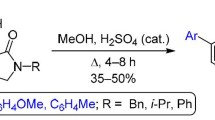
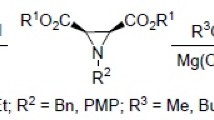
Abstract
The reaction mechanisms of cyclic 1,3-diketones with trialkyl orthoformates and trimethyl orthoacetate in the absence of activators were theoretically and experimentally studied. The reactions proceed via C-C-coupling of the ionized forms of the reactants giving dialkyl acetals, which further transform into vinyl ethers. The reactions of the latter with ortho esters afford aldehydes (ketones). Related 2-acetylcyclopentanone reacts with trimethyl orthoacetate under argon to form a mixture of methyl enol ethers, whereas in the presence of air oxygen dimethyl glutarate is formed.
Similar content being viewed by others
References
Z. Khademi, K. Nikoofar, RSC Adv., 2020, 10, 30314; DOI: https://doi.org/10.1039/d0ra05276k.
A. V. Komkov, T. V. Potapova, M. I. Zuev, S. V. Baranin, Yu. N. Bubnov, Russ. Chem. Bull., 2019, 68, 365; DOI: https://doi.org/10.1007/s11172-019-2394-6.
R. H. DeWolfe, Carboxylic Ortho Acid Derivatives: Preparations and Synthetic Applications, Acad. Press, New York-London, 1970.
V. V. Mezheritskii, E. P. Olekhnovich, S. M. Luk’yanov, G. N. Dorofeenko, Ortoefiry v organicheskom sinteze [Ortho Esters in Organic Synthesis], Izd-vo Rostov. Gos. Un-ta, Rostov-on-Don, 1976, 176 pp. (in Russian).
U. Pindur, Acid Derivatives, John Wiley and Sons Ltd., New York, 1992; DOI: https://doi.org/10.1002/9780470772508.
V. V. Mezheritskii, E. P. Olekhnovich, G. N. Dorofeenko, Russ. Chem. Rev., 1973, 42, 392; DOI: https://doi.org/10.1070/RC1973v042n05ABEH002612.
R. H. DeWolfe, Synthesis, 1974, 153; DOI: https://doi.org/10.1055/s-1974-23268.
L. A. Pavlova, Yu. A. Davidovich, S. V. Rogozhin, Russ. Chem. Rev., 1986, 55, 1026; DOI: https://doi.org/10.1070/RC1986v055n11ABEH003238.
S. Ghosh, U. R. Ghatak, Proc. Indian Acad. Sci. Chem. Sci., 1988, 100, 235; DOI: https://doi.org/10.1007/BF02839451.
R. A. Fernandes, A. K. Chowdhury, P. Kattanguru, Eur. J. Org. Chem., 2014, 2833; DOI: https://doi.org/10.1002/ejoc.201301033.
Y. Chen, Chem. - Eur. J., 2019, 25, 3405; DOI: https://doi.org/10.1002/chem.201803642.
Z. Nazarian, M. Dabiri, ChemistrySelect, 2020, 5, 4394; DOI: https://doi.org/10.1002/slct.201903901.
D. L. Rakhmankulov, O. B. Chalova, T. K. Kiladze, E. A. Kantor, R. A. Karakhanov, M. Bartok, A. Molnar, Acta Phys. Chem., 1982, 28, 181; WOS: A1982QN45900009.
D. L. Rakhmankulov, V. V. Zorin, F. N. Latypova, S. S. Zlotsky, R. A. Karakhanov, Russ. Chem. Rev., 1983, 52, 350; DOI: https://doi.org/10.1070/RC1983v052n04ABEH002822.
F. N. Latypova, F. Sh. Vil’danov, R. R. Chanyshev, S. S. Zlotsky, Izv. Vuzov. Khim. Khim. Tekhnol. [Bulletin of Higher Educational Institutions. Chemistry and Chemical Technology], 2015, 58, No, 8, 3 (in Russian); journals.isuct.ru/public/journals/2/2015/v58-_n08_2015_full.pdf.
