Abstract
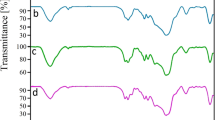
The chemoselective oxidative coupling of thiols to disulfides and oxidation of sulfides to corresponding sulfoxides are significant procedures in organic chemistry. Therefore, we have investigated 5-nitro-salicylaldehyde Schiff-base complex of vanadium oxide on MCM-41 (V(O)-5NSA-MCM-41) as a stable, efficient, reusable, and chemoselective nanocatalyst for the synthesis of disulfide and sulfoxide derivatives using hydrogen peroxide (H2O2) as biocompatibility, inexpensive, and available oxidant. V(O)-5NSA-MCM-41 was characterized using various techniques such as SEM, EDS, WDX, XRD, TEM, TGA, BET, and FTIR. This catalyst can be recovered and reused several times without significant loss in catalytic activity. In addition, the recovered catalyst was characterized with several techniques, which are showing an excellent agreement with fresh catalyst before reusing. A comparison of reused and un-reused catalysts confirmed the heterogeneous nature of this catalyst.

















Similar content being viewed by others
Availability of data and materials
All data are available in the main manuscript file.
References
F. Poovan, V. Chandrashekhar, K. Natte, R.V. Jagadeesh, Catal Sci. Technol. 12, 6623 (2022)
M. Miceli, P. Frontera, A. Macario, A. Malara, Catalysts 11, 591 (2021)
D.J. Cole-Hamilton, Science 299, 1702 (2003)
V.S. Shende, V.B. Saptal, B.M. Bhanage, Chem. Rec. 19, 1 (2019)
F. Habeche, M. Hachemaoui, A. Mokhtar, K. Chikh, F. Benali, A. Mekki, F. Zaoui, Z. Cherif, B. Boukoussa, J. Inorg. Organomet. Polym. 30, 4245 (2020)
C.W. Lim, I.S. Lee, Nano Today 5, 412 (2010)
D. Astruc, Chem. Rev. 120, 461 (2020)
C. Testa, A. Zammataro, A. Pappalardo, G.T. Sfrazzetto, RSC Adv. 9, 27659 (2019)
S.B. Somwanshi, S.B. Somvanshi, P.B. Kharat, J. Phys. Conf. Ser. 1644, 012046 (2020). https://doi.org/10.1088/1742-6596/1644/1/012046
M. Nikoorazm, B. Tahmasbi, S. Gholami, P. Moradi, Appl. Organomet. Chem. 34, e5919 (2020)
P. Moradi, M. Hajjami, RSC Adv. 11, 25867–25879 (2021)
K. Biradha, A. Goswami, R. Moi, Chem. Commun. 56, 10824 (2020)
A. L. Corcho-Valdés, C. Iriarte-Mesa, J. Calzadilla-Maya, Y. Matos-Peralta, L. F. Desdín-García, M. Antuch (2022) Carbon Composite Catalysts 223–266.
M. Melchionna, S. Marchesan, M. Prato, P. Fornasiero, Catal Sci. Technol. 5, 3859 (2015)
S. Khazalpour, M. Yarie, E. Kianpour, A. Amani, S. Asadabadi, J.Y. Seyf, M. Rezaeivala, S. Azizian, M.A. Zolfigol, J. Iran. Chem. Soc. 17, 1775 (2020)
P. Moradi, RSC Adv. 12, 33459 (2022)
M. Nikoorazm, P. Moradi, N. Noori, G. Azadi, J. Iran. Chem. Soc. 18, 467 (2021)
B. Atashkar, A. Rostami, H. Gholami, B. Tahmasbi, Res. Chem. Intermed. 41, 3675 (2015)
B. Tahmasbi, A. Ghorbani-Choghamarani, P. Moradi, New J. Chem. 44, 3717 (2020)
A. Ghorbani-Choghamarani, P. Moradi, B. Tahmasbi, Polyhedron 163, 98 (2019)
P. Moradi, M. Hajjami, RSC Adv. 12, 13523 (2022)
P. Moradi, M. Hajjami, New J. Chem. 45, 2981 (2021)
C.T. Kresge, M.E. Leonowicz, W.J. Roth, J.C. Vartuli, J.S. Beck, Nature 359, 710 (1992)
