Abstract
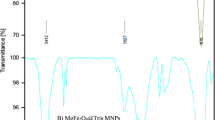
In this work, the surface of Fe3O4 nanoparticles was functionalized with the glycine, furfural, and cobalt (II) nitrate hexahydrate to synthesize a novel support nanocatalyst (Fe3O4@gly@Furfural@Co(NO3)2). The characteristics of the nanocatalyst were confirmed by various techniques, such as energy-dispersive X-ray spectroscopy (EDX), Fourier transform infrared spectrometer, EDX mapping, scanning electron microscopy, X-ray diffraction, thermal gravimetric/derivative thermal gravimetric, vibrating sample magnetometer, and TEM. Finally, the performance of the nanocatalyst was investigated for the one-pot multicomponent synthesis of benzo[b]pyran and pyrano[2,3-d] pyrimidine derivatives. The good–excellent yield of the products was achieved in a green solvent under mild conditions at short reaction time. In addition, the green heterogeneous catalyst has features, such as environmental friendliness, easy separation from the reaction mixture by an external magnet, high catalytic activity, and reusability up to 6 stages without significant reduction of its catalytic activity.













Similar content being viewed by others
Availability of data and materials
Not applicable.
References
G.H. Mohammadi Ziarani, Z. Kheilkordi, F. Mohajer, A. Badiei, R. Luque, RSC Adv. 11, 17456 (2021)
H. Faroughi Niya, N. Hazeri, M. Fatahpour, M.T. Maghsoodlou, Res. Chem. Intermed. 46, 3651 (2020)
D. Wang, J. He, N. Rosenzweig, Z. Rosenzweig, Nano Lett. 4, 409 (2004)
A.K. Gupta, M. Gupta, Biomaterials 26, 3995 (2005)
J.M. Perez, T. Ooughin, F.J. Simone, R. Weissleder, L. Josephson, J. Am. Chem. Soc. 124, 2856 (2002)
A. Ito, Y. Kuga, H. Honda, H. Kikkawa, A. Horiuchi, Y. Watanabe, T. Kobayashi, Cancer Lett. 212, 167 (2004)
X.J. Zhu, Sci. Iran. 18, 1627 (2011)
A.K. Gupta, A.S. Curtis, J. Mater. Sci. 15, 493 (2004)
H. Faroughi Niya, N. Hazeri, M. Fatahpour, M.T. Maghsoodlou, Res. Chem. Intermed. 46, 1685 (2019)
M. Mohammadi, M. Khodamoradi, B. Tahmasbi, K. Bahrami, A. Ghorbani-Choghamarani, J. Ind. Eng. Chem. 97, 1 (2021)
M. Kazemi, M. Mohammadi, Appl. Organomet. Chem. 34, e5400 (2019)
F. Ghobakhloo, D. Azarifar, M. Mohammadi, H. Keypour, H. Zeynali, Inorg. Chem. 61, 4786 (2022)
M. Mohammadi, A. Ghorbani-Choghamarandi, Res. Chem. Intermed. 48, 2641 (2022)
A. Ghorbani-Choghamarandi, M. Mohammadi, L. Shiri, Z. Taherinia, Res. Chem. Intermed. 45, 5705 (2019)
S. Motahari, A. Khazaei, Polycycl. Aromat. Compd. 43, 945 (2021)
M. Rezaie Kahkhaie, N. Hazeri, M.T. Maghsoodlou, A. Yazdani-Elah-Abadi, Res. Chem. Intermed. 47, 5007 (2021)
Q. Zhang, W. Cui, H. Guo, B. Wang, H. Wang, J. Zhang, W. Li, J. Exp. NanoSci. 17, 326 (2022)
T. Tamoradi, S.M. Mousavi, M. Mohammadi, Chem. Select. 5, 5077 (2020)
M. Mohammadi, A. Ghorbani-Choghamarani, N. Hussain-Khil, Phys. Chem. Solids. 177, 111300 (2023)
