Abstract
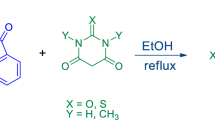
An efficient and easy method has been developed for the preparation of some new aminonaphthoquinone derivatives from a one-pot three-component condensation of 2-hydroxy-1,4-naphthoquinone, ninhydrin and heteroaryl amines under catalyst and solvent-free conditions at 75 °C. The protocol describes in situ generated imine as intermediate from the condensation reaction of ninhydrin with heteroaryl amines followed by the addition of 2-hydroxynaphthalene-1,4-dione to the imine, afforded the desired products. The operational simplicity of the procedure, shorter reaction times, simple workup procedure, clean reaction, easy purification of products by nonchromatographic methods, environmentally friendly conditions and high yields make this method much attractive.

Similar content being viewed by others
References
D.C. Mcmillan, S.D. Sarvate, J.E. Oatis Jr., D.J. Jollow, Toxicol. Sci. 2, 647 (2004)
J. Araldi, S.S. Guterres, Infarma 17, 78 (2005)
R. Pradhan, P. Dandawate, A. Vyas, S. Padhye, B. Biersack, R. Schobert, A. Ahmad, F.H. Sarkar, Curr. Drug Targets 13, 1777 (2012)
A. Kosmalska, M. Zaborski, A. Masek, Przem. Chem. 89, 420 (2010)
R.R. Korac, K.M. Khambholja, Pharmacogn. Rev. 5, 164 (2011)
R. Andoong, H. Chihchiang, Environ. Sci. Technol. 39, 7460 (2005)
A. Ostovari, S.M. Hoseinieh, M. Peikari, S.R. Shadizadeh, S.J. Hashemi, Corros. Sci. 51, 1935 (2009)
Y.M. Hijji, B. Barare, Y. Zhang, Sensors Actuat. B Chem. 169, 106 (2012)
S. Ruhemann, Trans. Chem. Soc. 97, 1438 (1910)
M. Friedman, J. Agric. Food Chem. 52, 385 (2004)
A. Sinhababu, S. Basu, H. Dey, Res. Chem. Intermed. 41, 2785 (2015)
G.M. LaPorte, R.S. Ramotowski, J. Forensic. Sci. 48, 658 (2003)
L. Schwarz, I. Frerichs, J. Forensic. Sci. 47, 1274 (2002)
P. Nagaraja, M.S. Hemantah Kumar, H.S. Yathirajan, J.S. Prakash, Anal. Sci. 18, 1027 (2002)
M. Pavia, A.J. Trujillo, B. Guamis, V. Ferragut, J. Dairy Sci. 83, 1441 (2000)
M. Moulin, C. Deleu, F.R. Larher, A. Bouchereau, Anal. Biochem. 308, 320 (2002)
G. Mohammadi Ziarani, N. Lashgari, F. Azimian, H.G. Kruger, P. Gholamzadeh, ARKIVOC 6, 1 (2015)
T.T. Guimaraes, F.R. Maria do Carmo, J.S. Lanza, M.N. Melo, L. Rubens, I.M. de Melo, E.B. Diogo, V.F. Ferreira, C.A. Camara, W.O. Valenca, R.N. de Oliveira, F. Frezard, E.N. da Silva Jr, Eur. J. Med. Chem. 63, 523 (2013)
M. Arundhati, C. Dipak, R. Mithun, Indian J. Pharm. Educ. Res. 52, 472 (2018)
A. Ahmad, K. Mahal, S. Padhye, F.H. Sarkar, R. Schobert, B. Biersack, J. Saudi. Chem. Soc. 21, 105 (2017)
A.P. Neves, C.C. Barbosa, S.J. Greco, M.D. Vargas, L.C. Visentin, C.B. Pinheiro, A.S. Mangrich, J.P. Barbosa, G.L. da Costae, J. Braz. Chem. Soc. 20, 712 (2009)
C. Festus, A.C. Ekennia, A.A. Osowole, L.O. Olasunkanmi, D.C. Onwudiwe, O.T. Ujam, Res. Chem. Intermed. 44, 5857 (2018)
N.M.F. Lima, C.S. Correia, P.A.L. Ferraz, A.V. Pinto, M.C.R.F. do Pinto, A.E.G. Santana, M.O.F. Goulart, J. Braz. Chem. Soc. 13, 822 (2002)
J.F. Allochio Filho, L.L. Roldi, M. Delarmelina, R.G. Fiorot, J.T. Andrade, Á.A. Aleixo, R.S. Carvalho, M.G.F. Araújo, J.M.S. Ferreira, A.G. Taranto, W. Romao, S.J. Greco, J. Braz. Chem. Soc. 27, 2127 (2016)
A.F.L. Vilela, B.M. Frugeri, A.L.F. Sarria, R.O.S. Kitamura, J.B. Fernandes, M.F.G.F. Silva, Q.B. Cass, C.L. Cardoso, J. Braz. Chem. Soc. 27, 535 (2016)
K. Mahal, A. Ahmad, F. Schmitt, J. Lockhauserbäumer, K. Starz, R. Pradhan, S. Padhye, F.H. Sarkar, W.S. Koko, R. Schobert, K. Ersfeld, B. Biersack, Eur. J. Med. Chem. 126, 421 (2017)
C.P.P. de Mello, N.S. Sardoux, L. Terra, L.C. Amorim, M.D. Vargas, G.B. da Silva, H.C. Castro, V.A. Giongo, L.F. Madeira, I.C.N.P. Paixao, Antivir. Ther. 21, 507 (2016)
M. Dabiri, Z. Noroozi Tisseh, A. Bazgir, Dyes Pigm. 89, 63 (2011)
H.R. Tavakoli, S.M. Moosavi, A. Bazgir, Res. Chem. Intermed. 41, 3041 (2015)
A. Olyaei, M. Sadeghpour, M. Khalaj, RSC Adv. 10, 30265 (2020)
A. Olyaei, M.S. Shahsavari, M. Sadeghpour, Res. Chem. Intermed. 44, 943 (2018)
A. Olyaei, R. Mohammad Ebrahimi, A. Adl, M. Sadeghpour, Chem. Heterocycl Compd. 55, 1104 (2019)
F. Noruzian, A. Olyaei, R. Hajinasiri, Res. Chem. Intermed. 45, 4383 (2019)
R. Khoeiniha, A. Olyaei, M. Saraei, J. Heterocyclic Chem. 54, 1746 (2017)
R. Khoeiniha, A. Olyaei, M. Saraei, Synth. Commun. 48, 155 (2018)
A. Olyaei, M. Saraei, R. Khoeiniha, Synlett 29, 1589 (2018)
Acknowledgements
The authors thank the Research Council of Payame Noor University for financial support.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Olyaei, A., Taheri, N. & Sadeghpour, M. Solvent and catalyst-free synthesis of some new aminonaphthoquinones from lawsone, ninhydrin and heteroaryl amines. Res Chem Intermed 47, 1211–1219 (2021). https://doi.org/10.1007/s11164-020-04325-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11164-020-04325-2