as 10–20 K is probably not feasible in the near future (to simulate interstellar and cometary ices), but a 100 K temperature level would be sufficient to simulate a large number of astrophysical environments for both chemistry and biology, and could be reached at relatively low cost and low mass and size on the basis of existing small cryocoolers developed for space infrared detectors. Mixtures of \(\mathrm{H}_{2}\mathrm{O}\), CO2, NH3 and, CH3OH for instance could be the first to test, as they have proven to have very interesting (photo)reactivity (Vinogradoff et al. 2015).
Future developments will also be necessary for long-term continuous biological studies (weeks to years), with life support across many (up to thousands of) generations for studies of natural selection, adaptive mutations, and directed evolution. Critical support capabilities will have to be implemented in exposure facilities to monitor/control temperature, pressure, humidity, pH, and for sub-culturing/sub-sampling, waste processing, nutrient/gas monitoring and replenishing. Experiments will have to be monitored from the ground and processes tuned according to telemetered results.
Finally, tools for the characterization of the local space environment relevant to astrobiological studies will have to be developed (if not already available) and concurrently implemented. For electromagnetic radiation, they should measure both spectrum and dose rate over the spectral range from the NIR to the wavelength of the Lyman \(\alpha\) emission (122 nm) or shorter. For particle radiation, including GCRs & SPEs, it is important to measure not only total ionizing dose, but the linear-energy-transfer characteristics of typical particle events in the vicinity of the exposure as a function of time: in the event of an SPE, these can be highly variable. Electric and magnetic fields may be relevant and should be recorded if their characteristics are unknown in the vicinity of exposure.
These measurement requirements will lead to special challenges for future facilities in terms of mass, volume, and power consumption of the next generation of instrumentation, especially if it is accommodated by small satellites like cubesats.
5 Summary, Conclusions and Recommendations
Hardware developments in the last ten years have substantially broadened the range of astrochemical and exobiological experiments conducted in low Earth orbit. Since the first biology experiments in space in the 1960’s and the first chemistry experiments in space in the 1980’s, the options to expose a large variety of samples have been expanded.
There are limitations to the current facilities, however. Passive exposure platforms like EXPOSE do not allow direct in-situ measurements of the evolution of the samples. Next-generation platforms such as the O/OREOS NASA nanosat (Ehrenfreund et al. 2014; Mattioda et al. 2012) and the forthcoming OREOcube platform outside the ISS (Elsaesser et al. 2014) allow in-situ measurement in the UV-visible-NIR domain. Beyond these recent platforms, the next generation of exposure facilities should enable mid-infrared diagnostics for greater science return—a significant improvement in exposure facilities. One step beyond, for the study of the organic chemistry of the outer Solar System (icy satellites, comets), dense molecular clouds, and microorganisms in cold environments such as Mars or Europa, will be to enable the exposure of samples at controlled low temperatures.
The space environment conditions provided by the exposure platforms are a final important issue to address. Experiments conducted in most low Earth orbits (see Table 1) are efficiently protected by Earth’s magnetosphere from much of the flux of most high-energy particles (GCRs and SPEs). New space exposure studies should be implemented in environments beyond low Earth orbit, either with higher orbital inclination (polar and near-polar orbits) or much higher altitudes (GTO, HEO, GEO). Major challenges are still ahead to develop and implement the most appropriate exposure platforms, but we are confident that the science return from such new, ambitious programs will be significant and well justified.
An ESA Topical Team focused on “Future Astrobiology Experiments in Earth orbit and beyond” has been working since 2011 to discuss the benefits of conducting experiments related to astrobiology in Earth orbit and to submit to ESA recommendations to improve the current facilities and overcome their limitations. This paper reports the work of this Topical Team; its recommendations are as follows:
-
Resumption of Biopan-like experiments is recommended.
The timing of preparation for experiments on the ISS is extremely long (a few months between sample delivery and actual launch, sometimes a few weeks inside the ISS before EVA, 18 to 24 months in space, and then a few weeks to return samples to terrestrial laboratories for analysis), while Biopan samples were prepared just 2 to 3 weeks before launch, and analyzed in laboratories a few days after returning to Earth. Some samples are not suitable for study on ISS due to their poor stabilities over such long storage and exposure durations; they should be exposed for short durations (a few weeks) in order to decide if they should occupy a valuable position in an exposure facility for more than a year in space. In addition, continuation of long-term exposure facilities is well suited to the study of highly UV-resistant molecules or microorganisms, and to the study of long-term evolution of bacterial colonies (generally in absence of solar UV). The orbits or surfaces of the Moon, Mars, or Mars’ satellites, as well as space-mission hitchhikers, could provide environmentally unique, privileged locations for such studies in the future.
-
STONE-like experiments should resume.
STONE experiments have no equivalent since 2007, despite the fact that science related to the stability of minerals and organic compounds during meteoritic infall has important astrobiological significance. A study of how best to resume this class of experiment—whether on a FOTON capsule or as impact modules ejected from the ISS—should be conducted.
-
New individual retrievable satellites should be developed.
The development of individual retrievable satellites that can be returned to the ground is needed for all experiments that benefit from post-flight on-ground analysis of samples in addition to in-situ measurements.
-
Experiments should be conducted in clean environments.
Facilities far from the polluted environment of ISS should be made available. Gas plumes from Soyuz and ISS resupply vehicles, as well as degradation and outgassing of external ISS materials, along with oxygen radicals from Earth’s residual atmosphere below 500 km, make protection of samples by optical components such as MgF2 windows necessary, blocking VUV photons below 115 nm. The flux at these short wavelengths, although low, is required for photodissociation of very relevant molecules such as \(\mathrm{N}_{2}\) (\(< 100~\mbox{nm}\)). Direct exposure of samples to full-spectrum solar radiation should be possible without protective windows to block chemical pollution.
-
Orbits should allow both photolysis and particle irradiation.
Environments with significant fluxes of energetic charged particles (GCRs, SPEs) should be accessible. ISS’s altitude is well within Earth’s magnetosphere, blocking many of the most damaging particle types and energies. An exposure environment combining high fluxes of both photons and energetic charged particles would be a major improvement compared to existing facilities in space as well as in Earth laboratories. Polar, transfer, geostationary, or other beyond-LEO orbits will be necessary for future facilities.
-
Samples should be analyzed throughout the duration of the experiments.
Sample analysis should be feasible throughout the exposure period, not only before launch and after return to Earth: in-situ analytical instruments are required. OREOcube will be equipped with a UV-vis-NIR spectrometer that provides useful information about the electronic structure of thin films, particularly those with informative bands in the visible and near-UV regions (organometallics as well as many metal oxides and most PAHs, for example). For most astrochemistry experiments, optimal molecular characterization, including structural changes and new product formation, also requires measurement of the details of molecular bonding. This can be provided by an IR or Raman spectrometer that covers the \(4000\mbox{--}1000~\mbox{cm}^{-1}\) energy range; such an instrument would make the return of samples to Earth less of a necessity. This capability for vibrational spectroscopy is well within the technical reach of nanosatellites, including those destined for polar or geotransfer orbits. In addition, other techniques for characterization may be required according to the details of the samples and their expected response to the space environment: laser-induced fluorescence and mass spectroscopic analysis (of headspace gases above samples) are two examples.
