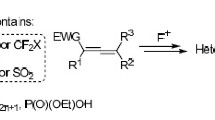
Abstract
The present review is focused on the main transformation routes of sp2- and sp3-hybridized carbanions derived from the compounds containing two or more fused N,O,S-heterocycles. The rearrangement and fragmentation reactions and their applications in the synthesis of poorly available organic compounds are discussed. In the review, separate consideration is given to the reactions leading to a decrease, retention, and increase in the number of annulated heterocycles.
Similar content being viewed by others
References
J.-M. L’Helgoual’ch, A. Seggio, F. Chevallier, M. Yonehara, E. Jeanneau, M. Uchiyama, F. Mongin, J. Org. Chem., 2008, 73, 177; DOI: https://doi.org/10.1021/jo7020345.
L. Marzo, I. Perez, F. Yuste, J. Aleman, J. L. G. Ruano, Chem. Commun., 2015, 51, 346; DOI: https://doi.org/10.1039/c4cc07574a.
Topics in Heterocyclic Chemistry, Eds M. Schnürch, M. D. Mihovilovic, Springer, London, 2013, Vol. 31, 272 pp.; DOI: https://doi.org/10.1007/978-3-642-35022-1.
H. K. Khartabil, P. C. Gros, Y. Fort, M. F. Ruiz-Lopez, J. Am. Chem. Soc., 2010, 132, 2410; DOI: https://doi.org/10.1021/ja910350q.
M. Schlosser, F. Mongin, Chem. Soc. Rev., 2007, 36, 1161; DOI: https://doi.org/10.1039/b706241a.
M. Balkenhohl, P. Knochel, SynOpen, 2018, 2, 78; DOI: https://doi.org/10.1055/s-0036-1591966.
S. Javed, D. M. Hoffman, Dalton Trans., 2010, 39, 11439; DOI: https://doi.org/10.1039/c0dt00847h.
C. Samann, E. Coya, P. Knochel, Angew. Chem., Int. Ed., 2014, 53, 1430; DOI: https://doi.org/10.1002/anie.201309217.
K. M. Luedecke, Y. Wang, P. M. Tran, H. L. Threlkeld, P. Wei, Y. Xie, H. F. Schaefer, G. H. Robinson, Organometallics, 2020, 39, 4178; DOI: https://doi.org/10.1021/acs.organomet.0c00201.
I. Avinash, S. Parveen, G. Anantharaman, Inorg. Chem., 2020, 59, 5646; DOI: https://doi.org/10.1021/acs.inorgchem.0c00348.
F. Bellina, R. Rossi, Adv. Synth. Catal., 2010, 352, 1223; DOI: https://doi.org/10.1002/adsc.201000144.
M. Mosrin, P. Knochel, Chem. Eur. J., 2009, 15, 1468; DOI: https://doi.org/10.1002/chem.200801831.
K. Groll, S. M. Manolikakes, X. M. du Jourdin, M. Jaric, A. Bredihhin, K. Karaghiosoff, T. Carell, P. Knochel, Angew. Chem., Int. Ed., 2013, 52, 6776; DOI: https://doi.org/10.1002/anie.201301694.
A. Seggio, F. Chevallier, M. Vaultier, F. Mongin, J. Org. Chem., 2007, 72, 6602; DOI: https://doi.org/10.1021/jo0708341.
F. H. Lutter, L. Grokenberger, L. A. Perego, D. Broggini, S. Lemaire, S. Wagschal, P. Knochel, Nature Commun., 2020, 11, 4443; DOI: https://doi.org/10.1038/s41467-020-18188-z.
S. H. Wiedemann, M. M. Bio, L. M. Brown, K. B. Hansen, N. F. Langille, Synlett, 2012, 23, 2231; DOI: https://doi.org/10.1055/s-0032-1316737.
J. Y. F. Wong, A. Lewandowska, B. R. Trowse, G. Barker, Org. Lett., 2019, 21, 7069; DOI: https://doi.org/10.1021/acs.orglett.9b02633.
