Abstract
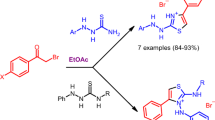
Acylation of indole and 2,5-dimethylthiophene with 2-(3-indolyl)-2-oxoacetyl chloride afforded the corresponding diketones. 1-(2,5-Dimethyl-3-thienyl)-2-(3-indolyl)ethanedione reacted with thiosemicarbazide under atmosperic and elevated pressure to give 6-(2,5-dimethyl-3-thienyl)-5-(3-indolyl)-2,3-dihydro-1,2,4-triazine-3-thione whose structure was studied in detail by the X-ray diffraction method. Reactions of 6-(2,5-dimethyl-3-thienyl)-5-(3-indolyl)-2,3-dihydro-1,2,4-triazine-3-thione with amines and hydrazine resulted in formation of fused triazolo- and tetrazolotriazines.
Similar content being viewed by others
REFERENCES
Irie, M., Chem. Rev., 2000, vol. 100, p. 1685.
Beccalli, E.M., Gelmi, M.L., and Marchesini, A., Eur. J. Org. Chem., 1999, p. 1421.
Pereira, E.R., Sancelme, M., Voldoire, A., and Prudhomme, M., Bioorg. Med. Chem. Lett., 1997, vol. 7, p. 2503.
Hughes, T.V. and Cava, M.P., Tetrahedron Lett., 1998, vol. 39, p. 9629.
Davis, P.D., Hill, C.H., Lawton, G., Nixon, J.S., Wilkinson, S.E., Hurst, S.A., Keech, E., and Turner, S.E., J. Med. Chem., 1992, vol. 35, p. 177.
Ivanov, S.N., Lichitskii, B.V., Dudinov, A.A., Martynkin, A.Yu., and Krayushkin, M.M., Khim. Geterotsikl. Soedin., 2001, p. 89.
Millich, F. and Becker, E.I., J. Org. Chem., 1958, vol. 23, p. 1096.
Ouchi, Y., Saito, H., and Hatayama, K., JPN Patent no. 02-188 579, 1990; Chem. Abstr., 1990, vol. 113, no. 211 985 q.
Krayushkin, M.M., Shirinyan, V.Z., Belen’kii, L.I., Shadronov, A.Yu., Vorontsova, L.G., and Starikova, Z.A., Izv. Ross. Akad. Nauk, Ser. Khim., 2002, p. 1392.
Matsumoto, K., Kaneko, M., Katsura, H., Hayashi, N., Uchida, T., and Acheeson, R.M., Heterocycles, 1998, vol. 47, p. 1135.
Zavarzin, I.V., Zhulin, V.M., Yarovenko, V.N., and Krayushkin, M.M., Izv. Akad. Nauk SSSR, Ser. Khim., 1988, p. 1168.
Gazieva, G.A., Lyssenko, K.A., Gaziev, R.G., Kravchenko, A.N., Lebedev, O.V., and Zhulin, V.M., Mendeleev Commun., 2001, p. 107.
Database of Cambridge Crystallographic Data Centre, 2003, CSD version 5.24 (Jul).
Krayushkin, M.M., Vorontsova, L.G., and Uzhinov, B.M., Int. J. Photoenergy, 2001, vol. 3, p. 25.
Zefirov, Yu.V. and Zorkii, P.M., Usp. Khim., 1995, vol. 64, p. 446.
Allen, F.H., Kennard, O., Watson, D.G., Brammer, L., Orpen, A.G., and Taylor, R., J. Chem. Soc., Perkin Trans. 2, 1987, p. S1.
Kishikawa, K., Iwashima, C., Yamaguchi, K., and Yamamoto, M., J. Chem. Soc., Perkin Trans. 1, 2000, p. 2217.
Golubev, S.N. and Kondrashev, Yu.D., Zh. Strukt. Khim., 1984, vol. 25, p. 143.
Zefirov, Yu.V., Kristallografiya, 1998, vol. 43, p. 313.
Krayushkin, M.M., Ivanov, S.N., Martynkin, A.Yu., Lichitskii, B.V., Dudinov, A.A., and Uzhinov, B.M., Izv. Ross. Akad. Nauk, Ser. Khim., 2001, p. 2315.
Krayushkin, M.M., Ivanov, S.N., Martynkin, A.Yu., Lichitskii, B.V., Dudinov, A.A., Vorontsova, L.G., Starikova, Z.A., and Uzhinov, B.M., Izv. Ross. Akad. Nauk, Ser. Khim., 2002, p. 1588.
Toda, F., Tanaka, K., and Tange, H., J. Chem. Soc., Perkin Trans. 1, 1989, p. 1555.
Zoeller, J.R. and Ackerman, C.J., J. Org. Chem., 1990, vol. 55, p. 1354.
Stevens, M.F.G., J. Chem. Soc., Perkin Trans. 1, 1972, p. 1221.
Nikishin, G.I., Spektor, S.S., Shakhovskoi, G.P., Glukhovtsev, V.G., and Zhulin, V.M., Izv. Akad. Nauk SSSR, Ser. Khim., 1976, p. 1664.
Bruker. SMART. Bruker Molecular Analysis Research Tool. V. 5.059, Madison, Wisconsin, USA: Bruker AXS, 1998.
Sheldrick, G.M., SHELXTL v. 5.10, Structure Determination Software Suite, Madison, Wisconsin, USA: Bruker AXS, 1998.
Author information
Authors and Affiliations
Additional information
Dedicated to Full Member of the Russian Academy of Sciences V.I. Minkin on his Jubilee
__________
Translated from Zhurnal Organicheskoi Khimii, Vol. 41, No. 6, 2005, pp. 895–902.
Original Russian Text Copyright © 2005 by Krayushkin, Yarovenko, Sedishev, Zavarzin, Vorontsova, Starikova.
Rights and permissions
About this article
Cite this article
Krayushkin, M.M., Yarovenko, V.N., Sedishev, I.P. et al. Synthesis and Structure of 5-Indolyl-6-thienyl-1,2,4-triazines. Russ J Org Chem 41, 875–883 (2005). https://doi.org/10.1007/s11178-005-0258-z
Received:
Issue Date:
DOI: https://doi.org/10.1007/s11178-005-0258-z