-
Temperature should be controlled and allow experimentation on icy samples.
Temperature control is an additional priority. Currently, solid films and gaseous mixtures are exposed at temperatures that are largely uncontrolled during exposure. Large temperature fluctuations have been observed on the Biopan, EXPOSE, and O/OREOS experiments (\(-20^{\circ}\mbox{C to } {+}40^{\circ}\mbox{C}\)). This wide temperature range prevents the study of compounds having significant vapor pressure above 30°C. Thermal control can therefore broaden the range of compounds that can be studied in exposure facilities. Moreover, with regard to organic astrochemistry, radiation-driven changes occur in the gaseous and solid phases, but also, for a large fraction, in the ice phase at low temperature (\(<100~\mbox{K}\)) at the surfaces of the icy moons of the giant planets, in comets, and in interstellar dark clouds. Future exposure facilities should include cold sample locations where icy mixtures can be prepared, after launch, from gaseous mixtures (H\(_{2}\mathrm{O}\), CO2, NH3 and CH3OH for instance). Also for the exposure of organisms to environments similar to e.g. Mars or Europa with respect to temperature, cold exposure platforms for long-term exposure (\(> 2~\mbox{weeks}\)) are needed.
-
Active organisms should be exposed.
Living organisms should be exposed to the space environment in the metabolically active and, for many, reproductively active forms. It is already known that, in actively metabolizing organisms, non-terrestrial gravity levels including microgravity can have significant effects, whether direct or due to modification of mass-transport mechanisms, as can the other space environmental parameter most perturbing to living biological systems removed from their terrestrial habitats, radiation. To understand the cellular and molecular mechanisms of adaptation and evolution, e.g. under Mars or space conditions, investigations require active metabolism, at least temporarily, and in some cases for multiple generations. Long-term experiments with actively growing organisms offer a new horizon for biology exposure experiments. Adaptation and optimization of existing, well-developed laboratory technologies are necessary for space application, including monitoring and control of temperature, relative humidity, pressure, pH, gas-phase composition, the supply of nutrients, the removal of waste products, and the delivery of reagents. Small space bioreactors and microevolution chambers are currently in the infancy of conception and development; they should include options for automatic sub-/sampling at regular, predefined but variable intervals, options to adjust or redirect the experiments from the ground by tele-monitoring and tele-command, as well as appropriate in-situ/in-orbit observation and analysis to detect metabolic changes and quantify adaptation processes.
Notes
References
L.J. Allamandola, D.M. Hudgins, S.A. Sandford, Astrophys. J. 511, L115–L119 (1999)
K. Altwegg, H. Balsiger, A. Bar-Nun, J.-J. Berthelier, A. Bieler, P. Bochsler, C. Briois, U. Calmonte, M.R. Combi, H. Cottin et al., Sci. Adv. 2(5), e1600285 (2016)
M. Ansdell, P. Ehrenfreund, C. McKay, Acta Astronaut. 68, 2098–2113 (2011)
A. Aubrey, H.J. Cleaves, J.H. Chalmers, A.M. Skelley, R.A. Mathies, F.J. Grunthaner, P. Ehrenfreund, J.L. Bada, Geology 34, 357–360 (2006)
W. Balogh, L. Canturk, S. Chernikov, T. Doi, S. Gadimova, H. Haubold, V. Kotelnikov, Space Policy 26, 185–188 (2010)
M. Baqué, G. Scalzi, E. Rabbow, P. Rettberg, D. Billi, Orig. Life Evol. Biosph. 43, 377–389 (2013)
M. Baqué, C. Verseux, U. Böttger, E. Rabbow, J.-P.P. Vera, D. Billi, Orig. Life Evol. Biosph. 46, 289–310 (2015)
G.A. Baratta, D. Chaput, H. Cottin, L.F. Cascales, M.E. Palumbo, G. Strazzulla, Planet. Space Sci. 118, 211–220 (2015)
B. Barbier, A. Chabin, D. Chaput, A. Brack, Planet. Space Sci. 46, 391–398 (1998)
B. Barbier, O. Henin, F. Boillot, A. Chabin, D. Chaput, A. Brack, Planet. Space Sci. 50, 353–359 (2002)
T. Berger, M. Hajek, P. Bilski, G. Reitz, Int. J. Astrobiol. 14, 27–32 (2015)
M. Bertrand, A. Chabin, A. Brack, H. Cottin, D. Chaput, F. Westall, Astrobiology 12, 426–435 (2012)
M. Bertrand, A. Chabin, C. Colas, M. Cadène, D. Chaput, A. Brack, H. Cottin, F. Westall, Int. J. Astrobiol. 14, 89–97 (2015)
D.A. Beysens, J.J.W.A. van Loon, The space gravity environment, in Generation and Applications of Extra-Terrestrial Environments on Earth, ed. by D.A. Beysens, J.J.W.A. van Loon (River Publishers, Aalborg, 2015), pp. 5–9
J.P. Bibring, Y. Langevin, J.F. Mustard, F. Poulet, R. Arvidson, A. Gendrin, B. Gondet, N. Mangold, P. Pinet, F. Forget, Science 312, 400–404 (2006)
F. Boillot, A. Chabin, C. Buré, M. Venet, A. Belsky, M. Bertrand-Urbaniak, A. Delmas, A. Brack, B. Barbier, Orig. Life Evol. Biosph. 32, 359–385 (2002)
A.C.A. Boogert, K.M. Pontoppidan, C. Knez, F. Lahuis, J. Kessler-Silacci, E.F. van Dishoeck, G.A. Blake, J.C. Augereau, S.E. Bisschop, S. Bottinelli et al., Astrophys. J. 678, 985 (2008)
W.V. Boynton, D.W. Ming, B. Sutter, R.E. Arvidson, J. Hoffman, P.B. Niles, P. Smith, P.S. Team, Lunar Planet. Sci. Conf. Abstr. 40, 2434 (2009)
