Abstract
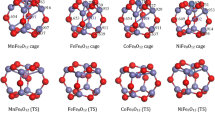
The transition metals (TM) doping a Fe9O12 cage have been systemically studied by density functional theory. And then the structures, electronic and magnetic properties of them have been discussed. It can be found that V, Nb, Mo, Ta and W atoms prefer to encapsulate in a Fe9O12 cage and then form a core@shell structure. Here, Ta@Fe9O12 is proposed to be the most favorable structure. Zr, Hf and Ta atoms are conducive to stabilize a Fe9O12 cage. All the TM doping Fe9O12 clusters are more kinetically active. The highest occupied molecular orbital (HOMO) and the lowest unoccupied molecular orbital (LUMO) of TM doping Fe9O12 clusters are mostly by virtue of d orbital distribution. More electrons are transferred from the Zr and Hf atoms to the Fe9O12 cage than from other transition metals. Only the spin values of Mn, Cr, and Ti atoms in a Fe9O12 cage are higher than 2.5 μB.






Similar content being viewed by others
References
Y. Li, C. Cai, C. Zhao, Y. Gu, Mod. Phys. Lett. B. 30, 1650239 (2018)
L.T. Lu, N.T. Dung, L.D. Tung, C.T. Thanh, O.K. Quy, N.V. Chuc, S. Maenosono, N.T.K. Thanh, Nanoscale 7, 19596 (2015)
J. Liu, Z. Sun, Y. Deng, Y. Zou, C. Li, X. Guo, L. Xiong, Y. Gao, F. Li, D. Zhao, Angew. Chem. Int. Ed. 48, 5875 (2009)
B. Ankamwar, T.C. Lai, J.H. Huang, R.S. Liu, M. Hsiao, C.H. Chen, Y.K. Hwu, Nanotechnology 21, 75102 (2010)
X. Liu, X. Duan, Q. Qin, Q. Wang, W. Zheng, CrystEngComm 15, 3284 (2013)
M.J. Bradley, A.J. Biacchi, R.E. Schaak, Chem. Mater. 25, 1886 (2013)
Z. Xu, Y. Hou, S. Sun, J. Am. Chem. Soc. 129, 8698 (2007)
D.J. Huang, C.F. Chang, H.-T. Jeng, G.Y. Guo, H.-J. Lin, W.B. Wu, H.C. Ku, A. Fujimori, Y. Takahashi, C.T. Chen, Phys. Rev. Lett. 93, 077204 (2004)
D. Odkhuu, P. Taivansaikhan, W.S. Yun, S.C. Hong, J. Appl. Phys. 115, 17A916 (2014)
R.C. Rai, S. Wilser, M. Guminiak, B. Cai, M.L. Nakarmi, Appl. Phys. A 106, 207 (2012)
Q.-C. Sun, H. Sims, D. Mazumdar, J.X. Ma, B.S. Holinsworth, K.R. O’Neal, G. Kim, W.H. Butler, A. Gupta, J.L. Musfeldt, Phys. Rev. B. 86, 205106 (2012)
Q. Song, Z.J. Zhang, J. Am. Chem. Soc. 134, 10182 (2012)
J.C. Garcia, W.V.M. Machado, L.V.C. Assali, J.F. Justo, Diam. Relat. Mater. 20, 1222 (2011)
Y. Hu, C. Ji, X. Wang, J. Huo, Q. Liu, Y. Song, Sci. Rep-UK 7, 16485 (2017)
D.Z. Zhang, Y.E. Sun, C.X. Jiang, Y. Zhang, Sensor. Actuat. B-Chem. 242, 15 (2017)
D.Z. Zhang, J.F. Wu, P. Li, Y.H. Cao, J. Mater. Chem. A. 5, 20666 (2017)
D.Z. Zhang, M.S. Pang, J.F. Wu, Y.H. Cao, New J. Chem. 43, 4900 (2019)
D.Z. Zhang, H.Y. Chang, Y.E. Sun, C.X. Jiang, Y. Yao, Y. Zhang, Sens. Actuator B-Chem. 252, 624 (2017)