A. Nikseresht, R. Bagherinia, M. Mohammadi, R. Mehrvar, RSC Adv. 13, 674 (2023)
A. Ghorbani-Chghamarandi, M. Mohammadi, T. Tamoradi, M. Ghadermazi, Polyhedron 158, 25 (2019)
R. Achagar, A. Elmakssoudi, A. Thoume, M. Dakir, A. Elamrani, Y. Zouheir, M. Zahouily, Z. Ait-Touchente, J. Jamaleddine, M.M. Chehimi, Appl. Sci. 12, 5487 (2022)
B. Shaabani, H. Maleki, J. Rakhtshah, J. Organomet. Chem. 897, 139 (2019)
M. Maleki, M. Azizi, Z. Emdadi, Green Chem. Lett. Rev. 11, 573 (2018)
B. Adrom, N. Hazeri, M. Lashkari, M.T. Maghsoodlou, J. Chem. Res. 40, 458 (2016)
D. Dharmendra, P. Chundawat, Y. Vyas, P. Chaubisa, M. Kumawat, C. Ameta, Sustain Chem Pharm. 28, (2022)
N. Zheng, Q. Hao, K. Lin, J. Pan, Y. Li, W. Zhou, Bioorgan. Med. Chem. Lett. 29, 225 (2019)
M. Davidson, P. Ma, E.A. Stein, A.M. Guotto Jr., A. Raza, R. Chitra, H. Hutchinson, Am. J. Cardiol. 89, 268 (2002)
J.S. Debenham, C. Madsen-Duggan, M.J. Clements, T.F. Walsh, J.T. Kuethe, M. Reibarkh, S.P. Salowe, L.M. Sonatore, R. Hajdu, J.A. Milligan, J. Med. Chem. 59, 11039 (2016)
L. Sorbera, J. Castaner, Drags Future 30, 765 (2005)
J.A. Makawana, M.P. Patel, R.G. Patel, Arch. Pharm. 345, 314 (2012)
A. Venkatesham, R.S. Rao, K. Nagaiah, J.S. Yadav, G. Roopa Jones, S.J. Basha, B. Sridhar, A. Addlagatta, MedChemComm 3, 652 (2012)
A.R. Saundane, K. Vijaykumar, A.V. Vaijinath, Bioorg. Med. Chem. Lett 23, 1978 (2013)
S.A. Patil, J. Wang, X.S. Li, J.J. Chen, T.S. Jones, A. HosniAhmed, R. Patil, W.L. Seibel, W. Li, D.D. Miller, Bioorg. Med. Chem. Lett. 22, 4458 (2012)
E. Alimohammadi, K. Khosravi, A.M. Zolfigol, J. Iran. Chem. Soc. 19, 4721 (2022)
T.E. Ali, M.A. Assiri, A.A. Shati, M.Y. Alfaifi, S.E.I. Elbehairi, Russ. J. Org. Chem. 58, 106 (2022)
S. Tabassum, K.R. Sunaja Devi, S. Govindaraju, Mater. Today Proc. 45, 3716 (2021)
A. Saha, S. Payra, A. Asatkar, A.R. Patel, S. Banerjee, Curr. Organocatal. 6, 177 (2019)
K.C. Barick, P.A. Hassan, J. Colloid Interface Sci. 369, 96 (2012)
K. Kumar Gangu, S. Maddila, S. Babu Mukkamala, S.B. Jonnalagadda, J. Mol. Struct. 1143, 153 (2017)
S.M. Mousavifar, H. Kefayati, S. Shariati, Appl. Organomet. Chem. 32, e4242 (2018)
K.C. Barick, S. Singh, D. Bahadur, M.A. Lawande, D.P. Patkar, P.A. Hassan, J. Colloid Interface Sci. 418, 120 (2014)
Kh. Niknam, N. Borazjani, R. Rashidian, A. Jamali, Chinese J. Catal. 13, 2245 (2013)
S. Rostamnia, A. Nuri, H. Xin, A. Pourjavadi, S.H. Hosseini, Tetrahedron Lett. 54, 3344 (2013)
F. Mohammadpour, Org. Prep. Proceed. Int. 54, 306 (2022)
N. Azizi, S. Dezfooli, M. Khajeh, M.M. Hashemi, J. Mol. Liq. 186, 76 (2013)
A. Rostami, B. Atashkar, H. Gholami, Catal. Commun. 37, 67 (2013)
D.D. Pham, G. Vo-Thanh, T.N. Le, Synth. Commun 47, 1684 (2017)
B. Maleki, N. Nasiri, R. Tayebee, A. Khojastehnezhad, H.A. Akhlaghi, Rsc Adv. 6, 79128 (2016)
R. Heydari, A. Mansouri, F. Shahrekipour, R. Shahraki, Lett. Org. Chem. 15, 302 (2018)