A. Brack, P. Baglioni, G. Borruat, F. Brandstatter, R. Demets, H.G.M. Edwards, M. Genge, G. Kurat, M.F. Miller, E.M. Newton et al., Planet. Space Sci. 50, 763–772 (2002). doi:10.1016/S0032-0633(02)00018-1
N.E. Bramall, R. Quinn, A. Mattioda, K. Bryson, J.D. Chittenden, A. Cook, C. Taylor, G. Minelli, P. Ehrenfreund, A.J. Ricco et al., Planet. Space Sci. 60, 121–130 (2012)
F. Brandstaetter, A. Brack, P. Baglioni, C.S. Cockell, R. Demets, H.G.M. Edwards, G. Kurat, G.R. Osinski, J.M. Pillinger, C.-A. Roten et al., Planet. Space Sci. 56, 976–984 (2008)
A. Brandt, J.-P. de Vera, S. Onofri, S. Ott, Int. J. Astrobiol. 14, 411–425 (2015)
A. Brandt, E. Posthoff, J.-P. de Vera, S. Onofri, S. Ott, Orig. Life Evol. Biosph. 46, 311–321 (2016)
J.J. Brocks, G.A. Logan, R. Buick, R.E. Summons, Science 285, 1033–1036 (1999)
J.J. Brocks, R. Buick, G.A. Logan, R.E. Summons, Geochim. Cosmochim. Acta 67, 4289–4319 (2003)
K.L. Bryson, Z. Peeters, F. Salama, B. Foing, P. Ehrenfreund, A.J. Ricco, E. Jessberger, A. Bischoff, M. Breitfellner, W. Schmidt et al., Adv. Space Res. 48, 1980–1996 (2011)
K.L. Bryson, F. Salama, A. Elsaesser, Z. Peeters, A.J. Ricco, B.H. Foing, Y. Goreva, Int. J. Astrobiol. 14, 55–66 (2015)
H. Bucker, G. Horneck, Acta Astronaut. 2, 247–264 (1975)
M.P. Callahan, K.E. Smith, H.J. Cleaves, J. Ruzicka, J.C. Stern, D.P. Glavin, C.H. House, J.P. Dworkin, Proc. Natl. Acad. Sci. USA 108, 13995–13998 (2011)
F. Capaccioni, A. Coradini, G. Filacchione, S. Erard, G. Arnold, P. Drossart, M.C. De Sanctis, D. Bockelee-Morvan, M.T. Capria, F. Tosi et al., Science 347, aaa0628 (2015)
N. Carrasco, H. Cottin, M. Cloix, M. Jérome, Y. Bénilan, P. Coll, M.-C. Gazeau, F. Raulin, K. Saiagh, D. Chaput et al., Int. J. Astrobiol. 14, 79–87 (2015)
Y.-J. Chen, K.-J. Chuang, G.M.M. Caro, M. Nuevo, C.-C. Chu, T.-S. Yih, W.-H. Ip, C.-Y.R. Wu, Astrophys. J. 781, 15 (2014)
H. Chennaoui Aoudjehane, G. Avice, J.-A. Barrat, O. Boudouma, G. Chen, M.J.M. Duke, I.A. Franchi, J. Gattacceca, M.M. Grady, R.C. Greenwood et al., Science 338, 785–788 (2012)
C. Chyba, C. Sagan, Nature 355, 125–132 (1992)
M.W. Claire, J. Sheets, M. Cohen, I. Ribas, V.S. Meadows, D.C. Catling, Astrophys. J. 757, 95 (2012)
S.J. Clemett, C.R. Maechling, R.N. Zare, P.D. Swan, R.M. Walker, Science 262, 721–725 (1993)
C.S. Cockell, D.C. Catling, W.L. Davis, K. Snook, R.L. Kepner, P. Lee, C.P. McKay, Icarus 146, 343–359 (2000)
C.S. Cockell, A. Brack, D.D. Wynn-Williams, P. Baglioni, F. Brandstaetter, R. Demets, H.G.M. Edwards, A.L. Gronstal, G. Kurat, P. Lee et al., Astrobiology 7, 1–9 (2007)
C.S. Cockell, P. Rettberg, E. Rabbow, K. Olsson-Francis, ISME J. 5, 1671–1682 (2011)
L. Colangeli, J.R. Brucato, A. Bar-Nun, R.L. Hudson, M.H. Moore, in Laboratory Experiments on Cometary Materials, ed. by M.C. Festou, H.U. Keller, H.A. Weaver (University of Arizona Press, Tucson, 2004), pp. 695–717
A. Colaprete, P. Schultz, J. Heldmann, D. Wooden, M. Shirley, K. Ennico, B. Hermalyn, W. Marshall, A. Ricco, R.C. Elphic et al., Science 330, 463–468 (2010)
A.M. Cook, A.L. Mattioda, R.C. Quinn, A.J. Ricco, P. Ehrenfreund, N.E. Bramall, G. Minelli, E. Quigley, R. Walker, R. Walker, Astrophys. J. Suppl. Ser. 210, 15 (2014)
H. Cottin, M.C. Gazeau, F. Raulin, Planet. Space Sci. 47, 1141–1162 (1999)
H. Cottin, M.H. Moore, Y. Bénilan, Astrophys. J. 590, 874–881 (2003)
H. Cottin, A. Noblet, Y.Y. Guan, O. Poch, K. Saiagh, M. Cloix, F. Macari, M. Jérome, P. Coll, F. Raulin et al., Astrobiology 12, 412–425 (2012)
H. Cottin, J.M. Kotler, K. Bartik, H.J. Cleaves, C.S. Cockell, J.-P.P. de Vera, P. Ehrenfreund, S. Leuko, I.L. Ten Kate, Z. Martins et al., Space Sci. Rev. 1–42 (2015a). doi:10.1007/s11214-015-0196-1
H. Cottin, K. Saiagh, Y.Y. Guan, M. Cloix, D. Khalaf, F. Macari, M. Jérome, J.-M. Polienor, Y. Bénilan, P. Coll et al., Int. J. Astrobiol. 14, 67–77 (2015b)
J. Cronin, R.S. Chang, in Organic Matter in Meteorites: Molecular and Isotopic Analyses of the Murchison Meteorites, ed. by J.M. Greenberg, C.X. Mendoza-Gomez, V. Pirronello (Kluwer Academic, Dordrecht, 1993), pp. 209–258
L.M. Crowe, J.H. Crowe, Adv. Space Res. 12, 239–247 (1992)
T.P. Dachev, B. Tomov, Y. Matviichuk, P. Dimitrov, N. Bankov, Adv. Space Res. 44, 1433–1440 (2009)
T. Dachev, G. Horneck, D.-P. Häder, M. Schuster, P. Richter, M. Lebert, R. Demets, Astrobiology 12, 403–411 (2012)
T. Dachev, G. Horneck, D.-P. Häder, M. Schuster, M. Lebert, Int. J. Astrobiol. 14, 17–25 (2015)
E. Dartois, C. Engrand, R. Brunetto, J. Duprat, T. Pino, E. Quirico, L. Remusat, N. Bardin, G. Briani, S. Mostefaoui et al., Icarus 224, 243–252 (2013)
A.F. Davila, D. Schulze-Makuch, Astrobiology 16, 159–168 (2016)
R. de la Torre, L.G. Sancho, G. Horneck, A. de los Rios, J. Wierzchos, K. Olsson-Francis, C.S. Cockell, P. Rettberg, T. Berger, J.-P.P. de Vera et al., Icarus 208, 735–748 (2010)
P. de Marcellus, C. Meinert, M. Nuevo, J.J. Filippi, G. Danger, D. Deboffle, L. Nahon, L.L.S. d’Hendecourt, U.J. Meierhenrich, Astrophys. J. Lett. 727, L27 (2011)
J.-P. de Vera, Fungal Ecol. 5, 472–479 (2012)
J.P. de Vera, S. Ott, in Resistance of Symbiotic Eukaryotes to Simulated Space Conditions and Asteroid Impact Catastrophes, ed. by J. Seckbach, M. Grube (2010), pp. 595–611