G. Martínez-Edo, A. Balmori, I. Pontón, A.M. del Rio, D. Sánchez-García, Catalysts 8(12), 617 (2018)
J.A.S. Costa, R.A. de Jesus, D.O. Santos, J.F. Mano, L.P.C. Romão, C.M. Paranhos, Microporous Mesoporous Mater. 291, 109698 (2020)
M. Nikoorazm, P. Moradi, N. Noori, J. Porous Mater. 27, 1159 (2020)
M. Nikoorazm, A. Ghorbani-Choghamaranai, M. Khanmoradi, P. Moradi, J. Porous Mater. 25, 1831 (2018)
G. Martínez-Edo, A. Balmori, I. Pontón, A. Martí del Rio, D. Sánchez-García, Catalysts 8(12), 617 (2018)
P. Polikarpova, A. Akopyan, A. Shigapova, A. Glotov, A. Anisimov, E. Karakhanov, Energ. Fuel. 32(10), 10898 (2018)
T. Kikhavani, P. Moradi, M. Mashari-Karir, J. Naji, Appl. Organomet. Chem. 36, e6895 (2022)
A. Mavrogiorgou, M. Baikousi, V. Costas, E. Mouzourakis, Y. Deligiannakis, M.A. Karakassides, M. Louloudi, J. Mol. Catal. A Chem. 413, 40 (2016)
E. Ali, M.R. Naimi-Jamal, M.G. Dekamin, Sci. Iran. 20, 592 (2013)
A.M. Abu-Dief, I.M.A. Mohamed, Beni-Suef Univ. J. Basic Appl. Sci. 4(2), 119 (2015)
M. Sunjuk, L. Al-Najjar, M. Shtaiwi, B. El-Eswed, M. Al-Noaimi, L. Al-Essa, K. Sweidan, Inorganics 10(4), 43 (2022)
W. Al Zoubi, Y. Gun Ko, Appl. Organomet. Chem. 31(3), e3574 (2017)
C.M. da Silva, D.L. da Silva, L.V. Modolo, R.B. Alves, M.A. de Resende, C.V.B. Martins, Â. Fátima, J. Adv. Res. 2(1), 1 (2011)
H. Schiff, Ann. Der Chem. Und Pharm. 131, 118 (1864)
L. Shiri, B. Tahmasbi, Phosphorus Sulfur Silicon Relat. Elem. 192, 53 (2017)
C. Liu, Synth. Commun. 51, 2237 (2021)
A. Ghorbani-Choghamarani, G. Azadi, B. Tahmasbi, M. Hadizadeh-Hafshejani, Z. Abdi, Phosphorus Sulfur Silicon Relat. Elem. 189, 433 (2014)
M.M.D. Pramanik, N. Rastogi, Chem. Commun. 52, 8557 (2016)
K.G.M. Koua, V.M. Dong, Org. Biomol. Chem. 13, 5844 (2015)
A. Ghorbani-Choghamarani, P. Moradi, B. Tahmasbi, J. Iran. Chem. Soc. 16, 511 (2019)
Q. Jing, X. Hu, Y. Ma, J. Mu, W. Liu, F. Xu, Z. Li, J. Bai, H. Hua, D. Li, Mar. Drugs 17, 384 (2019)
A. Singh, V. Purohit, Drugs Ther. Perspect. 35, 278 (2019)
A. Jabbari, B. Tahmasbi, M. Nikoorazm, A. Ghorbani-Choghamarani, Appl. Organometal. Chem. 32, e4295 (2018)
B. Tahmasbi, M. Nikoorazm, P. Moradi, Y. Abbasi Tyula, RSC Adv. 12, 34303 (2022)
A. Jabbari, P. Moradi, M. Hajjami, B. Tahmasbi, Sci. Rep. 12, 11660 (2022)
A. Ghorbani-Choghamarani, Z. Darvishnejad, B. Tahmasbi, Inorg. Chim. Acta 435, 223 (2015)
K. Yoshiya, T. Shousuke, H. Kozo, Bull. Chem. Soc. Jpn. 40, 2458 (1967). https://doi.org/10.1246/bcsj.40.2458
R.K. Bindiya Devi, S. Pramodini Devi, R.K. Hemakumar Singh, Spectrosc. Lett. 45, 93 (2012). https://doi.org/10.1080/00387010.2011.603791
W. Chen, L. Qiang Mai, J. Feng Peng, Q. Xu, Q. Yao Zhu, J. Mater. Sci. 39, 2625 (2004)
M. Nikoorazm, N. Noori, B. Tahmasbi, S. Faryadi, Trans. Met. Chem. 42, 469 (2017)
M. Nikoorazm, A. Ghorbani-Choghamarani, A. Panahi, B. Tahmasbi, N. Noori, J. Iran. Chem. Soc. 15, 181 (2018)
M. Nikoorazm, Z. Rezaei, B. Tahmasbi, J. Porous Mater. 27, 671 (2020)
M. Szeląg, M. Janek, R. Panek, J. Madej, J. Fronczyk, Constr. Build. Mater. 344, 128253 (2022)