S. M. Ivanov, J. Heterocycl. Chem., 2020, 57, 3510; DOI: https://doi.org/10.1002/jhet.4071.
Topics in Heterocyclic Chemistry, Eds B. U. W. Maes, G. W. Gribble, Springer, London, 2012, Vol. 29, 445 pp.; DOI: https://doi.org/10.1007/978-3-642-31791-0.
R. Chinchilla, C. Nájera, M. Yus, ARKIVOC, 2007, Part x, 152; DOI: https://doi.org/10.3998/ark.5550190.0008.a13.
S. B. Mogensen, M. K. Taylor, J.-W. Lee, Molecules, 2020, 25, 5950; DOI:https://doi.org/10.3390/molecules25245950.
Science of Synthesis: Houben-Weyl Methods of Molecular Transformations, Eds M. Majewski, V. Snieckus, M. A. Y. Astiz, Georg Thieme Verlag, New York, 2005, Vol. 8a, 952 pp.
B. Haag, M. Mosrin, H. Ila, V. Malakhov, P. Knochel, Angew. Chem., Int. Ed., 2011, 50, 9794; DOI: https://doi.org/10.1002/anie.201101960.
G. A. El-Hiti, K. Smith, A. S. Hegazy, M. B. Alshammari, A. M. Masmali, ARKIVOC, 2015, Part iv, 19; DOI: https://doi.org/10.24820/ark.5550190.p008.744.
Y. Fort, C. Comoy, in Lithium Compounds in Organic Synthesis, Eds R. Luisi, V. Capriati, Wiley-VCH, Weinheim, 2014, pp. 423–462; DOI: https://doi.org/10.1002/9783527667512.ch15.
E. A. Chugunova, A. S. Gazizov, A. R. Burilov, L. M. Yusupova, M. A. Pudovik, O. G. Sinyashin, Russ. Chem. Bull., 2019, 68, 887; DOI: https://doi.org/10.1007/s11172-019-2503-6.
L. Bischoff, in Comprehensive Heterocyclic Chemistry III, Eds A. R. Katritzky, C. A. Ramsden, E. F. V. Scriven, R. J. K. Taylor, Elsevier, Oxford, 2008, pp. 589–627; DOI: https://doi.org/10.1016/B978-008044992-0.01013-0.
V. Gayakhe, Y. S. Sanghvi, I. J. S. Fairlamb, A. R. Kapdi, Chem. Commun., 2015, 51, 11944; DOI: https://doi.org/10.1039/c5cc03416g.
Yu. G. Shtyrlin, A. S. Petukhov, A. D. Strelnik, N. V. Shtyrlin, A. G. Iksanova, M. V. Pugachev, R. S. Pavelyev, M. S. Dzyurkevich, M. R. Garipov, K. V. Balakin, Russ. Chem. Bull., 2019, 68, 911; DOI: https://doi.org/10.1007/s11172-019-2504-5].
V. N. Charushin, O. N. Chupakhin, Russ. Chem. Bull., 2019, 68, 453; DOI: https://doi.org/10.1007/s11172-019-2441-3.
N. N. Makhova, L. I. Belen’kii, G. A. Gazieva, I. L. Dalinger, L. S. Konstantinova, V. V. Kuznetsov, A. N. Kravchenko, M. M. Krayushkin, O. A. Rakitin, A. M. Starosotnikov, L. L. Fershtat, S. A. Shevelev, V. Z. Shirinian, V. N. Yarovenko, Russ. Chem. Rev., 2020, 89, 55; DOI: https://doi.org/10.1070/RCR4914.
L.-H. Chung, S.-W. Ng, C.-F. Yeung, H.-L. Shek, S.-Y. Tse, H.-S. Lo, S.-C. Chan, M.-K. Tse, S.-M. Yiu, C.-Y. Wong, Dalton Trans., 2018, 47, 12838; DOI: https://doi.org/10.1039/C8DT02408A.
T. D. Lash, Macroheterocycles, 2008, 1, 9.
S. A. Al-Jibori, H. M. Gergees, M. S. Al-Rubaye, S. Basak-Modi, S. Ghosh, H. Schmidt, M. Laguna, M. A. Luquin, G. Hogarth, Inorg. Chim. Acta, 2016, 450, 50; DOI: https://doi.org/10.1016/j.ica.2016.04.046.