J.-P. de Vera, U. Boettger, R. de la Torre Noetzel, F.J. Sánchez, D. Grunow, N. Schmitz, C. Lange, H.-W. Hübers, D. Billi, M. Baqué et al., Planet. Space Sci. 74, 103–110 (2012)
C. DeDuve, in Possible Starts for Primitive Life. Clues from Present-day Biologie: The Thioester World, ed. by A. Brack (Cambridge University Press, Cambridge, 1998), pp. 219–236
R. Delonge, DLR Forschungsbericht DLR-FB 93-06 (1995)
R. Demets, W. Schulte, P. Baglioni, Adv. Space Res. 36, 311–316 (2005)
R. Demets, M. Bertrand, A. Bolkhovitinov, K. Bryson, C. Colas, H. Cottin, J. Dettmann, P. Ehrenfreund, A. Elsaesser, E. Jaramillo et al., Int. J. Astrobiol. 14(1), 33–45 (2015)
A.W. DeWolfe, A. Wilson, D.M. Lindholm, C.K. Pankratz, M.A. Snow, T.N. Woods, in AGU 2010, San Francisco (2010), GC21B-0881
R. dos Santos, M. Patel, J. Cuadros, Z. Martins, Icarus 277, 342–353 (2016)
K. Dose, A. Bieger-Dose, R. Dillmann, M. Gill, O. Kerz, A. Klein, H. Meinert, T. Nawroth, S. Risi, C. Stridde, Adv. Space Res. 16, 119–129 (1995)
K. Dose, A. Bieger-Dose, R. Dillmann, M. Gill, O. Kerz, A. Klein, C. Stridde, Adv. Space Res. 18(12), 51–60 (1996)
B.L. Ehlmann, J.F. Mustard, S.L. Murchie, F. Poulet, J.L. Bishop, A.J. Brown, W.M. Calvin, R.N. Clark, D.J. Des Marais, R.E. Milliken et al., Science 322, 1828 (2008)
P. Ehrenfreund, S.B. Charnley, Annu. Rev. Astron. Astrophys. 38, 427–483 (2000)
P. Ehrenfreund, M. Sephton, Faraday Discuss. 133, 277 (2006)
P. Ehrenfreund, R. Ruiterkamp, Z. Peeters, B. Foing, F. Salama, Z. Martins, Planet. Space Sci. 55, 383–400 (2007)
P. Ehrenfreund, A.J. Ricco, D. Squires, C. Kitts, E. Agasid, N. Bramall, K. Bryson, J. Chittenden, C. Conley, A. Cook et al., Acta Astronaut. 93, 501–508 (2014)
A. Elsaesser, R.C. Quinn, P. Ehrenfreund, A.L. Mattioda, A.J. Ricco, J. Alonzo, A. Breitenbach, Y.K. Chan, A. Fresneau, F. Salama et al., Langmuir 30(44), 13217–13227 (2014)
J.E. Elsila, D.P. Glavin, J.P. Dworkin, Meteorit. Planet. Sci. 44, 1323–1330 (2009)
E-t. Es-sebbar, Y. Bénilan, N. Fray, H. Cottin, A. Jolly, M.-C. Gazeau, Astrophys. J. Suppl. 218 (2015)
F. Ferrari, E. Szuszkiewicz, Astrobiology 9, 413–436 (2009)
G.J. Flynn, Earth Moon Planets 72, 469–474 (1996)
F. Foucher, F. Westall, F. Brandstaetter, R. Demets, J. Parnell, C.S. Cockell, H.G.M. Edwards, J.-M. Beny, A. Brack, Icarus 207, 616–630 (2010)
N. Fray, A. Bardyn, H. Cottin, K. Altwegg, D. Baklouti, C. Briois, L. Colangeli, C. Engrand, H. Fischer, A. Glasmachers et al., Nature 538, 72–74 (2016). doi:10.1038/nature19320
C. Freissinet, D.P. Glavin, P.R. Mahaffy, K.E. Miller, J.L. Eigenbrode, R.E. Summons, A.E. Brunner, A. Buch, C. Szopa, P.D. Archer Jr. et al., J. Geophys. Res., Planets 120, 495–514 (2015)
S.F. Fung, in Radiation Belts: Models and Standards. Geophysical Monograph Series, vol. 97 (1996), pp. 79–91
T. Gaisser, Cosmic Rays and Particle Physics (Cambridge University Press, Cambridge, 1990)
J.R.C. Garry, I.L. Ten Kate, Z. Martins, P. Nornberg, P. Ehrenfreund, Meteorit. Planet. Sci. 41, 391–405 (2006)
E.L. Gibb, D.C.B. Whittet, A.C.A. Boogert, A.G.G.M. Tielens, Astrophys. J. Suppl. Ser. 151, 35–73 (2004)
G.P. Ginet, T.P. O’Brien, S.L. Huston, W.R. Johnston, T.B. Guild, R. Friedel, C.D. Lindstrom, C.J. Roth, P. Whelan, R.A. Quinn et al., Space Sci. Rev. 179, 579–615 (2013)
F. Goesmann, H. Rosenbauer, J.H. Bredehöft, M. Cabane, P. Ehrenfreund, T. Gautier, C. Giri, H. Krüger, L. Le Roy, A.J. MacDermott et al., Science 349(6247), aab0689 (2015)
D.O. Gough, Sol. Phys. 74, 21–34 (1981)
J.M. Greenberg, A. Li, C.X. Mendoza-Gómez, W.A. Schutte, P.A. Gerakines, M. de Groot, Astrophys. J. 455, L177–L180 (1995)
Y.Y. Guan, N. Fray, P. Coll, F. Macari, F. Raulin, D. Chaput, H. Cottin, Planet. Space Sci. 58, 1327–1346 (2010)
M. Güdel, J.F. Kasting, in Origins and Evolution of Life, ed. by M. Gargaud, P. Lopez-Garcia, H. Martin (Cambridge University Press, Cambridge, 2011)
K. Gustafsson, L. Sihver, D. Mancusi, T. Sato, K. Niita, Acta Astronaut. 65, 279–288 (2009)
K.S. Habicht, M. Gade, B. Thamdrup, P. Berg, D.E. Canfield, Science 298, 2372–2374 (2002)
J.P. Harrison, N. Gheeraert, D. Tsigelnitskiy, C.S. Cockell, Trends Microbiol. 21, 204–212 (2013)
D.M. Hassler, C. Zeitlin, R.F. Wimmer-Schweingruber, S. Böttcher, C. Martin, J. Andrews, E. Böhm, D.E. Brinza, M.A. Bullock, S. Burmeister et al., Space Sci. Rev. 170, 503–558 (2012)
D.M. Hassler, C. Zeitlin, R.F. Wimmer-Schweingruber, B. Ehresmann, S. Rafkin, J.L. Eigenbrode, D.E. Brinza, G. Weigle, S. Böttcher, E. Böhm et al., Science 343(6169), 1244797 (2014)
E. Herbst, E. van Dishoeck, Annu. Rev. Astron. Astrophys. 47, 427–480 (2009)
J.R. Hörandel, Prog. Part. Nucl. Phys. 64, 351–359 (2010)
G. Horneck, H. Bücker, G. Reitz, H. Raquardt, K. Dose, K.D. Martens, H.D. Mennigmann, P. Weber, Science 225, 226–228 (1984a)
G. Horneck, H. Bücker, G. Reitz, H. Requardt, K. Dose, K.D. Martens, A. Bieger, H.D. Mennigmann, P. Weber, Adv. Space Res. 4, 19–27 (1984b)
G. Horneck, H. Bücker, G. Reitz, Adv. Space Res. 14, 41–45 (1994)
G. Horneck, P. Rettberg, G. Reitz, J. Wehner, U. Eschweiler, K. Strauch, C. Panitz, V. Starke, C. Baumstark-Khan, Orig. Life Evol. Biosph. 31, 527–547 (2001)
G. Horneck, D. Stöffler, S. Ott, U. Hornemann, C.S. Cockell, R. Moeller, C. Meyer, J.-P. de Vera, J. Fritz, S. Schade et al., Astrobiology 8, 17–44 (2008)