D.Z. Zhang, Y.E. Sun, C.X. Jiang, Y. Yao, D.Y. Wang, Y. Zhang, Sens. Actuator B-Chem. 253, 1120 (2017)
J.F. Wu, D.Z. Zhang, Y.H. Cao, J. Colloid Interface Sci. 529, 556 (2018)
Z. Zhao, Z. Li, Q. Wang, Mater. Res. Express. 5, 065605 (2018)
B. Delley, J. Chem. Phys. 92, 508 (1990)
B. Delley, J. Chem. Phys. 113, 7756 (2000)
J.P. Perdew, K. Burke, M. Ernzerhof, Phys. Rev. Lett. 77, 3865 (1996)
S.A. Khandy, D.C. Gupta, RSC Adv. 6, 48009 (2016)
Z. Zhao, Z. Li, Q. Wang, D. Wang, C. Wu, Z. Zhou, Comput. Theor. Chem. 1095, 9 (2016)
Z. Li, Z. Zhou, H. Wang, S. Li, Z. Zhao, J. Cryst. Growth 449, 22 (2016)
Z. Li, Z. Zhao, Phase Transit. 91, 426 (2018)
R.S. Mülliken, J. Chem. Phys. 23, 1841 (1955)
S.A. Khandy, I. Islam, D.C. Gupta, R. Khenata, A. Laref, Sci. Rep-UK 9, 1475 (2019)
S.A. Khandy, Mater. Res. Express. 5, 056516 (2018)
S.A. Khandy, I. Islam, D.C. Gupta, A. Laref, J. Mol. Model. 24, 131 (2018)
Z. Li, Z. Zhou, Z. Zhao, Q. Wang, Int. J. Mod. Phys. B. 32, 1850187 (2018)
C. Zhang, H. Cui, J. Shen, Chin. Phys. B. 21, 103102 (2012)
X.L. Ding, W. Xue, Y.P. Ma, Z.C. Wang, S.G. He, J. Chem. Phys. 130, 014303 (2009)
J. Fan, L.-S. Wang, J. Chem. Phys. 102, 8714 (1995)
X. Xiao, G. Liu, B. Hu, J. Wang, A. Ullah, Mater. Charact. 82, 130 (2013)
Z. Li, Z. Zhao, Q. Wang, X. Yin, Mater. Res. Express. 5, 046105 (2018)
Z. Li, Z. Zhao, Z. Zhou, Q. Wang, Mater. Res. Express. 5, 026524 (2018)
Z. Li, Z. Zhao, S. Li, Q. Wang, Comput. Mater. Sci. 110, 340 (2015)
Z. Li, Z. Zhao, S. Li, Q. Wang, Solid State Commun. 221, 5 (2015)
Z. Li, Z. Zhao, Mater. Chem. Phys. 187, 54 (2017)
Z. Li, Z. Zhao, Res. Chem. Intermediat. 45, 833 (2019)
X. Wang, H. Qin, Y. Chen, J. Hu, J. Phys. Chem. C 118, 28548 (2014)
P. Tereshchuk, J.L.F.D. Silva, Phys. Rev. B. 85, 195461 (2012)
G.S. Shahane, K.V. Zipare, S.S. Bandgar, V.L. Mathe, J. Mater. Sci. Mater. Electron. 28, 4146 (2017)
Acknowledgements
We gratefully acknowledge the financial support from the Key Fund Project of the National Science Foundation, People’s Republic of China (Grant No. 51634004), the Doctoral Scientific Research Foundation of Liaoning Province (Grant No. 20180551213), Key Laboratory of Chemical Metallurgy Engineering Liaoning Province, University of Science and Technology Liaoning (Grant No. USTLKFSY201711) and the Fund Project of University of Science and Technology Liaoning (Grant No. 2017YY02).
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Li, Z., Zhao, Z. The selectivity of the transition metals encapsulated in a Fe9O12 cage. Res Chem Intermed 45, 4573–4582 (2019). https://doi.org/10.1007/s11164-019-03850-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11164-019-03850-z