K. Undheim, Synthesis, 2017, 49, 705; DOI: https://doi.org/10.1055/s-0036-1588339.
U. Pindur, J. Muller, C. Flo, H. Witzel, Chem. Soc. Rev., 1987, 16, 75; DOI: https://doi.org/10.1039/as9871600075.
F. Perron-Sierra, M. A. Promo, V. A. Martin, K. F. Albizati, J. Org. Chem., 1991, 56, 6188; DOI: https://doi.org/10.1021/jo00021a043.
H. W. Post, E. R. Erickson, J. Org. Chem., 1937, 2, 260; DOI: https://doi.org/10.1021/jo01226a008.
R. C. Fuson, W. E. Parham, L. J. Reed, J. Org. Chem., 1946, 11, 194; DOI: https://doi.org/10.1021/jo01172a014.
Yu. S. Kudyakova, D. N. Bazhin, M. V. Goryaeva, Ya. V. Burgart, V. I. Saloutin, Russ. Chem. Rev., 2014, 83, 120; DOI: https://doi.org/10.1070/RC2014v083n02ABEH004388.
R. G. Jones, J. Am. Chem. Soc., 1952, 74, 4889; DOI: https://doi.org/10.1021/ja01139a-046.
T.-L. Su, J.-T. Huang, T.-Ch. Chou, G. M. Otter, F. M. Sirotnak, K. A. Watanabe, J. Med. Chem., 1988, 31, 1209; DOI: https://doi.org/10.1021/jm00401a023.
Pat. WO 2011113789; Chem. Abstr., 2011, 155, 431861.
M. V. Pryadeina, Ya. V. Burgart, V. I. Saloutin, P. A. Slepukhin, O. N. Kazheva, G. V. Shilov, O. A. D’yachenko, O. N. Chupakhin, Russ. J. Org. Chem., 2007, 43, 945; DOI: https://doi.org/10.1134/S107042800707001.
V. I. Saloutin, Z. E. Skryabina, I. T. Bazyl’, O. N. Chupakhin, J. Fluorine Chem., 1993, 65, 37; DOI: https://doi.org/10.1016/S0022-1139(00)80470-4.
E. B. Knott, J. Chem. Soc., 1954, 1482; DOI: https://doi.org/10.1039/JR9540001482.
J. Soulier, M. Farines, R. M. Authier, M. Fournier, J. Heterocycl. Chem., 1976, 13, 1125; DOI: https://doi.org/10.1002/jhet.5570130543.
R. T. Akhmatdinov, O. B. Chalova, E. A. Kantor, D. L. Rakhmankulov, Russ. J. Org. Chem., 1980, 16, 962.
A. Kankaanperä, M. Mattsén, Acta Chem. Scand., 1975, 29A, 419; DOI: https://doi.org/10.3891/acta.chem.scand.29a-0419.
L. A. Yanovskaya, S. S. Yufit, V. F. Kucherov, in Khimiya atsetalei [Chemistry of Acetals], Nauka, Moscow, 1975, pp. 138 and 217 (in Russian).
S. Forsen, F. Merenyi, M. Nilsson, Acta. Chem. Scand., 1967, 21, 620; DOI: https://doi.org/10.3891/acta.chem.scand.21-0620.
T. Miki, K. Hiraga, T. Asako, H. Masuya, Chem. Pharm. Bull., 1967, 15, 670; DOI: https://doi.org/10.1248/cpb.15.670.
Yu. A. Pentin, L. V. Vilkov, in Fizicheskie metody issledovaniya v khimii [Physical Methods of Investigation in Chemistry], Mir, Moscow, 2003, p. 454 (in Russian).
V. V. Gromak, J. Mol. Struct.: THEOCHEM, 2005, 726, 213; DOI: https://doi.org/10.1016/j.theochem.2005.01.0436.
M. C. Etter, D. L. Parker, S. R. Ruberu, T. W. Panunto, D. Britton, J. Inclusion Phenom. Mol. Recognit. Chem., 1990, 8, 395; DOI: https://doi.org/10.1007/BF01041196.
R. E. Valters, Kol’chato-tsepnaya isomeriya v organicheskoi khimii [Ring-Chain Isomerism in Organic Chemistry], Zinatne, Riga, 1978, 238 pp. (in Russian).