G. Horneck, D.M. Klaus, R.L. Mancinelli, Microbiol. Mol. Biol. Rev. 74, 121 (2010)
G. Horneck, R. Moeller, J. Cadet, T. Douki, R.L. Mancinelli, W.L. Nicholson, C. Panitz, E. Rabbow, P. Rettberg, A. Spry et al., Astrobiology 12, 445–456 (2012)
S. Hörst, R. Yelle, A. Buch, N. Carrasco, G. Cernogora, O. Dutuit, E. Quirico, E. Sciamma-O’Brien, M. Smith, A. Somogyi et al., Astrobiology 12, 809–817 (2012)
J. Hotchin, P. Lorenz, A. Markusen, C. Hemenway, Life Sci. Space Res. 5, 1–6 (1967)
J. Hotchin, P. Lorenz, C. Hemenway, Life Sci. Space Res. 6, 108–114 (1968)
C. Huber, G. Wächtershäuser, Science 276, 245–247 (1997)
C. Huber, G. Wächtershäuser, Science 281, 670–672 (1998)
L. Innocenti, D.A.M. Mesland, Adv. Space Res. 16(8), 1–3 (1995)
ISO-21348:2007 (2007)
G. Israël, C. Szopa, F. Raulin, M. Cabane, H.B. Niemann, S.K. Atreya, S.J. Bauer, J.-F. Brun, E. Chassefière, P. Coll et al., Nature 438, 796–799 (2005)
R.K. Jagpal, B.M. Quine, H. Chesser, S.M. Abrarov, R. Lee, J. Appl. Remote Sens. 4, 049501 (2010)
K.I. Jönsson, E. Rabbow, R.O. Schill, M. Harms-Ringdahl, P. Rettberg, Curr. Biol. 18, R729–R731 (2008)
B.A. Kahn, P.J. Stoffella, J. Am. Soc. Hortic. Sci. 121, 414–418 (1996)
Y. Kawaguchi, Y. Yang, N. Kawashiri, K. Shiraishi, M. Takasu, I. Narumi, K. Satoh, H. Hashimoto, K. Nakagawa, Y. Tanigawa et al., Orig. Life Evol. Biosph. 43, 411–428 (2013)
Y. Kawaguchi, S. Yokobori, H. Hashimoto, H. Yano, M. Tabata, H. Kawai, A. Yamagishi, Astrobiology 16, 363–376 (2016)
M.A. Khenokh, E.A. Kuzicheva, N.V. Tsupkina, N.I. Konshin, A.L. Mashinsky, G.S. Nechitailo, Dokl. Akad. Nauk SSSR 249, 749–752 (1979)
S. Kimura, K. Tomita-Yokotani, Y. Igarashi, S. Sato, H. Katoh, T. Abe, K. Sonoike, M. Ohmori, Biol. Sci. Space 29, 12–18 (2015)
S. Kitayama, S. Asaka, K. Totsuka, J. Bacteriol. 155, 1200–1207 (1983)
C. Kitts, M. Rasay et al., in 25th Annual AIAA/USU Conference on Small Satellites, Logan, USA (2011), SSC11-II-3
G. Kopp, J.L. Lean, Geophys. Res. Lett. 38, L01706 (2011)
H. Koshiishi, H. Matsumoto, J. Atmos. Sol.-Terr. Phys. 99, 129–133 (2013)
E.A. Kuzicheva, N.B. Gontareva, Adv. Space Res. 23, 393–396 (1999)
E.A. Kuzicheva, N.B. Gontareva, Astrobiology 3, 253–261 (2003)
E.A. Kuzicheva, M.B. Simakov, Adv. Space Res. 23, 387–391 (1999)
E.A. Kuzicheva, N.V. Tzupkina, N.G. Potapova, Adv. Space Res. 9, 53–56 (1989)
E.A. Kuzicheva, M.B. Simakov, I.L. Mal’ko, N.Y. Dodonova, N.B. Gontareva, Adv. Space Res. 18, 65–68 (1996)
J. Lederberg, Science 132, 393–400 (1960)
L. Leshin, P. Mahaffy, C. Webster, M. Cabane, P. Coll, P. Conrad, P. Archer, S. Atreya, A. Brunner, A. Buch et al., Science 341, 1238937 (2013)
H. Lowenstam, Science 211, 1126–1131 (1981)
N.A. Lyons, R. Kolter, Curr. Opin. Microbiol. 24, 21–28 (2015)
R.L. Mancinelli, M.R. White, L.J. Rothschild, Adv. Space Res. 22, 327–334 (1998)
S. Mann, Biomineralization: Principles and Concepts in Bioinorganic Materials Chemistry (Oxford University Press, New York, 2001)
K. Marti, T. Graf, Annu. Rev. Earth Planet. Sci. 20, 221–243 (1992)
Z. Martins, Elements 7, 3540 (2011)
Z.M. Martins, A. Sephton, in Extraterrestrial Amino Acids, ed. by A.B. Hughes (Wiley-VCH, Weinheim, 2009), pp. 3–42
Z. Martins, O. Botta, M.L. Fogel, M.A. Sephton, D.P. Glavin, J.S. Watson, J.P. Dworkin, A.W. Schwartz, P. Ehrenfreund, Earth Planet. Sci. Lett. 270, 130–136 (2008)
Z. Martins, M.A. Sephton, B.H. Foing, P. Ehrenfreund, Int. J. Astrobiol. 10, 231–238 (2011)
Z. Martins, H. Cottin, J.M. Kotler, N. Carrasco, C. Cockell, R.d.l. Torre, R. Demets, J.-P. d. Vera, L. d’Hendecourt, P. Ehrenfreund et al., Space Sci. Rev. (2017). doi:10.1007/s11214-017-0369-1
J.S. Mathis, P.G. Mezger, N. Panagia, Astron. Astrophys. 128, 212–229 (1983)
G. Matrajt, G. Flynn, D. Brownlee, D. Joswiak, S. Bajt, Astrophys. J. 765, 145–162 (2013)
A. Mattioda, A. Cook, P. Ehrenfreund, R. Quinn, A.J. Ricco, D. Squires, N. Bramall, K. Bryson, J. Chittenden, G. Minelli et al., Astrobiology 12, 841–853 (2012)
A. Mattioda, A. Cook, R. Quinn, A. Elsaesser, P. Ehrenfreund, A. Ricca, N.C. Jones, S. Hoffmann, A. Ricco, Abstr. Pap.—Am. Chem. Soc. 247 (2014)
J.E. Mazur, W.R. Crain, M.D. Looper, D.J. Mabry, J.B. Blake, A.W. Case, M.J. Golightly, J.C. Kasper, H.E. Spence, Space Weather 9, S07002 (2011)
M.B. McElroy, 2012
J. Meessen, F.J. Sanchez, A. Brandt, E.M. Balzer, R. de la Torre, L.G. Sancho, J.P. de Vera, S. Ott, Orig. Life Evol. Biosph. 43, 283–303 (2013)
C. Meinert, J.-J. Filippi, P. de Marcellus, L.L.S. d’Hendecourt, U.J. Meierhenrich, ChemPlusChem 77, 186–191 (2012)
C. Meinert, S.V. Hoffmann, P. Cassam-Chenaï, A.C. Evans, C. Giri, L. Nahon, U.J. Meierhenrich, Angew. Chem., Int. Ed. Engl. 53, 210–214 (2014)
C. Meinert, I. Myrgorodska, P. de Marcellus, T. Buhse, L. Nahon, S.V. Hoffmann, L.L.S. d’Hendecourt, U.J. Meierhenrich, Science 352, 208–212 (2016)
R.A. Mewaldt, Cosmic rays, in Macmillan Encyclopedia of Physics, vol. 1 (Macmillan, New York, 1996)
R. Moeller, G. Reitz, W.L. Nicholson, G. Horneck (The PROTECT Team), Astrobiology 12, 457–468 (2012)
B.E. Moseley, H.J. Copland, J. Bacteriol. 121, 422–428 (1975)
B.E. Moseley, D.M. Evans, J. Gen. Microbiol. 129, 2437–2445 (1983)
