Abstract
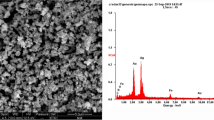
In this procedure, the synthesis of ecofriendly, magnetically retrievable BaFe12O19 was reported using a sulfated polysaccharide of algal origin, Irish moss (IM), as bio-matrix and capping agent. The characterization of BaFe12O19@IM was performed by some physicochemical characterization tools, such as Fourier transform infrared spectroscopy, X-ray powder diffraction, scanning electron microscopy, energy-dispersive X-ray spectroscopy, vibrating sample magnetometer, and thermogravimetric analysis. The catalytic performance and recyclability of the introduced heterogeneous catalyst have been explored in the one-pot synthesis of 2-amino-4H-pyrans and pyrans annulated heterocyclic compounds via a three-component reaction between aldehydes, malononitrile, and various C–H activated acidic compound under green reaction conditions. This environmentally benign catalyst displayed high catalytic activity and effective reusability and could maintain its high catalytic efficiency even after six recycling runs. Furthermore, the antibacterial activity of BaFe12O19@IM and the selected compound (4a) toward two types of clinically isolated bacterial strains Escherichia coli (E. coli) and Staphylococcus aureus (S. aureus) was evaluated. The results revealed that BaFe12O19@IM and 2-amino-4-(4-chlorophenyl)-7,7-dimethyl-5-oxo-5,6,7,8-tetrahydro-4H-chromene-3-carbonitrile (4a) showed an effective antibacterial activity against gram-positive S. aureus.
Graphic abstract












Similar content being viewed by others
Change history
26 September 2021
A Correction to this paper has been published: https://doi.org/10.1007/s11164-021-04593-6
References
R.G. Saratale, G.D. Saratale, H.S. Shin, J.M. Jacob, A. Pugazhendhi, M. Bhaisare, G. Kumar, Environ. Sci. Pollut. Res. 25, 10164 (2018)
A. Pugazhendhi, T.N.J.I. Edison, I. Karuppusamy, B. Kathirvel, Int. J. Pharm. 539, 104 (2018)
S. Jeyarani, N.M. Vinita, P. Puja, S. Senthamilselvi, U. Devan, A.J. Velangani, M. Biruntha, A. Pugazhendhi, P. Kumar, J. Photochem. Photobiol. B 202, 111715 (2020)
T. Parandhaman, N. Pentela, B. Ramalingam, D. Samanta, S.K. Das, ACS Sustain. Chem. Eng. 5, 489 (2017)
Z. Jiang, K. Shan, J. Song, J. Liu, S. Rajendran, A. Pugazhendhi, J.A. Jacob, B. Chen, Life Sci. 220, 156 (2019)
P.S. Murphin Kumar, S. Thiripuranthagan, T. Imai, G. Kumar, A. Pugazhendhi, S.R. Vijayan, ACS Sustain. Chem. Eng. 5, 11290 (2017)
P.S.M. Kumar, V.K. Ponnusamy, K.R. Deepthi, G. Kumar, A. Pugazhendhi, H. Abe, J. Mater. Chem. A. 6, 23435 (2018)
J. Kothandapani, S.S. Ganesan, Curr. Org. Chem. 23, 313 (2019)
K.R. Reddy, P.A. Reddy, C.V. Reddy, N.P. Shetti, B. Babu, K. Ravindranadh, M.V. Shankar, M.C. Reddy, S. Soni, S. Naveen, Methods Microbiol. 46, 227 (2019)
O. Zaitseva, D.A. Vinnik, E.A. Trofimov, Mater. Sci. Forum 946, 186 (2019)
K. Watanabe, T. Fujihara, K. Watanabe, K. Kakizaki, K. Kamishima, J. Phys. Soc. Jpn. 89, 014704 (2020)