G. Albertin, S. Antoniutti, M. Bortoluzzi, J. Castro, F. Sibilla, E. Trave, New J. Chem., 2017, 41, 12976; DOI: https://doi.org/10.1039/c7nj02224g.
C. G. Neochoritis, T. Zhao, A. Dömling, Chem. Rev., 2019, 119, 1970; DOI: https://doi.org/10.1021/acs.chemrev.8b00564.
L. Cuesta, D. Gross, V. M. Lynch, Z. Ou, W. Kajonkijya, K. Ohkubo, S. Fukuzumi, K. M. Kadish, J. L. Sessler, J. Am. Chem. Soc., 2007, 129, 11696; DOI: https://doi.org/10.1021/ja075613r.
Á. Vivancos, C. Segarra, M. Albrecht, Chem. Rev., 2018, 118, 9493; DOI: https://doi.org/10.1021/acs.chemrev.8b00148.
J. Pan, J. Wang, M. M. B. Holl, J. W. Kampf, A. J. Ashe, Organometallics, 2006, 25, 3463; DOI: https://doi.org/10.1021/om060239+.
T. R. Helgert, C. E. Webster, T. K. Hollis, H. U. Valle, P. Hillesheim, A. G. Oliver, Inorg. Chim. Acta, 2018, 469, 164; DOI: https://doi.org/10.1016/j.ica.2017.08.050.
N. V. Onvamboko, R. Weber, A. Fauconnier, A. Renson, Bull. Soc. Chim. Belg., 1983, 92, 53; DOI: https://doi.org/10.1002/bscb.19830920108.
B. Hill, M. De Vleeschauwer, K. Houde, M. Belley, Synlett, 1998, 407; DOI: https://doi.org/10.1055/s-1998-1674.
L. S. Fuller, B. Iddon MP, K. A. Smith, Chem. Commun., 1997, 2355; DOI: https://doi.org/10.1039/A707033K.
R. Hull, P. J. van den Broek, M. L. Swain, J. Chem. Soc., Perkin Trans. 1, 1975, 2271; DOI: https://doi.org/10.1039/P19750002271.
I. Iijima, K. C. Rice, J. Heterocycl. Chem., 1978, 15, 1527; DOI: https://doi.org/10.1002/jhet.5570150863.
H. Reimlinger, W. R. F. Lingier, J. J. M. Vandewalle, R. Merényi, Chem Ber., 1971, 104, 3965; DOI: https://doi.org/10.1002/cber.19711041227.
H. Reimlinger, W. R. F. Lingier, J. J. M. Vandewalle, Chem. Ber., 1971, 104, 3940; DOI: https://doi.org/10.1002/cber.19711041223.
S. M. Ivanov, A. M. Shestopalov, J. Heterocycl. Chem., 2019, 56, 2210; DOI: https://doi.org/10.1002/jhet.3615.
S. M. Ivanov, A. O. Dmitrienko, M. G. Medvedev, L. M. Mironovich, J. Organomet. Chem., 2019, 896, 168; DOI: https://doi.org/10.1016/j.jorganchem.2019.06.009.
S. M. Ivanov, Tetrahedron Lett., 2020, 61, 152404; DOI: https://doi.org/10.1016/j.tetlet.2020.152404.
S. Shiotani, H. Morita, J. Heterocycl. Chem., 1992, 29, 413; DOI: https://doi.org/10.1002/jhet.5570290221.
R. L. Meline, R. L. Elsenbaumer, Synthesis, 1997, 617; DOI: https://doi.org/10.1055/s-1997-1407.
R. L. Meline, R. L. Elsenbaumer, J. Chem. Soc., Perkin Trans. 1, 1998, 2467; DOI: https://doi.org/10.1039/A805226C.
R. Andreu, J. Gar’in, J. Orduna, J. M. Royo, Tetrahedron Lett., 2001, 42, 875; DOI: https://doi.org/10.1016/S0040-4039(00)02147-X.
D. Liu, S. A. Kozmin, Angew. Chem., Int. Ed., 2001, 40, 4757; DOI: https://doi.org/10.1002/1521-3773(20011217)40:24<4757::AID-ANIE4757>3.0.CO;2-S.
