Abstract
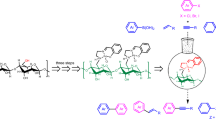
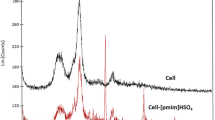
Surface-modified cellulose-based heterogeneously active catalyst was prepared by covalent anchoring of chlorosulphonic acid on amino-functionalized cellulose (SA@Cell-AEPC). The structure of synthesized catalyst was confirmed by analytical methods such as FT-IR, FE-SEM, EDX, TGA, XRD, TEM and CP/MAS 13C-NMR spectroscopy. The catalytic effect was evaluated for the formation of oxygen and nitrogen heterocycles. Reusability, shorter reaction time, high conversion, wide substrate scope, easy work-up procedure are noteworthy measures of this synthetic route.
Graphical abstract














Similar content being viewed by others
Data availability
All relevant data are within the manuscript and included in the electronic supplementary information.
References
P. Marion, B. Bernela, A. Piccirilli, B. Estrine, N. Patouillard, J. Guilbot, F. Jerome, Green Chem. 19, 4973 (2017)
P. Gupta, A. Mahajan, RSC Adv. 5, 26686 (2015)
M.B. Gawande, V.D.B. Bonifacio, R. Luque, P.S. Branco, R.S. Varma, Chem. Soc. Rev. 42, 5522 (2013)
R.A. Sheldon, I. Arends, U. Hanefeld, Green Chemistry and Catalysis (Wiely-VCH, Weinheim, 2007)
X.-L. Shi, H. Yang, M. Tao, W. Zhang, RSC Adv. 3, 3939 (2013)
H. Koga, T. Kitaoka, A. Isogai, Molecules 20, 1495 (2015)
X.-L. Shi, Q. Hu, Y. Chen, F. Wang, P. Duan, J. Ind. Eng. Chem. 65, 264 (2018)
C.M. Gowen, S.S. Fong, Chem. Biodiversity 7, 1086 (2010)
B.B.F. Mirjalili, A. Bamoniri, S.A. Fazeli-Attar, Res. Chem. Intermed. 48, 839 (2022)
B.B.F. Mirjalili, F. Aref, Res. Chem. Intermed. 44, 4519 (2018)
F. Osanlou, F. Nemati, S. Sabaqian, Res. Chem. Intermed. 43, 2159 (2017)
B. Sadeghi, Res. Chem. Intermed. 45, 4897 (2019)
S. Karhale, C. Bhenki, G. Rashinkar, V. Helavi, New J. Chem. 41, 5133 (2017)
S. Kamino, M. Uchiyama, Org. Biomol. Chem. 21, 2458 (2023)
C.F. Silva, D.C. Pinto, P.A. Fernandes, A.M.S. Silva, Pharmaceuticals 15, 148 (2022)
U.N. Guria, S.K. Manna, K. Maiti, S.K. Samanta, A. Ghosh, P. Datta, D. Mandal, A.K. Mahapatra, New J. Chem. 45, 15869 (2021)
M. Maia, D.I.S.P. Resende, F. Duraes, M.M. Pinto, E. Sousa, Eur. J. Med. Chem. 210, 113085 (2021)
K. Colas, S. Doloczki, M.P. Urrutia, C. Dyrager, Eur. J. Org. Chem. 15, 2133 (2021)
H. Ding, L. Peng, G. Yuan, L. Zhou, Dyes Pigm. 176, 108232 (2020)
P. Kushwaha, A.K. Tripathi, S. Gupta, P. Kothari, A. Upadhyay, N. Ahmad, T. Sharma, M.I. Siddiqi, R. Trivedi, K.V. Sashidhara, Eur. J. Med. Chem. 156, 103 (2018)
M. Rahimifard, G.M. Ziarani, A. Badiei, S. Asadi, A.A. Soorki, Res. Chem. Intermed. 42, 3847 (2016)
S. Karhale, V. Helavi, SN Appl. Sci. 2, 1227 (2020)
S. Karhale, Res. Chem. Intermed. 46, 3085 (2020)
S. Karhale, D. Survase, R. Bhat, P. Ubale, V. Helavi, Res. Chem. Intermed. 43, 3915 (2017)
S. Karhale, M. Patil, G. Rashinkar, V. Helavi, Res. Chem. Intermed. 43, 7073 (2017)
L. Jiao, Y. Hu, H. Ju, C. Wang, M.-R. Gao, Q. Yang, J. Zhu, S.-H. Yu, H.L. Jiang, J Mater Chem A. 5, 23170 (2017)
F. Shirini, M. Mamaghani, M. Seddighi, Catal. Commun. 36, 31 (2013)
R. Kurane, S. Khanapure, D. Kale, R. Salunkhe, G. Rashinkar, RSC Adv. 6, 44135 (2016)
B. Banerjee, M. Kaur, V. Sharma, V.K. Gupta, J. Kaur, A. Sharma, A. Priya, A. Singh, Res. Chem. Intermed. 49, 4639 (2023)
K. Hoseinzade, S.A. Mousavi-Mashhadi, A. Shiri, Mol. Divers. 26, 2745 (2022)
D. Thirumalai, S. Gajalakshmi, Res. Chem. Intermed. 46, 2657 (2020)
A. Fallah, M. Tajbakhsh, H. Vahedi, A. Bekhradnia, Res. Chem. Intermed. 43, 29 (2017)
A. Ilangovan, S. Muralidharan, P. Sakthivel, S. Malayappasamy, S. Karuppusamy, M.P. Kaushik, Tetrahedron Lett. 54, 491 (2013)
