Abstract
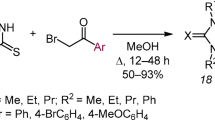
A reaction of 3-benzylthiotriazole-4-amines with aromatic aldehydes leads to the formation of 6,7-dihydro-5H-[1,2,4]triazolo[3,4-b][1,3,4]thiadiazines. A dihydrothiadiazine ring opening along the N-N bond occurs by the action of strong bases.
Similar content being viewed by others
References
L. P. Olekhnovich, E. P. Ivakhnenko, S. N. Lyubchenko, V. I. Simakov, G. S. Borodkin, A. V. Lesin, I. N. Shcherbakov, S. V. Kurbatov, Ros. Khim. Zh., 2004, 48, 103 [Mendeleev Chem. J., 2004, 48, No. 1 (Engl. Transl.)].
G. M. Sheldrick, SHELXTL v 5.10, Structure Determination Software Suite, Bruker AXS, Madison, Wisconsin, USA, 1998.
J. R. Reid, N. D. Heindel, J. Heterocycl. Chem., 1976, 13, 925.
Author information
Authors and Affiliations
Corresponding author
Additional information
Published in Russian in Izvestiya Akademii Nauk. Seriya Khimicheskaya, No. 6, pp. 1249–1252, June, 2008.
Rights and permissions
About this article
Cite this article
Kolodina, A.A., Gaponenko, N.I. & Lesin, A.V. Synthesis and opening of the thiadiazine ring in 6,7-dihydro-5H-[1,2,4]triazolo[3,4-b][1,3,4]thiadiazines. Russ Chem Bull 57, 1273–1276 (2008). https://doi.org/10.1007/s11172-008-0164-y
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11172-008-0164-y