Abstract
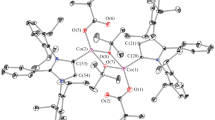
The chiral coordination compounds [Co(L)(H2O)2(MeOH)] n (1) and Co2(L)2(H2O)(MeOH)4 (2) were synthesized by the reaction of bis-2,4-[N-(S)-phenylalanyl]6-chlorotriazine (LH2) with cobalt(II) pivalate or acetate. In molecules 2, the CoII ions are linked by a bridging water molecule and two carboxy groups of the L2– anion. By passing the racemate of butan-2-ol through a chromatographic column containing complex 1 as the stationary phase, a mixture enriched with the (S)-isomer of this alcohol can be obtained.
Similar content being viewed by others
References
D. Dang, P. Wu, Ch. He, Zh. Xie, Ch. Duan, J. Am. Chem. Soc., 2010, 132, 14321.
Y. Liu, W. Xuan, Y. Cui, Adv. Mat., 2010, 22, 4112.
M. Yoon, R. Srirambalaji, K. Kim, Chem. Rev., 2012, 112, 1196.
K. Gedrich, M. Heitbaum, A. Notzon, I. Senkovska, R. Frçhlich, J. Getzschmann, U. Mueller, F. Glorius, S. Kaskel, Chem. Eur. J., 2011, 17, 2099.
R. Vaidhyanathan, D. Bradshaw, J.-N. Rebilly, J. P. Barrio, J. A. Gould, N. G. Berry, M. J. Rosseinsky, Angew. Chem., Int. Ed., 2006, 45, 6495.
M. C. Das, Q. Guo, Y. He, J. Kim, C.-G. Zhao, K. Hong, S. Xiang, Z. Zhang, K. M. Thomas, R. Krishna, B. Chen, J. Am. Chem. Soc., 2012, 134, 8703.
M. Padmanaban, P. Müller, C. Lieder, K. Gedrich, R. Grünker, V. Bon, I. Senkovska, S. Baumgärtner, S. Opelt, S. Paasch, E. Brunner, F. Glorius, E. Klemm, S. Kaskel, Chem. Commun., 2011, 47, 12089.
S.-M. Xie, Z.-J. Zhang, Z.-Y. Wang, L.-M. Yuan, J. Am. Chem. Soc., 2011, 133, 11892.
A. L. Nuzhdin, D. N. Dybtsev, K. P. Bryliakov, E. P. Talsi, V. P. Fedin, J. Am. Chem. Soc., 2007, 129, 12958.
C. Train, R. Gheorghe, V. Krstic, L.-M. Chamoreau, N. S. Ovanesyan, G. L. J. A. Rikken, M. Gruselle, M. Verdaguer, Nature Materials, 2008, 7, 729.
E. Coronado, M. C. Giménez-Lòpez, T. Korzeniak, G. Levchenko, F. M. Romero, A. Segura, V. García-Baonza, J. C. Cezar, F. M. F. de Groot, A. Milner, M. PazPasternak, J. Am. Chem. Soc., 2008, 130, 15519.
T. Wu, X.-Z. You, P. Bou, Coord. Chem. Rev., 2015, 284, 1.
H.-T. Ye, C.-Y. Ren, G.-F. Hou, Y.-H. Yu, X. Xu, J.-S. Gao, P.-F. Yan, S.-W. Ng, Cryst. Growth Des., 2014, 14, 3309.
S. Allenmark, Chromatographic Enantioseparation: Methods and Applications, Halsted Press/Wiley, Chichester, 1988, 224.
S.-M. Xie, X.-H. Zhang, Z.-J. Zhang, M. Zhang, J. Jia, L.-M. Yuan, Anal Bioanal Chem., 2013, 405, 3407.
S.-M. Xie, X.-H. Zhang, Z.-J. Zhang, L.-M. Yuan, Anal. Lett., 2013, 46, 753.
S. Xie, B. Wang, X. Zhang, J. Zhang, M. Zhang, L. Yuan, Chirality, 2014, 26, 27.
D. N. Dybtsev, M. P. Yutkin, E. V. Peresypkina, A. V. Virovets, C. Serre, G. Férey, V. P. Fedin, Inorg. Chem., 2007, 46, 6843.
D. N. Dybtsev, M. P. Yutkin, D. G. Samsonenko, V. P. Fedin, A. L. Nuzhdin, A. A. Bezrukov, K. P. Bryliakov, E. P. Talsi, R. V. Belosludov, H. Mizuseki, Y. Kawazoe, O. S. Subbotin, V. R. Belosludov, Chem. Eur. J., 2010, 16, 10348.
D. N. Dybtsev, A. L. Nuzhdin, H. Chun, K. P. Bryliakov, E. P. Talsi, V. P. Fedin, K. Kim, Angew. Chem., Int. Ed., 2006, 45, 916.