R. E. Valters, F. Fulop, D. Korbonits, Adv. Heterocycl. Chem., 1996, 66, 1; DOI: https://doi.org/10.1016/S0065-2725(08)60304-9.
V. I. Vysotskii, V. A. Kaminskii, T. I. Akimova, O. Yu. Slabko, A. N. Andin, N. P. Bagrina, 1,5-Dikarbonil’nye soedineniya v organicheskom sinteze [1,5-Dicarbonyl Compounds in Organic Synthesis], Izd-vo Dal’nevostoch. Fed. Un-ta, Vladivostok, 2014, 390 pp. (in Russian).
R. M. Mohareb, F. M. Manhi, M. A. A. Mahmoud, A. Abdelwahab, Med. Chem. Res., 2020, 29, 1536; DOI: https://doi.org/10.1007/s00044-020-02579-4.
E. G. Meek, J. H. Turnbull, W. Wilson, J. Chem. Soc., 1953, 811; DOI: https://doi.org/10.1039/JR9530000811.
V. P. Glazunov, D. V. Berdyshev, V. A. Denisenko, J. Appl. Spectr., 2011, 78, 489; DOI: https://doi.org/10.1007/s10812-011-9489-7.
V. L. Novikov, O. P. Shestak, Russ. Chem. Bull., 2012, 61, 1099; DOI: https://doi.org/10.1007/s11172-012-0149-8.
M. A. P. Martins, G. P. Bastos, A. P. Sinhorin, A. F. C. Flores, H. G. Bonacorso, N. Zanatta, Synlett, 1999, 6, 789; DOI: https://doi.org/10.1055/s-1999-2725.
P. J. Stephens, F. J. Devlin, C. F. Chabalowski, M. J. Frisch, J. Phys Chem., 1994, 98, 11623; DOI: https://doi.org/10.1021/j100096a001.
S. Miertus, E. Scrocco, J. Tomasi, Chem. Phys., 1981, 55, 117; DOI: https://doi.org/10.1016/0301-0104(81)85090-2.
Gaussian 16 W, Version 1.1, Gaussian Inc., Wallingford (CT), 2019.
V. F. Kucherov, L. N. Ivanova, Dokl. Akad. Nauk SSSR [Reports Acad. Sci. USSR], 1960, 131, 1077; http://www.mathnet.ru (in Russian).
V. L. Novikov, O. P. Shestak, A. V. Kamernitsky, G. B. Elyakov, Bull. Acad. Sci. USSR. Div. Chem. Sci., 1980, 1002; DOI: https://doi.org/10.1007/BF00958826.
H. Roedig, L. Hornig, Chem. Ber., 1955, 88, 2003; DOI: https://doi.org/10.1002/cber.19550881232.
F. Merényi, M. Nilsson, Acta Chem. Scand., 1963, 17, 1801; DOI: https://doi.org/10.3891/acta.chem.scand.17-1801.
B. D. Akehurst, J. R. Bartels-Keith, J. Chem. Soc., 1957, 4798; DOI: https://doi.org/10.1039/JR570004798.
A. A. Akhrem, F. A. Lakhvich, S. I. Budai, T. S. Khlebnicova, I. I. Petrusevich, Synthesis, 1978, 925; DOI: https://doi.org/10.1055/s-1978-24943.
O. P. Shestak, V. L. Novikov, E. A. Martyyas, M. M. Anisimov, Pharm. Chem. J., 2009, 43, 498; DOI: https://doi.org/10.1007/s11094-009-0338-4.
O. P. Shestak, V. L. Novikov, Russ. Chem. Bull., 2010, 59, 81; DOI: https://doi.org/10.1007/s11172-010-0048-9.
Author information
Authors and Affiliations
Additional information
This paper does not contain descriptions of studies on animals or humans.
The authors declare no competing interests.
Published in Russian in Izvestiya Akademii Nauk. Seriya Khimicheskaya, No. 8, pp. 1584–1598, August, 2021.
Rights and permissions
About this article
Cite this article
Balaneva, N.N., Shestak, O.P., Novikov, V.L. et al. Activator-free reactions of carboxylic ortho esters with cyclic β-diketones. Russ Chem Bull 70, 1584–1598 (2021). https://doi.org/10.1007/s11172-021-3255-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11172-021-3255-7