L.M. Mukhin, A.P. Koscheevi, Y.P. Dikov, J. Huth, H. Wänke, Nature 379, 141–143 (1996)
G.M. Muñoz Caro, G. Matrajt, E. Dartois, M. Nuevo, L. D’Hendecourt, D. Deboffle, G. Montagnac, N. Chauvin, C. Boukari, D. Le Du, Astron. Astrophys. 459, 147–159 (2006)
L. Nahon, N. de Oliveira, G.A. Garcia, J.F. Gil, B. Pilette, O. Marcouille, B. Lagarde, F. Polack, J. Synchrotron Radiat. 19, 508–520 (2012)
I. Narumi, K. Satoh, S. Cui, T. Funayama, S. Kitayama, H. Watanabe, Mol. Microbiol. 54, 278–285 (2004)
NASA U.S. Standard Atmosphere, U.S. Government Printing Office, Washington, D.C., 1962, NASA-TM-X-74335 (NASA, Washington, D.C., 1976), 241 pp
H.E. Newell, J.E. Naugle, Science 132, 1465–1472 (1960)
W.L. Nicholson, Trends Microbiol. 17, 243–250 (2009)
W.L. Nicholson, A.J. Ricco, R. Mancinelli, O. Santos, D. Ly, M. Parra, P. Ehrenfreund, D. Squires, C. Kitts, E. Agasid et al., Astrobiology 11, 951–958 (2011)
A. Noblet, Y.Y. Guan, F. Stalport, O. Poch, P. Coll, C. Szopa, M. Cloix, F. Macari, F. Raulin, D. Chaput et al., Astrobiology 12, 436–444 (2012)
J.W. Norbury, Adv. Space Res. 47, 611–621 (2011)
J.A. Nuth III, N.M. Johnson, S. Manning, Astrophys. J. Lett. 673, L225–L228 (2008)
K.I. Öberg, A.C.A. Boogert, K.M. Pontoppidan, S. van den Broek, E.F. van Dishoeck, S. Bottinelli, G.A. Blake, I. Neal, J. Evans, Astrophys. J. 740, 109 (2011)
K. Olsson-Francis, C.S. Cockell, J. Microbiol. Methods 80, 1–13 (2010)
K. Olsson-Francis, R. Torre, M.C. Towner, C.S. Cockell, Orig. Life Evol. Biosph. 39, 565–579 (2009)
K. Olsson-Francis, R. de la Torre, C.S. Cockell, Appl. Environ. Microbiol. 76, 2115–2121 (2010)
S. Onofri, R. de la Torre, J.-P. de Vera, S. Ott, L. Zucconi, L. Selbmann, G. Scalzi, K.J. Venkateswaran, E. Rabbow, F.J. Sanchez Inigo et al., Astrobiology 12, 508–516 (2012)
S. Onofri, J.-P. de Vera, L. Zucconi, L. Selbmann, G. Scalzi, K.J. Venkateswaran, E. Rabbow, R. de la Torre, G. Horneck, Astrobiology 15, 1052–1059 (2015)
A.I. Oparin, The Origin of Life (Dover, New York, 1953). Republication of the 1938 edition with the addition of a new Introduction by the translator
C.K. Pankratz, B.G. Knapp, R.A. Reukauf, J. Fontenla, M.A. Dorey, L.M. Connelly, A.K. Windnagel, Sol. Phys. 230, 389–413 (2005)
J. Parnell, S.A. Bowden, D. Muirhead, N. Blamey, F. Westall, R. Demets, S. Verchovsky, F. Brandstaetter, A. Brack, Icarus 212, 390–402 (2011)
B. Pasquale, J. Phys. Conf. Ser. 203, 012017 (2010)
M.R. Patel, J.C. Zarnecki, D.C. Catling, Planet. Space Sci. 50, 915–927 (2002)
A.A. Pavlov, G. Vasilyev, V.M. Ostryakov, A.K. Pavlov, P. Mahaffy, Geophys. Res. Lett. 39, L13202 (2012)
Z. Peeters, R. Quinn, Z. Martins, M.A. Sephton, L. Becker, M.C.M. van Loosdrecht, J. Brucato, F. Grunthaner, P. Ehrenfreund, Int. J. Astrobiol. 8, 301–315 (2009)
C. Pilorget, J.-P. Bibring, Planet. Space Sci. 99, 7–18 (2014)
O. Poch, A. Noblet, F. Stalport, J.J. Correia, N. Grand, C. Szopa, P. Coll, Planet. Space Sci. 85, 188–197 (2013)
O. Poch, M. Jaber, F. Stalport, S. Nowak, T. Georgelin, J.-F. Lambert, C. Szopa, P. Coll, Astrobiology 15, 221–237 (2015)
K.M. Pontoppidan, A.C.A. Boogert, H.J. Fraser, E.F. van Dishoeck, G.A. Blake, F. Lahuis, K.I. Öberg, I.N.J. Evans, C. Salyk, Astrophys. J. 678, 1005 (2008)
M.S. Potgieter, Adv. Space Res. 46, 402–412 (2010)
S.S. Prasad, S.P. Tarafdar, Astrophys. J. 267, 603–609 (1983)
R. Quinn, A.P. Zent, C.P. McKay, Astrobiology 6, 581–591 (2006)
E. Rabbow, P. Rettberg, S. Barczyk, M. Bohmeier, A. Parpart, C. Panitz, G. Horneck, R. von Heise-Rotenburg, T. Hoppenbrouwers, R. Willnecker et al., Astrobiology 12, 374–386 (2012)
E. Rabbow, A. Parpart, G. Reitz, Microgravity Sci. Technol. 28(3), 215–229 (2015a)
E. Rabbow, P. Rettberg, S. Barczyk, M. Bohmeier, A. Parpart, C. Panitz, G. Horneck, J. Burfeindt, F. Molter, E. Jaramillo et al., Int. J. Astrobiol. 14, 3–16 (2015b)
F. Raulin, P. Bruston, Adv. Space Res. 18, 41–49 (1996)
G. Reitz, R. Beaujean, E. Benton, S. Burmeister, T. Dachev, S. Deme, M. Luszik-Bhadra, P. Olko, Radiat. Prot. Dosim. 116, 374–379 (2005)
P. Rettberg, G. Horneck, A. Zittermann, M. Heer, Adv. Space Res. 22, 1643–1652 (1998)
P. Rettberg, U. Eschweiler, K. Strauch, G. Reitz, G. Horneck, H. Wanke, A. Brack, B. Barbier, in Survival of Microorganisms in Space Protected by Meteorite Material: Results of the Experiment ‘EXOBIOLOGIE’ of the PERSEUS Mission, ed. by M.P. Bernstein, P. Rettberg, R.L. Mancinelli, M.S. Race (Pergamon/Elsevier, Oxford, 2002), pp. 1539–1545
P. Rettberg, E. Rabbow, C. Panitz, G. Horneck, Adv. Space Res. 33, 1294–1301 (2004)
P. Rettberg, R. Moeller, E. Rabbow, T. Douki, J. Cadet, C. Panitz, G. Horneck, H. Lammer, in 37th COSPAR Scientific Assembly, Montreal, Canada (2008), F33-0007-08
A.J. Ricco, J.W. Hines, M. Piccini, M. Parra, L. Timucin, V. Barker, C. Storment, C. Friedericks, E. Agasid, C. Beasley et al., in TRANSDUCERS 2007—2007 International Solid-State Sensors, Actuators and Microsystems Conference (IEEE Press, New York, 2007), pp. 33–37
A.J. Ricco, M. Parra, D. Niesel, M. Piccini, D. Ly, M. McGinnis, A. Kudlicki, J.W. Hines, L. Timucin, C. Beasley et al., in Microfluidics, BioMEMS, and Medical Microsystems IX. Proceedings SPIE, vol. 7929, 79290T (SPIE, Bellingham, 2011) (9 pp.)