M. Kruk, M. Jaroniec, Langmuir 15(5279), 5284 (1999)
R.K. Khaled, M.A. Wahba, M.D. Badry, M.F. Zawrah, E.A. Heikal, J. Mater. Sci. 57, 4504 (2022)
X. Cao, J. Lu, Y. Zhao, R. Tian, W. Zhang, D. He, Y. Luo, Processes 9(2), 400 (2021)
D. Das, J.F. Lee, S. Cheng, J. Catal. 223, 152 (2004)
A. Ghorbani-Choghamarani, P. Moradi, B. Tahmasbi, RSC Adv. 6, 56458 (2016)
A. Rezaei, A. Ghorbani-Choghamarani, B. Tahmasbi, Catal. Lett. (2022). https://doi.org/10.1007/s10562-022-04135-8. (in press)
S. Molaei, M. Ghadermazi, Appl. Organometal. Chem. 33, e5328 (2020)
A. Saxena, A. Kumar, S. Mozumdar, J. Mol. Catal. A: Chem. 269, 35 (2007)
A. Ghorbani-Choghamarani, M. Nikoorazm, H. Goudarziafshar, B. Tahmasbi, Bull. Korean Chem. Soc. 30, 1388 (2009). https://doi.org/10.5012/bkcs.2009.30.6.1388
L. Song, W. Li, W. Duan, J. An, S. Tang, L. Li, G. Yang, Green Chem. 21, 1432 (2019)
L. Yang, S. Li, Y. Dou, S. Zhen, H. Li, P. Zhang, B. Yuan, G. Yang, Asian. J. Org. Chem. 6, 265 (2017)
M. Shariati, A. Rostami, G. Imanzadeha, S. Kheirjou, Mol. Catal. 461, 48 (2018)
S. Pakvojoud, M. Hatefi Ardakani, S. Saeednia, E. Heydari-Bafrooei, J. Sulphur Chem. 41, 561 (2020)
A. Ghorbani-Choghamarani, B. Tahmasbi, F. Arghand, S. Faryadi, RSC Adv. 5, 92174 (2015)
M. Zohrevandi, R. Mozafari, M. Ghadermazi, RSC Adv. 11, 14717 (2021)
A. Mahdian, M. Hatefi Ardakani, E. Heydari-Bafrooei, S. Saeednia, Appl. Organomet. Chem. 35, e6170 (2021)
S. Gao, L. Zhang, H. Yu, H. Wang, Z. He, C. Song, K. Huang, Macromol. Chem. Phys. 222, 2000375 (2021)
Acknowledgements
Authors thank the research facilities of Ilam University, Ilam, Iran, for financial support of this research project. Also, financial support for this work by the research affairs of Islamic Azad University, Qeshm Branch, Qeshm, Iran is gratefully acknowledged.
Funding
There are no financial resources.
Author information
Authors and Affiliations
Contributions
AJ contributed to methodology and investigation. MN contributed to supervision. PM contributed to writing—original draft, writing—review and editing.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Ethical approval and consent to participate
Authors declare no conflict of interest. All research results are absolutely correct. The article is only this one part that has been submitted to “Research on Chemical Intermediates.” Results are presented clearly, honestly and without fabrication, falsification or inappropriate data manipulation (including image-based manipulation). There is no plagiarism in the article
Consent for publication
All authors consent to the publication of this manuscript in “Research on Chemical Intermediates.”
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Jabbari, A., Nikoorazm, M. & Moradi, P. A V(O)-Schiff-base complex on MCM-41 as an efficient, reusable, and chemoselective nanocatalyst for the oxidative coupling of thiols and oxidation of sulfides. Res Chem Intermed 49, 1485–1505 (2023). https://doi.org/10.1007/s11164-023-04977-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11164-023-04977-w