P. O’Brien, C. D. Pilgram, Org. Biomol. Chem., 2003, 1, 523; DOI: https://doi.org/10.1039/B210608F.
D. M. Hodgson, C. D. Bray, P. G. Humphreys, Synlett, 2006, 1, 1; DOI: https://doi.org/10.1055/s-2005-921925.
D. M. Hodgson, C. R. Maxwell, I. R. Matthews, Tetrahedron: Asymmetry, 1999, 10, 1847; DOI: https://doi.org/10.1016/S0957-4166(99)00168-8.
D. M. Hodgson, C. R. Maxwell, I. R. Matthews, Synlett, 1998, 1349; DOI: https://doi.org/10.1055/s-1998-1963.
M. Suzuki, K. Tomooka, Synlett, 2004, 4, 651; DOI: https://doi.org/10.1055/s-2004-817772.
S. Carini, V. Cerè, F. Peri, S. Pollicino, Synthesis, 2000, 1756; DOI: https://doi.org/10.1055/s-2000-8206.
M. Carpintero, C. Jaramillo, A. Fernandez-Mayoralas, Eur. J. Org. Chem., 2000, 1285; DOI: https://doi.org/10.1002/1099-0690(200004)2000:7<1285::AID-EJOC1285>3.0.CO;2-Z.
C. J. Nichols, N. S. Simpkins, Tetrahedron Lett., 2004, 45, 7469; DOI: https://doi.org/10.1016/j.tetlet.2004.08.058.
D. Moffat, C. J. Nichols, D. A. Riley, N. S. Simpkins, Org. Biomol. Chem., 2005, 3, 2953; DOI: https://doi.org/10.1039/B506444A.
A. Ishii, T. Nakaniwa, K. Umezawa, J. Nakayama, Tetrahedron, 1999, 55, 10341; DOI: https://doi.org/10.1016/S0040-4020(99)00589-X.
S. Hanessian, B. Deschenes-Simard, D. Simard, Tetrahedron, 2009, 65, 6656; DOI: https://doi.org/10.1016/j.tet.2009.06.033.
D. Moderhack, D.-O. Bode, D. Schomburg, Chem. Ber., 1993, 126, 129; DOI: https://doi.org/10.1002/cber.19931260120.
R. F. Fibiger, A. R. Banks, T. Jones, R. C. Haltiwanger, D. S. Watt, J. Heterocycl. Chem., 1978, 15, 307; DOI: https://doi.org/10.1002/jhet.5570150225.
J. Sisko, A. J. Kassick, S. B. Shetzline, Org. Lett., 2000, 2, 2877; DOI: https://doi.org/10.1021/ol006302n.
N. N. Volkova, E. V. Tarasov, M. I. Kodess, W. Dehaen, V. A. Bakulev, Mendeleev Commun., 2002, 12, 131; DOI: https://doi.org/10.1070/MC2002v012n04ABEH001625.
T. L. Gilchrist, D. Hughes, W. Stretch, E. J. T. Chrystal, J. Chem. Soc., Perkin Trans. 1, 1987, 2505; DOI: https://doi.org/10.1039/P19870002505.
B. Furman, S. Molotov, R. Thürmer, Z. Kałuża, W. Voelter, M. Chmielewski, Tetrahedron, 1997, 53, 5883; DOI: https://doi.org/10.1016/S0040-4020(97)00248-2.
T. E. Gunda, G. N. Szöke, Tetrahedron, 1998, 54, 6565; DOI: https://doi.org/10.1016/S0040-4020(98)00316-0.
J. C. Hinshaw, J. Org. Chem., 1975, 40, 47; DOI: https://doi.org/10.1021/jo00889a010.
O. De Lucchi, P. Maglioli, G. Delogu, G. Valle, Synlett, 1991, 841; DOI: https://doi.org/10.1055/s-1991-20898.
D. M. Hodgson, I. D. Cameron, M. Christlieb, R. Green, G. P. Lee, L. A. Robinson, J. Chem. Soc., Perkin Trans. 1, 2001, 2161; DOI: https://doi.org/10.1039/B105369H.