R. Rose, J. Dickinson, A. Ridley, in 2012 IEEE Aerospace Conference (IEEE Press, New York, 2012)
L.J. Rothschild, R.L. Mancinelli, Nature 409, 1092–1101 (2001)
G. Rottman, Sol. Phys. 230, 7–25 (2005)
G.J. Rottman, T.N. Woods, W. McClintock, Adv. Space Res. 37, 201–208 (2006)
K. Saiagh, M. Cloix, N. Fray, H. Cottin, Planet. Space Sci. 90, 90–99 (2014)
K. Saiagh, H. Cottin, A. Alean, N. Fray, Astrobiology (2015)
F.J. Sanchez, E. Mateo-Marti, J. Raggio, J. Meessen, J. Martinez-Frias, L.G. Sancho, S. Ott, R. de la Torre, Planet. Space Sci. 72, 102–110 (2012)
L.G. Sancho, R. de la Torre, G. Horneck, C. Ascaso, A. de los Rios, A. Pintado, J. Wierzchos, M. Schuster, Astrobiology 7, 443–454 (2007)
L. Sancho, R. de la Torre, A. Pintado, Fungal Biol. Rev. 22, 103–109 (2008)
K. Sarda, S. Eagleson, E. Caillibot, C. Grant, D. Kekez, F. Pranajaya, R.E. Zee, Acta Astronaut. 59, 236–245 (2006)
G. Scalzi, L. Selbmann, L. Zucconi, E. Rabbow, G. Horneck, P. Albertano, S. Onofri, Orig. Life Evol. Biosph. 42, 253–262 (2012)
L. Selbmann, G.S. De Hoog, A. Mazzaglia, E.I. Friedmann, S. Onofri, Stud. Mycol. 51, 1–32 (2005)
S.M. Seltzer, NIST, Chicago (1994)
D. Selva, D. Krejci, Acta Astronaut. 74, 50–68 (2012)
P. Serrano, A. Hermelink, U. Boettger, J.-P. de Vera, D. Wagner, Planet. Space Sci. 98, 191–197 (2014)
B.R.T. Simoneit, R.E. Summons, L.L. Jahnke, Orig. Life Evol. Biosph. 28, 475–483 (1998)
D.F. Smart, M.A. Shea, Galactic cosmic radiation and solar energetic particles, in Handbook of Geophysics and the Space Environment (Air Force Geophysics Laboratory, Bedford, 1985). Chapter 6
S.W. Squyres, J.P. Grotzinger, R.E. Arvidson, J.F. Bell, W. Calvin, P.R. Christensen, B.C. Clark, J.A. Crisp, W.H. Farrand, K.E. Herkenhoff et al., Science 306, 1709–1714 (2004)
F. Stalport, P. Coll, C. Szopa, F. Raulin, Adv. Space Res. 42, 2014–2018 (2008)
F. Stalport, P. Coll, C. Szopa, H. Cottin, F. Raulin, Astrobiology 9, 543–549 (2009)
F. Stalport, Y.Y. Guan, P. Coll, C. Szopa, F. Macari, F. Raulin, D. Chaput, H. Cottin, Astrobiology 10, 449–461 (2010a)
F. Stalport, Y.Y. Guan, A. Noblet, P. Coll, C. Szopa, F. Macari, A. Person, F. Raulin, D. Chaput, H. Cottin, Planet. Space Sci. 58, 1617–1624 (2010b)
J.P.W. Stark, G.G. Swinerd, in Mission Analysis, ed. by P.W. Fortescue, L.J. Mottershead, G. Swinerd, J.P.W. Stark (Wiley, New York, 2003)
E.C. Stone, A.M. Frandsen, R.A. Mewaldt, E.R. Christian, D. Margolies, J.F. Ormes, in The Advanced Composition Explorer, ed. by C.T. Russell, R.A. Mewaldt, T.T. Von Rosenvinge (Springer, Berlin, 1998), pp. 1–22
G. Strazzulla, M.E. Palumbo, Planet. Space Sci. 46, 1339–1348 (1998)
M. Swartwout, J. Speech Sci. 2, 213–233 (2013)
M. Tabata, H. Yano, H. Kawai, E. Imai, Y. Kawaguchi, H. Hashimoto, A. Yamagishi, Orig. Life Evol. Biosph. 45, 225–229 (2015)
Y. Takano, A. Ohashi, T. Kaneko, K. Kobayashi, Appl. Phys. Lett. 84, 1410–1412 (2004)
G.R. Taylor, J. Spizizen, B.G. Foster, P.A. Volz, H. Bücker, R.C. Simmonds, A.M. Heimpel, E.V. Benton, Bioscience 24, 505–511 (1974)
I.L. Ten Kate, J.R.C. Garry, Z. Peeters, R. Quinn, B. Foing, P. Ehrenfreund, Meteorit. Planet. Sci. 40, 1185 (2005)
G. Thuillier, L. Floyd, T.N. Woods, R. Cebula, E. Hilsenrath, M. Hersé, D. Labs, Adv. Space Res. 34, 256–261 (2004b)
G. Thuillier, L. Floyd, T. Woods, R. Cebula, E. Hisenrath, M. Herse, D. Labs, in Solar Irradiance Reference Spectra, ed. by J.M. Pap, P. Fox (American Geophysical Union, Washington DC, 2004a), pp. 171–194
A.G.G.M. Tielens, Annu. Rev. Astron. Astrophys. 46, 289–337 (2008)
M.G. Trainer, A.A. Pavlov, D.B. Curtis, C.P. McKay, D.R. Worsnop, A.E. Delia, D.W. Toohey, O.B. Toon, M.A. Tolbert, Astrobiology 4, 409–419 (2004)
P. Truscott, QinetiQ Ltd. Farnborough, Hampshire, UK (2010)
S.R. Tsitas, J. Kingston, Aeronaut. J. 114, 689–697 (2010)
A.J. Tylka, J.H. Adams, P.R. Boberg, B. Brownstein, W.F. Dietrich, E.O. Flueckiger, E.L. Petersen, M.A. Shea, D.F. Smart, E.C. Smith, IEEE Trans. Nucl. Sci. 44, 2150–2160 (1997)
J.J.W.A. van Loon, in The Gravity Environment in Space Experiments (Wiley-VCH, Weinheim, 2007), pp. 17–32
F. Vigier, A. Le Postollec, G. Coussot, D. Chaput, H. Cottin, T. Berger, S. Incerti, S. Triqueneaux, M. Dobrijevic, O. Vandenabeele-Trambouze, Adv. Space Res. 52, 2168–2179 (2013)
V. Vinogradoff, N. Fray, F. Duvernay, G. Briani, G. Danger, H. Cottin, P. Theulé, T. Chiavassa, Astron. Astrophys. 551, A128 (2013), 9 pp