D. Vinnik, F. Podgornov, N. Zabeivorota, E. Trofimov, V. Zhivulin, A. Chernukha, M. Gavrilyak, S. Gudkova, D. Zherebtsov, A. Ryabov, J. Magn. Magn. Mater. 498, 166190 (2020)
S. Dagar, A. Hooda, S. Khasa, M. Malik, J. Alloys Compd. 826, 154214 (2020)
T. Piri, R. Peymanfar, S. Javanshir, S. Amirnejat, Catal. Lett. 149, 3384 (2019)
S. Amirnejat, A. Nosrati, S. Javanshir, M.R. Naimi-Jamal, Int. J. Biol. Macromol. 152, 834 (2020)
C. Chellapandian, B. Ramkumar, P. Puja, R. Shanmuganathan, A. Pugazhendhi, P. Kumar, Process Biochem. 80, 58 (2019)
M. Srinivasan, M. Venkatesan, V. Arumugam, G. Natesan, N. Saravanan, S. Murugesan, S. Ramachandran, Process Biochem. 80, 197 (2019)
M. Venkatesan, V. Arumugam, R. Pugalendi, K. Ramachandran, K. Sengodan, S.R. Vijayan, U. Sundaresan, Process Biochem. 84, 196 (2019)
B. Hemmati, S. Javanshir, Z. Dolatkhah, RSC Adv. 6, 50431 (2016)
R. Prabhu, R. Mohamed Asik, R. Anjali, G. Archunan, N.M. Prabhu, A. Pugazhendhi, N. Suganthy, Process Biochem. 84, 39 (2019)
A. Maleki, Z. Hajizadeh, Silicon Chem. 11, 2789 (2019)
E. Pourian, S. Javanshir, Z. Dolatkhah, S. Molaei, A. Maleki, ACS Omega 3, 5012 (2018)
F. Norouzi, S. Javanshir, BMC Chem. 14, 1 (2020)
D. Kumar, P. Sharma, H. Singh, K. Nepali, G.K. Gupta, S.K. Jain, F. Ntie-Kang, RSC Adv. 7, 36977 (2017)
K. Niknam, A. Piran, Green Sustain. Chem. 3, 1 (2013)
D. Armesto, W.M. Horspool, N. Martin, A. Ramos, C. Seoane, J. Org. Chem. 54, 3069 (1989)
G. Reynolds, K.H. Drexhage, Opt. Commun. 13, 222 (1975)
N. Gouault, J.-F. Cupif, A. Sauleau, M. David, Tetrahedron Lett. 41, 7293 (2000)
E.A.A. Hafez, M.H. Elnagdi, A.G.A. Elagamey, F.M.A.A. El-Taweel, Heterocycles 26, 903 (1987)
P. Rai, A. Ibad, H. Sagir, I. Siddiqui, ChemistrySelect. 1, 1300 (2016)
A. Maleki, Z. Varzi, F. Hassanzadeh-Afruzi, Polyhedron 171, 193 (2019)
M.S. Esmaeili, M.R. Khodabakhshi, A. Maleki, Z. Varzi, Polycycl. Aromat. Compd. (2020). https://doi.org/10.1080/10406638.2019.1708418
S. Pan, P. Li, G. Xu, J. Guo, L. Ke, C. Xie, Z. Zhang, Y. Hui, Res. Chem. Intermed. 46, 1353 (2020)
A. Khazaei, F. Gholami, V. Khakyzadeh, A.R. Moosavi-Zare, J. Afsar, RSC Adv. 5, 14305 (2015)
M.T. Maghsoodlou, N. Hazeri, M. Lashkari, F.N. Shahrokhabadi, B. Naghshbandi, M.S. Kazemi-doost, M. Rashidi, F. Mir, M. Kangani, S. Salahi, Res. Chem. Intermed. 41, 6985 (2015)
P.B. Hiremath, K. Kantharaju, ChemistrySelect. 5, 1896 (2020)
M.S. Samuel, S. Jose, E. Selvarajan, T. Mathimani, A. Pugazhendhi, J. Photochem. Photobiol. B. 202, 111642 (2020)
A. Pugazhendhi, D. Prabakar, J.M. Jacob, I. Karuppusamy, R.G. Saratale, Microb. Pathog. 114, 41–45 (2018)
E. Tacconelli, E. Carrara, A. Savoldi, S. Harbarth, M. Mendelson, D.L. Monnet, C. Pulcini, G. Kahlmeter, J. Kluytmans, Y. Carmeli, Lancet Infect. Dis. 18, 318 (2018)
M. Saravanan, S.K. Barik, D. MubarakAli, P. Prakash, A. Pugazhendhi, Microb. Pathog. 116, 221 (2018)