D. M. Hodgson, L. A. Robinson, Chem. Commun., 1999, 309; DOI: https://doi.org/10.1039/A809105F.
M. Schnurch, M. Spina, A. F. Khan, M. D. Mihovilovic, P. Stanetty, Chem. Soc. Rev., 2007, 36, 1046; DOI: https://doi.org/10.1039/b607701n.
A. J. Guildford, M. A. Tometzki, R. W. Turner, Synthesis, 1983, 987; DOI: https://doi.org/10.1055/s-1983-30593.
J. M. Chezal, E. Moreau, O. Chavignon, C. Lartigue, Y. Blache, J. C. Teulade, Tetrahedron, 2003, 59, 5869; DOI: https://doi.org/10.1016/S0040-4020(03)00938-4.
J. Frey, A. D. Bond, A. B. Holmes, Chem. Commun., 2002, 2424; DOI: https://doi.org/10.1039/B207403F.
M. Miyasaka, A. Rajca, J. Org. Chem., 2006, 71, 3264; DOI: https://doi.org/10.1021/jo060038f.
K. Kato, H. Hayakawa, H. Tanaka, H. Kumamoto, T. Miyasaka, Tetrahedron Lett., 1995, 36, 6507; DOI: https://doi.org/10.1016/0040-4039(95)01302-X.
K. Kato, H. Hayakawa, H. Tanaka, H. Kumamoto, S. Shindoh, S. Shuto, T. Miyasaka, J. Org. Chem., 1997, 62, 6833; DOI: https://doi.org/10.1021/jo970398q.
K.-I. Tanji, H. Kato, T. Higashino, Chem. Pharm. Bull., 1991, 39, 2793; DOI: https://doi.org/10.1248/cpb.39.2793.
B. Abarca, R. Ballesteros, M. Elmasnaouy, Tetrahedron, 1998, 54, 15287; DOI: https://doi.org/10.1016/S0040-4020(98)00955-7.
B. Abarca, R. Ballesteros, M. Elmasnaouy, ARKIVOC, 2002, Part vi, 145; DOI: https://doi.org/10.3998/ark.5550190.0003.613.
S. M. Ivanov, J. K. Voronina, A. N. Fakhrutdinov, A. M. Shestopalov, J. Fluorine Chem., 2019, 220, 16; DOI: https://doi.org/10.1016/j.jfluchem.2019.02.004.
S. M. Ivanov, M. E. Minyaev, J. Organomet. Chem., 2020, 906, 121060; DOI: https://doi.org/10.1016/j.jorganchem.2019.121060.
E. Meyle, P. Schwenkkraus, M. Zsigmondy, H.-H. Otto, Arch. Pharm., 1989, 322, 17; DOI: https://doi.org/10.1002/ardp.19893220105.
M. Zhan, S. Zhang, Z. Huang, Z. Xi, Org. Lett., 2015, 17, 1026; DOI: https://doi.org/10.1021/acs.orglett.5b00136.
O. Miyata, R. R. Schmidt, Angew. Chem., Int. Ed. Engl., 1982, 21, 637; DOI: https://doi.org/10.1002/anie.198206371.
Z. Huang, M. Zhan, S. Zhang, Q. Luo, W.-X. Zhang, Z. Xi, Org. Chem. Front., 2017, 4, 1785; DOI: https://doi.org/10.1039/C7QO00287D.
L. R. Takaoka, A. J. Buckmelter, T. E. LaCruz, S. D. Rychnovsky, J. Am. Chem. Soc., 2005, 127, 528; DOI: https://doi.org/10.1021/ja044642o.
E. Roulland, F. Cecchin, H.-P. Husson, J. Org. Chem., 2005, 70, 4474; DOI: https://doi.org/10.1021/jo050258d.
Author information
Authors and Affiliations
Corresponding author
Additional information
Published in Russian in Izvestiya Akademii Nauk. Seriya Khimicheskaya, No. 4, pp. 647–664, April, 2021.
This paper does not contain descriptions of studies on animals or humans.
The author declares no competing interests.
Rights and permissions
About this article
Cite this article
Ivanov, S.M. Rearrangements of polyaza(oxa-,thia-)heterocyclic carbanions in organic synthesis. Russ Chem Bull 70, 647–664 (2021). https://doi.org/10.1007/s11172-021-3133-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11172-021-3133-3