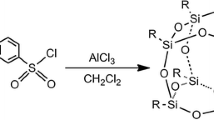
Abstract
The bromination and characterization of octaphenylsilsesquioxane is reported. The reaction is successful with a variety of methods, including microwave assisted synthesis. The analysis of the obtained product mix was carried out with a combination of high performance liquid chromatography and matrix assisted laser desorption ionization time-of-flight mass spectrometry (MALDI-TOF-MS). The analysis indicates that the bromination leads to multiple bromines per octaphenylsilsesquioxane molecule with a distribution dominated by statistical odds and the reaction conditions. The results showed that the bromination is not selective and does not lead to a single defined product.
Similar content being viewed by others
Abbreviations
- MALDI-TOF-MS:
-
matrix assisted laser desorption ionization time-of-flight mass spectra
- POSS:
-
polyhedral oligomeric silsesquioxane
- OPS:
-
octaphenylsilsesquioxane
References
Harrison, P.G. 1997, J. Organometallic Chemistry 542, 141–183.
Sellinger, A. & Laine, R.M. 1996, Macromolecules 29, 2327–2330.
Sellinger, A. & Laine, R.M. 1996, Chem. Mater. 8, 1592–1593.
Zhang, C., Babonneau, F., Bonhomme, C., Laine, R.M., Soles, C.L., Hristov, H.A., Yee, & A.F. 1998, J. Am. Chem. Soc. 120, 8380–8391.
Choi, J., Kim, S.G., & Laine, R.M. 2004, Macromolecules 37, 99–109.
Schwab, J.J. & Lichtenhan, J.D. 1998, Appl. Organomet. Chem. 12, 707.
Laine, R.M., Tamaki, R., & Choi, J. 2005, U. S. Patent 6,927,301 B2.
Su, R. Q., Müller, T.E., Procházka, J., & Lercher, J.A. 2002, Adv. Mater. 14(19), 1369–1373.
Tegou, E., Bellas, V., Gogolides, E., Argitis, P., Eon, D., Cartry, G., & Cardinaud, C. 2004, Chem. Mater. 16, 2567–2577.
Feher, F.J. & Wyndham, K.D. 1998, Chem. Commun. 323–324.
Voronkov, M.G. & Lavrent'ev, V. 1982, Top. Curr. Chem. 102, 199.
Feher, F.J. & Budzichowski, T.A. 1989, J. Organometallic Chem. 379, 33–40.
Bassindale, A.R., Liu, Z., MacKinnon, I.A., Taylor, P.G., Yang, Y., Light, M.E., Horton, P.N., & Hursthouse, M. B. 2003, Dalton Trans. 2945–2949.
Sprung, M.M. & Guenther, F.O. 1955, J. Am. Chem. Soc. 77, 3996–4002.
Sprung, M.M. & Guenther, F.O. 1955, J. Am. Chem. Soc. 77, 6045–6047.
Scott, D.W. 1946, J. Am. Chem. Soc. 68, 356–359.
Olsson, K. & Grönwall, C. 1966, Arkiv for Kemi 17(54), 529–540.
Brown, J.F., Jr., Vogt, L.H., Jr., & Prescott, P.I. 1964, J. Am. Chem. Soc. 86, 1120–1125.
Brown, J.F., Jr. & Vogt, L.H., Jr. 1965, J. Am. Chem. Soc. 87(19), 4313–4317.
(a) He, C., Xiao, Y., Huang, J., Lin, T., Mya, K.Y., & Zhang, X. 2004, J. Am. Chem. Soc. 126, 7792–7793; (b) It has been pointed out during the review of this article that the Suzuki coupling, not recommended by He et al., is a viable synthetic method for polyaromatic octasilsesquioxane (see ref. [22]).
Suppliers include Aldrich, HybridPlastics and Mayaterials. Octabromophenyl-octasilsesquioxane is available from Mayaterials.
Brick, C.M., Ouchi, Y., Chujo, Y., & Laine, R.M. 2005, Macromolecules 38, 4661–4665.
Brick, C.M., Tamaki, R., Kim, S.-G., Asuncion, M.Z., Roll, M., Nemoto, T., Ouchi, Y., Chujo, Y., & Laine, R.M. 2005, Macromolecules 38, 4655–4660.
Militzer, W. 1938, J. Am. Chem. Soc. 60(2), 256–257.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Erben, C., Grade, H. & Goddard, G.D. Bromination of octaphenylsilsesquioxane. Silicon Chem 3, 43–49 (2006). https://doi.org/10.1007/s11201-005-9000-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11201-005-9000-5