V. Vinogradoff, F. Duvernay, N. Fray, M. Bouilloud, T. Chiavassa, H. Cottin, Astrophys. J. Lett. 208, L18 (2015), 6 pp
J.H. Waite, D.T. Young, T.E. Cravens, A.J. Coates, F.J. Crary, B. Magee, J. Westlake, Science 316, 870–875 (2007)
M. Walt, Introduction to Geomagnetically Trapped Radiation (Cambridge University Press, Cambridge, 2005)
M. Wassmann, R. Moeller, G. Reitz, P. Rettberg, Astrobiology 10, 605–615 (2010)
M. Wassmann, R. Moeller, G. Reitz, P. Rettberg, Arch. Microbiol. 193, 823–832 (2011)
M. Wassmann, R. Moeller, E. Rabbow, C. Panitz, G. Horneck, G. Reitz, T. Douki, J. Cadet, H. Stan-Lotter, C.S. Cockell et al., Astrobiology 12, 498–507 (2012)
S. Weiner, P.M. Dove, Rev. Mineral. Geochem. 54, 1–29 (2003)
F. Westall, in Early Life on Earth and Analogies to Mars, ed. by T. Tokano (Springer, Berlin, 2005), p. 45
F. Westall, B. Cavalazzi, in Biosignatures in Rocks, ed. by J. Reitner, V. Thiel (Springer, Dordrecht, 2011), pp. 189–201
F. Westall, C.S. Cockell, Orig. Life Evol. Biosph. 46, 105–106 (2015)
F. Westall, F. Foucher, B. Cavalazzi, S.T. de Vries, W. Nijman, V. Pearson, J. Watson, A. Verchovsky, I. Wright, J.N. Rouzaud et al., Planet. Space Sci. 59, 1093–1106 (2011)
F. Westall, D. Loizeau, F. Foucher, N. Bost, M. Bertrand, J. Vago, G. Kminek, Astrobiology 13, 887–897 (2013)
F. Westall, F. Foucher, N. Bost, M. Bertrand, D. Loizeau, J.L. Vago, G. Kminek, F. Gaboyer, K.A. Campbell, J.-G. Bréhéret et al., Astrobiology 15, 998–1029 (2015)
K. Woellert, P. Ehrenfreund, A.J. Ricco, H. Hertzfeld, Adv. Space Res. 47, 663–684 (2011)
I.P. Wright, S. Sheridan, S.J. Barber, G.H. Morgan, D.J. Andrews, A.D. Morse, Science 349(6247), aab0673 (2015)
M.A. Xapsos, G.P. Summers, E.A. Burke, Sol. Phys. 183, 157–164 (1998)
M.A. Xapsos, C. Stauffer, T. Jordan, J.L. Barth, R.A. Mewaldt, IEEE Trans. Nucl. Sci. 54, 1985–1989 (2007)
A. Yamagishi, H. Yano, K. Okudaira, K. Kobayashi, S.I. Yokobori, M. Tabata, H. Kawai, M. Yamashita, H. Hashimoto, H. Naraoka et al., Trans. Jpn. Soc. Aeronaut. Space Sci. 7, Tk_49–Tk_55 (2009)
B.D. Yost, J.L. Fishman, M. Fonda, 2007
C. Zeitlin, D.M. Hassler, F.A. Cucinotta, B. Ehresmann, R.F. Wimmer-Schweingruber, D.E. Brinza, S. Kang, G. Weigle, S. Boettcher, E. Boehm et al., Science 340, 1080–1084 (2013)
Acknowledgements
This work has been supported by ESA, through a Topical Team grant from 2011 to 2016. We are grateful to Oliver Angerer, Jason Hatton, René Demets from ESA for their help and support. UPEC and LISA provided important administrative help (Mireille Paul, Sabrina Hammache, Valérie Simard, Charlotte Laurent, Nicolas Vautrin). Hervé Cottin acknowledges support from CNES and Labex ESEP (N° 2011-LABX-030). Kafila Saiagh acknowledges support from DIM ACAV (region Ile de France). Zita Martins is funded by a Royal Society University Research Fellowship (grant UF080820 and grant UF130324). Pascale Ehrenfreund and Richard Quinn acknowledge support from the NASA Astrobiology Institute. Jean-Pierre de Vera acknowledges the Helmholtz-Gemeinschaft for financial support through the Helmholtz-Alliance Planetary Evolution and Life. Daniela Billi and Silvano Onofri acknowledge Italian Space Agency (ASI) for financial support of the projects BIOMEX-Cyano, BOSS-Cyano, BIOMEX-MCF and E-GEM. Nathalie Carrasco acknowledges the financial support of the European Research Council (ERC Starting Grant PRIMCHEM, grant agreement n°636829). Hirofumi Hashimoto, Shin-ichi Yokobori, Eiichi Imai, Issay Narumi and the Tanpopo team are also warmly thanked. The two anonymous reviewers of this paper are warmly thanked for their detailed and constructive comments. Open Access for this paper has been funded by CNES.
Author information
Authors and Affiliations
Corresponding author
Additional information
Note by the editor: This is a Special Communication, supplementing the papers by Cottin et al. on “Astrobiology and the Possibility of Life on Earth and Elsewhere…”, 2015, Space Science Reviews, doi:10.1007/s11214-015-0196-1 and Martins et al. (2017) “Earth as a Tool for Astrobiology—A European Perspective”, Space Science Reviews; doi:10.1007/s11214-017-0369-1.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Cottin, H., Kotler, J.M., Billi, D. et al. Space as a Tool for Astrobiology: Review and Recommendations for Experimentations in Earth Orbit and Beyond. Space Sci Rev 209, 83–181 (2017). https://doi.org/10.1007/s11214-017-0365-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11214-017-0365-5