J. A. Rood, B. C. Noll, K. W. Henderson, J. Solid, State Chem., 2010, 183, 270.
M. P. Yutkin, M. S. Zavakhina, D. G. Samsonenko, D. N. Dybtsev, V. P. Fedin, Russ. Chem. Bull. (Int. Ed.), 2010, 59, 733 [Izv. Akad. Nauk, Ser. Khim., 2010, 719].
Z. Chen, X. Liu, C. Zhang, Z. Zhang, F. Liang, Dalton Trans., 2011, 40, 1911.
N. Hoshino, M. Nakano, H. Nojiri, W. Wernsdorfer, H. Oshio, J. Am. Chem. Soc., 2009, 131, 15100.
R. S. Forgan, R. A. Smaldone, J. J. Gassensmith, H. Furukawa, D. B. Cordes, Q. Li, C. E. Wilmer, Y. Y. Botros, R. Q. Snurr, A. M. Z. Slawin, J. F. Stoddart, J. Am. Chem. Soc., 2012, 134, 406.
G. Li, W. Yu, J. Ni, T. Liu, Y. Liu, E. Sheng, Y. Cui, Angew. Chem., Int. Ed., 2008, 47, 1245.
T. Liu, Y. Liu, W. Xuan, Y. Cui, Angew. Chem., Int. Ed., 2010, 49, 4121.
T. Ezuhara, K. Endo, Y. Aoyama, J. Am. Chem. Soc., 1999, 121, 3279.
G. Tian, G. Zhu, X. Yang, Q. Fang, M. Xue, J. Sun, Y. Wei, S. Qiu, Chem. Commun., 2005, 1396.
S. Wu, Y. Wu, Q. Kang, H. Zhang, L. Long, Z. Zheng, R. Huang, L. Zheng, Angew. Chem., Int. Ed., 2007, 46, 8475.
S. H. Jhung, J.-H. Lee, P. M. Forster, G. Férey, A. K. Cheetham, J.-S. Chang, Chem. Eur. J., 2006, 12, 7899.
V. I. Mur, Russ. Chem. Rev., 1964, 33, 92 [Usp. Khim., 1964, 33, 182].
C. J. Cavallito, D. C. Schlieper, T. B. O´Dell, J. Org. Chem., 1954, 19, 826.
E. A. Mikhalyova, S. V. Kolotilov, O. Cador, F. Pointillart, S. Golhen, L. Ouahab, V. V. Pavlishchuk, Inorg. Chim. Acta, 2010, 3453.
X. Zhao, H. He, T. Hu, F. Dai, D. Sun, Inorg. Chem., 2009, 48, 8057.
D. Sun, S. Ma, Y. Ke, D. J. Collins, H.-C. Zhou, J. Am. Chem. Soc., 2006, 128, 3896.
K. Suh, M. P. Yutkin, D. N. Dybtsev, V. P. Fedin, K. Kim, Chem. Commun., 2012, 48, 513.
K. Huang, X. Dong, R. Ren, W. Jin, AIChE J., 2013, 59, 4364.
P. Li, Y. He, J. Guang, L. Weng, J. C.-G. Zhao, S. Xiang, B. Chen, J. Am. Chem. Soc., 2014, 136, 547.
R.-G. Xiong, X.-Z. You, B. F. Abrahams, Z. Xue, C.-M. Che, Angew. Chem., Int. Ed., 2001, 40, 4422.
Yu. A. Satskaya, N. P. Komarova, K. S. Gavrilenko, O. V. Manoilenko, Zh. V. Chernenko, M. A. Kiskin, S. V. Kolotilov, I. L. Eremenko, V. M. Novotortsev, Theor. Exper. Chem., 2015, 51, 45 [Teor. Eksp. Khim., 2015, 51, 41].
I. G. Fomina, G. G. Aleksandrov, Zh. V. Dobrokhotova, O. Yu. Proshenkina, M. A. Kiskin, Yu. A. Velikodnyi, V. N. Ikorskii, V. M. Novotortsev, I. L. Eremenko, Russ. Chem. Bull. (Int. Ed.), 2006, 55, 1909 [Izv. Akad. Nauk, Ser. Khim., 2006, 1841].
SMART (control) and SAINT (integration) Software, Version 5.0, Bruker AXS Inc., Madison,WI, 1997.
G. M. Sheldrick, SHELX-97, Program for the Refinement of Crystal Structure, University of Göttingen, Göttingen (Germany), 1997.
Author information
Authors and Affiliations
Corresponding author
Additional information
Dedicated to Academician of the Russian Academy of Sciences V. I. Minkin on the occasion of his 80th birthday.
Published in Russian in Izvestiya Akademii Nauk. Seriya Khimicheskaya, No. 3, pp. 0630—0635, March, 2015.
Rights and permissions
About this article
Cite this article
Satskaya, Y.A., Komarova, N.P., Gavrilenko, K.S. et al. Cobalt(II) complexes with bis-2,4-[N-(S)-phenylalanyl]-6-chlorotriazine: synthesis, structure, and application for separation of enantiomers of butan-2-ol. Russ Chem Bull 64, 630–635 (2015). https://doi.org/10.1007/s11172-015-0910-x
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11172-015-0910-x