S. Khaksar, A. Rouhollahpour, S.M. Talesh, J. Fluor. Chem. 141, 11 (2012)
N. Taheri, M. Fallah-Mehrjardi, S. Sayyahi, Bull. Chem. Soc. Ethiop. 32, 531 (2018)
K. Madasamy, S.H. Kumaraguru, V. Sankar, S. Mannathan, M. Kathiresan, New J. Chem. 43, 3795 (2019)
M. Esmaeilpour, J. Javidi, F. Dehghani, F. Nowroozi Dodeji, RSC Adv. 34, 26625 (2015)
A. Pourkazemi, N. Assadi, M. Farahi, A. Zarnegaryan, B. Karami, Acta Chim. Solv. 69, 30 (2022)
H. Alinezhad, M. Tarahomi, B. Maleki, A. Amiri, Appl. Organomet. Chem. 33, e4661 (2019)
M. Bakherad, Gh. Bagherian, A. Rezaeifard, F. Mosavebi, B. Shokoohi, A. Keivanloo, J. Iran. Chem. Soc. 18, 839 (2021)
M. Abaszadeh, S.J. Roudbaraki, M. Ghashang, Org. Prep. Proceed. Int. 51, 255 (2019)
M.A. Zolfigol, R. Ayazi-Nasrabadi, S. Baghery, Appl. Organomet. Chem. 30, 273 (2016)
S.N. Maddila, S. Maddila, W.E. van Zyl, S.B. Jonnalagadda, Rsc Adv. 5, 37360 (2015)
A.A. Mohammadi, M.R. Asghariganjeh, A. Hadadzahmatkesh, Arab. J. Chem. 10, S2213 (2017)
S. Chauhan, A. Mishra, P. Verma, V. Srivastava, Org. Prep. Proceed. Int. 53, 441 (2021)
A.R. Nesaragi, T. Gasti, T.V. Metra, R.R. Kamble, R.B. Chougale, R.S. Keri, Super Mol. Chem. 7, e202200604 (2022)
D. Azarifar, R. Nejat-Yami, F. Sameri, Z. Akrami, Lett. Org. Chem. 9, 435 (2012)
G. Brahmachari, B. Banerjee, A.C.S. Sustain, Chem. Eng. 2, 2802 (2014)
N. Sheikhan-Shamsabadi, M. Ghashang, Main Group Met. Chem. 40, 19 (2017)
M.M. Heravi, A. Ghods, K. Bakhtiari, F. Derikvand, Synth. Commun. 40, 1927 (2010)
Acknowledgements
The authors gratefully appreciate the financial support from the Research Council of University of Sistan and Baluchestan.
Funding
Funding was provided by University of Sistan and Baluchestan.
Author information
Authors and Affiliations
Contributions
RK (the PhD. student) performed the practical laboratory work and wrote the manuscript. NH (Supervisor, PhD) designed the research project. HFN (Advisor, PhD) helped Miss RK to do her PhD. research project in optimized conditions. MF (Advisor, PhD) helped Miss RK with the formal analysis, writing, and editing of her manuscript.
Corresponding author
Ethics declarations
Ethical approval
The manuscript treats an original work; it has not been previously published and is not currently submitted for review to any other journal. We want to clarify that it will not be submitted elsewhere before a decision is made by this journal.
Conflict of interest
The authors declare no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Keshani, R., Hazeri, N., Faroughi Niya, H. et al. Synthesis of benzo[b]pyran and pyrano[2,3-d]pyrimidine derivatives using a new superparamagnetic nanocatalyst Fe3O4@gly@Furfural@Co(NO3)2. Res Chem Intermed 49, 3461–3479 (2023). https://doi.org/10.1007/s11164-023-05044-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11164-023-05044-0