Abstract
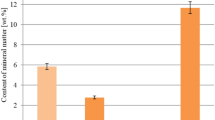
The efficient/low-cost activated carbons were prepared from inedible fruits such as Cerbera odollam Gaertn, Terminalia catappa, Ficus lyrata, Couroupita guianensis Aubl and Hevea brasiliensis as well as biochars of Combretum quadrangulare and Leucaena Leucocephala (LL). The physical and chemical activation processes were applied to improve the Fe3+ adsorption efficiency. As obtained results, LL heated at 500 °C for 2 h (LL502) exhibited best performance for Fe3+ adsorption with a qe value of Fe3+ (28.18 mg/g). The physicochemical properties of LL502 such as BET surface area, total acid–base amount and pHpzc were 247.3 m2/g, 2.2647 meq/g and 8.49, respectively. Moreover, the adsorption behaviors of Fe3+ onto the surface of LL502 were found to be monolayer, physisorption and rapid adsorption processes which could be confirmed by Langmuir and Dubinin–Radushkevich isotherms and pseudo-second order kinetic models, respectively. Adsorption thermodynamics also indicated that the Fe3+ adsorption processes were endothermic and spontaneous in nature. In addition, 98% of iron removal from ground/surface water was successfully achieved by using LL502. The quality of ground/surface water was acceptable based on comparison with a water quality standard. This research was as expected since as-prepared low-cost adsorbent could be verily applied in a practical treatment process.
Graphical abstract






Similar content being viewed by others
References
C. Dalai, R. Jha, V.R. Desai, Aquat. Procedia 4, 1126 (2015)
H. Jeong, H. Kim, T. Jang, Water 169, 1 (2016)
D. Vollprecht, L. Krois, K.P. Sedlazeck, P. Müller, R. Mischitz, T. Olbrich, R. Pomberger, J. Cleaner Prod. 208, 1409 (2019)
K. Sasaki, Y. Hayashi, K. Toshiyuki, B. Guo, Chemosphere 207, 139 (2018)
L. Cai, L. Cui, B. Lin, J. Zhang, Z. Huang, J. Cleaner Prod. 202, 759 (2018)
N. Chitpong, S.M. Husson, Sep. Purif. Technol. 179, 94 (2017)
J. Valentín-Reyes, R.B. García-Reyes, A. García-González, E. Soto-Regalado, F. Cerino-Córdova, J. Environ. Manage. 236, 815 (2019)
P. Maneechakr, S. Karnjanakom, J. Chem. Thermodyn. 106, 104 (2017)
P. Maneechakr, P. Chaturatphattha, S. Karnjanakom, Res. Chem. Intermed. 44, 7135 (2018)
W. Phairuang, M. Hata, M. Furuuchi, J. Environ. Sci. 52, 85 (2017)
W. Shen, Z. Li, Y. Liu, Chem. Eng. 1, 27 (2008)
R.B. Fidel, D.A. Laird, M.L. Thompson, J. Environ. Qual. 42, 1771 (2013)
Q. Shi, A. Terracciano, Y. Zhao, C. Wei, C. Christodoulatos, X. Meng, Sci. Total Environ. 648, 176 (2019)
Y. Gao, S. Xu, Q. Yue, Y. Wu, B. Gao, J. Taiwan Inst. Chem. Eng. 61, 327 (2016)
J.P. Chen, S. Wu, Langmuir 20, 2233 (2004)
L. Huang, J. Kong, W. Wang, C. Zhang, S. Niu, B. Gao, Desalination 286, 268 (2012)
M.H. Marzbali, M. Esmaieli, H. Abolghasemi, M.H. Marzbali, Process Saf. Environ. Prot. 102, 700 (2016)
M. Kılıç, E. Apaydın-Varol, A.E. Pütün, Appl. Surf. Sci. 261, 247 (2012)
Z. Sun, Y. Yu, S. Pang, D. Du, Appl. Surf. Sci. 284, 100 (2013)
H. Shayesteh, A. Rahbar-Kelishami, R. Norouzbeigi, J. Mol. Liq. 221, 1 (2016)
M. Ghaedi, A. Ansari, F. Bahari, A.M. Ghaedi, A. Vafaei, Spectrochim. Acta, Part A 137, 1004 (2015)
P. Roy, N.K. Mondal, K. Das, J. Environ. Chem. Eng. 2, 585 (2014)
C. Meng, W. Zhikun, L. Qiang, L. Chunling, S. Shuangqing, H. Songqing, J. Hazard. Mater. 341, 198 (2018)
H.D. Choi, W.S. Jung, J.M. Cho, B.G. Ryu, J.S. Yang, K. Baek, J. Hazard. Mater. 166, 642 (2009)
S. Karnjanakom, P. Maneechakr, J. Mol. Struct. 1186, 80 (2019)
H. Sayğılı, F. Güzel, Ecotoxicol. Environ. Saf. 131, 22 (2016)
M.N. Sepehr, A. Amrane, K.A. Karimaian, M. Zarrabi, H.R. Ghaffari, J. Taiwan Inst. Chem. Eng. 45, 635 (2014)
A. Sheibani, M. Shishehbor, H. Alaei, Int. J. Ind. Chem. 3, 1 (2012)
K. Leila, C. M. Luisa, S. N. Soaad, B. Mohamed, Sep. Sci. Technol.
M.E. Goher, A.M. Hassan, I.A. Abdel-Moniem, A.H. Fahmy, M.H. Abdo, S.M. El-sayed, Egypt. J. Aquat. Res. 41, 155 (2015)
Acknowledgements
The authors wish to acknowledge Department of Chemistry, Faculty of Science, Rangsit University for supporting all instruments and chemicals.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Maneechakr, P., Karnjanakom, S. The essential role of Fe(III) ion removal over efficient/low-cost activated carbon: surface chemistry and adsorption behavior. Res Chem Intermed 45, 4583–4605 (2019). https://doi.org/10.1007/s11164-019-03851-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11164-019-03851-y