B. Das, J. Kashanna, R.A. Kumar, P. Jangili, Synth. Commun. 42, 2876 (2012)
M. Marinescu, Chemistry and Applications of Benzimidazole and Its Derivatives, (United Kingdom, IntechOpen, 2019).
S.J. Lin, Y.C. Cheng, C.H. Chen, Y.Y. Zhang, J.H. Lee, M.K. Leung, B.Y. Lin, T.L. Chiu, J. Mater. Chem. C 11, 161 (2023)
S.M. Abu-Bakr, F.A. Bassyouni, M.A. Rehim, Res. Chem. Intermed. 38, 2523 (2012)
A.M.S. Hebishy, M.S. Abdelfattah, A. Elmorsy, A.H.M. Elwahy, J. Heterocycl. Chem. 57, 2256 (2020)
M. Marinescu, D.G. Tudorache, G.I. Marton, C.M. Zalaru, M. Popa, M.C. Chifiriuc, C.E. Stavarache, C. Constantinescu, J. Mol. Struct. 1130, 463 (2017)
R.P. Tayade, N. Sekar, Dyes Pigm. 128, 111 (2016)
A. Hameed, A. Hameed, T. Farooq, R. Noreen, S. Javed, S. Batool, A. Ahmad, T. Gulzar, M. Ahmad, BMC Chem. 13, 29 (2019)
S.R. Brishty, M.J. Hossain, M.U. Khandaker, M.R.I. Faruque, H. Osman, S.M.A. Rahman, Front. Pharmacol. 12, 762807 (2021)
S.K. Bagaria, N. Jangir, D.K. Jangid, Sustain. Chem. Pharm. 31, 100932 (2023)
A.K. Sharma, P. Sharma, P. Mehara, P. Das, Chem. Eng. J. 471, 144666 (2023)
M.A. Tzani, C. Gabriel, I.N. Lykakis, Nanomaterials 10, 2405 (2020)
S. Karhale, K. Patil, C. Bhenki, G. Rashinkar, V. Helavi, Res. Chem. Intermed. 42, 7257 (2016)
K.R. Balinge, S.K. Datir, V.B. Khajone, K.J. Bhansali, A.G. Khiratkar, P.R. Bhagat, Res. Chem. Intermed. 45, 155 (2019)
M. Goswami, M.M. Dutta, P. Phukan, Res. Chem. Intermed. 44, 1597 (2018)
M. Karthik, P. Suresh, New J. Chem. 42, 17931 (2018)
M.T. Maghsoodlou, N. Hazeri, M. Lashkari, F.N. Shahrokhabadi, B. Naghshbandi, M.S. Kazemi-doost, M. Rashidi, F. Mir, M. Kangani, S. Salahi, Res. Chem. Intermed. 41, 6985 (2015)
M.A. Chari, P. Sadanandam, D. Shobha, K. Mukkanti, J. Heterocycl. Chem. 47, 153 (2010)
Acknowledgements
SSK is gratefully acknowledged to the Principal, Karmaveer Bhaurao Patil Mahavidyalaya (Autonomous), Pandharpur, Solapur for providing the financial assistance under RUSA Scheme [RUSA/MRP/F.No.1/004/20-21, Dated: 5th September 2020]. Author is also thankful to the Head, Sophisticated Analytical Instrument Facility, IIT Bombay and Scientific Instrumentation Center, PAH, Solapur University, Solapur for providing spectral facilities.
Funding
This work was financially supported by the Principal, Karmaveer Bhaurao Patil Mahavidyalaya (Autonomous), Pandharpur, Solapur, Maharashtra, India under RUSA Scheme [RUSA/MRP/F.No.1/004/20-21, Dated: 5th September 2020].
Author information
Authors and Affiliations
Contributions
Shrikrishna Karhale: Data curation, Formal analysis. Project administration, Writing main manuscript and Supervision. Ankush Kadam: Conceptualization, Methodology, prepared figures and Investigation. Both authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethical approval
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Karhale, S., Kadam, A. Cellulose-based surface-modified heterogeneously feasible novel solid acid catalyst to access bioactive heterocycles. Res Chem Intermed 50, 31–48 (2024). https://doi.org/10.1007/s11164-023-05171-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11164-023-05171-8