A. Pugazhendhi, S.S. Kumar, M. Manikandan, M. Saravanan, Microb. Pathog. 122, 84 (2018)
P. Nallasamy, T. Ramalingam, T. Nooruddin, R. Shanmuganathan, P. Arivalagan, S. Natarajan, Process Biochem. 92, 355 (2020)
E. Nazarzadeh Zare, R. Jamaledin, P. Naserzadeh, E. Afjeh-Dana, B. Ashtari, M. Hosseinzadeh, R. Vecchione, A. Wu, F.R. Tay, A. Borzacchiello, P. Makvandi, Appl. Mater. Interfaces 12, 3279 (2019)
K. Gold, B. Slay, M. Knackstedt, A.K. Gaharwar, Adv. Ther. 1, 3 (2018)
S. Ma, L. Chen, X. Liu, D. Li, N. Ye, L. Wang, Int. J. Green Energy 9, 13–21 (2012)
W.A.K. Mahmood, M.M.R. Khan, T.C. Yee, J. Phys. Sci. 25, 123 (2014)
A. Maleki, J. Rahimi, K. Valadi, Nano Struct. Nano Objects 18, 100264 (2019)
A. Maleki, M. Aghaei, H.R. Hafizi-Atabak, M. Ferdowsi, Ultrason. Sonochem. 37, 260 (2017)
M. Bakherad, F. Moosavi, A. Keivanloo, R. Doosti, E. Moradian, M. Armaghan, Res. Chem. Intermed. 45, 2981 (2019)
M. Zabihzadeh, A. Omidi, F. Shirini, H. Tajik, M.S.N. Langarudi, J. Mol. Struct. 1206, 127730 (2020)
B. Maleki, O. Reiser, E. Esmaeilnezhad, H.J. Choi, Polyhedron 162, 129 (2019)
S. Banerjee, A. Horn, H. Khatri, G. Sereda, Tetrahedron Lett. 52, 1878 (2011)
M. Honarmand, A. Tzani, A. Detsi, J. Iran. Chem. Soc. 16, 571 (2019)
S. Gao, C.H. Tsai, C. Tseng, C.-F. Yao, Tetrahedron Lett. 64, 9143 (2008)
A.V. Chate, R.M. Dongre, M.K. Khaire, G.M. Bondle, J.N. Sangshetti, M. Damale, Res. Chem. Intermed. 44, 6119 (2018)
A. Maleki, A.A. Jafari, S. Yousefi, Carbohydr. Polym. 175, 409 (2017)
X.-S. Wang, Z.-S. Zeng, M.-M. Zhang, Y.-L. Li, D.-Q. Shi, S.-J. Tu, X.-Y. Wei, Z.-M. Zong, J. Chem. Res. Synop. 4, 228 (2006)
N. Azizi, S. Dezfooli, M. Khajeh, M.M. Hashemi, J. Mol. Liq. 186, 76 (2013)
I. Kharbangar, M.R. Rohman, H. Mecadon, B. Myrboh, Int. J. Org. Chem. 2, 282 (2012)
A. Fihri, C. Len, R.S. Varma, A. Solhy, Coord. Chem. Rev. 347, 48 (2017)
S.K. Dangolani, F. Panahi, M. Nourisefat, A. Khalafi-Nezhad, RSC Adv. 6, 92316 (2016)
M. Hajjami, F. Gholamian, R.H. Hudson, A.M. Sanati, Catal. Lett. 149, 228 (2019)
B. Eshtehardian, M. Rouhani, Z. Mirjafary, J. Iran. Chem. Soc. 17, 469 (2020)
Acknowledgements
The authors wish to express their gratitude for the financial support provided by the Research Council of Iran University of Science and Technology (IUST), Tehran, Iran
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The original online version of this article was revised: The corresponding author was incorrectly published as “Sara Amirnejat”. However, it should be “Shahrazad Javanshir”.
Rights and permissions
About this article
Cite this article
Amirnejat, S., Nosrati, A., Peymanfar, R. et al. Synthesis and antibacterial study of 2-amino-4H-pyrans and pyrans annulated heterocycles catalyzed by sulfated polysaccharide-coated BaFe12O19 nanoparticles. Res Chem Intermed 46, 3683–3701 (2020). https://doi.org/10.1007/s11164-020-04168-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11164-020-04168-x