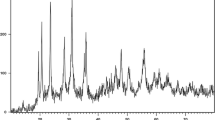
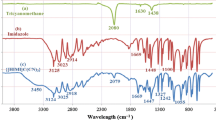
Abstract
A four-component reaction of hydrazine hydrate, ethyl cyanoacetate, malononitrile, and aromatic aldehydes was achieved in the presence of nanocrystalline MIIZr4(PO4)6 ceramics (MII: Mn, Fe, Co, Ni, Cu, Zn, Cd) as heterogeneous catalysts to produce N-amino-2-pyridones. The reactions were performed in the presence of different catalysts, and it is observed that CdZr4(PO4)6 nanocrystallines are the best catalysts among those examined. Atom economy, excellent yields in short times, high catalytic activity, recycling of catalyst, and environmental benignity are some of the important features of this protocol.



Similar content being viewed by others
References
J.P. Wan, Y. Liu, RSC Adv. 2, 9763 (2012)
A. Kumar, M. Kumar Gupta, M. Kumar, D. Saxena, RSC Adv. 3, 1673 (2013)
A. Banu, R.S. Lamani, I.A.M. Khazi, N. Shahina Begum, J. Saud. Chem. Soc. 18, 371 (2014)
H. Shojaei, Z.L. Böhmer, P.V. Zezschwitz, J. Org. Chem. 72, 5091 (2007)
P.S. Dragovich, T.J. Prins, R. Zhou, T.O. Johnson, Y. Hua, J. Med. Chem. 46, 4572 (2003)
Q. Li, L.A. Mitscher, L.L. Shen, Med. Res. Rev. 20, 231 (2000)
R.J. Cox, D. O’Hagan, J. Chem. Soc. Perkin Trans. 1, 2537 (1991)
M. Nagarajan, X.S. Xiao, S. Antony, G. Kohlhagen, Y. Pommier, M. Cushman, J. Med. Chem. 46, 5712 (2003)
R.K. Beckman, J.C. Potenza, E.J. Enholm, J. Org. Chem. 52, 469 (1987)
Q.Y. Zhuang, J.N. Xu, S.J. Tu, R.H. Jia, J.Y. Zhang, C.M. Li, D.X. Zhou, Chin. J. Chem. 25, 1568 (2007)
D.L. Comins, X. Chen, S.P. Joseph, Tetrahedron Lett. 37, 9275 (1996)
M. Seifi, M. Khajehasani Rabori, H. Sheibani, Mod. Res. Catal. 2, 8 (2013)
M.R.H. Elmoghayar, A.G.A. El-Agamey, M.Y. Abdel-Samad Nasr, M.M.M. Sallam, J. Heterocycl. Chem. 21, 1885 (1984)
F.F. Abdel-Latif, R. Mekheimer, E.K. Ahmed, T.B. Abdel-Aleem, Pharmazie 48, 736 (1993)
A.H.M. Hussein, Heteroat. Chem. 8, 1 (1997)
E.A. El-Rady, M.A. Khalil, J. Chin. Chem. Soc. 51, 779 (2004)
W.M. Basyouni, Acta Chem. Slov. 50, 223 (2003)
E.M. Zayed, E.A. Hafez, S.A.S. Ghozlan, A.A.H. Ibrahim, Heterocycles 22, 2553 (1984)
W.R. Abdel-Monem, Chem. Pap. 58, 276 (2004)
A.A. Harb, Chem. Pap. 58, 260 (2004)
M. Shankar Singh, S. Chowdhury, RSC Adv. 2, 4547 (2012)
V. Estévez, M. Villacampa, J.C. Menéndez, Chem. Soc. Rev. 39, 4402 (2010)
A. Bhunia, T. Kaicharla, D. Porwal, R.G. Gonnade, A.T. Biju, Chem. Commun. 50, 11389 (2014)
F. Alonso, Y. Moglie, G. Radivoy, M. Yus, J. Org. Chem. 76, 8394 (2011)
J. Safaei-Ghomi, H. Shahbazi-Alavi, S. Kalhor, Monatsh. Chem. (2016). doi:10.1007/s00706-016-1693-y
J. Safaei-Ghomi, P. Babaei, H. Shahbazi-Alavi, S. Zahedi, J. Saud. Chem. Soc. (2016). doi:10.1016/j.jscs.2016.01.003
M. Lakshmi Kantam, K.B. Shiva Kumar, C. Sridhar, Adv. Synth. Catal. 347, 1212 (2005)
V. Polshettiwar, R.S. Varma, Green Chem. 12, 743 (2010)
N. Gorodylova, V. Kosinova, Z. Dohnalova, P. Belina, P. Sulcova, Dyes Pigm. 98, 393 (2013)
D.M. Liu, Mater. Chem. Phys. 36, 350 (1994)
J. Safaei-Ghomi, M. Navvab, H. Shahbazi-Alavi, Ultrason. Sonochem. 31, 102 (2016)
K. Tanabe, Solid Acids and Bases (Academic Press, New York, 1970)
C. Zener, Phys. Rev. 82, 403 (1951)
J.Y. Guo, C.X. Xu, Appl. Phys. A 102, 333 (2011)
J. Ikhsan, B.B. Johnson, J.D. Wells, J. Colloid Interface Sci. 217, 403 (1999)
S. Myradalyyev, T. Limpanuparb, X. Wang, H. Hirao, Polyhedron 52, 96 (2013)
C.M. Aldao, I.M. Vitomirov, F. Xu, J.H. Weaver, Phys. Rev. B 37, 6019 (1988)
A.A. Ensafi, E. Heydari-Bafrooei, B. Rezaei, Anal. Chem. 85, 991 (2013)
Acknowledgments
The authors acknowledge a reviewer who provided helpful insights. Meanwhile, the authors are grateful to the University of Kashan for supporting this work by Grant No: 159196/XXII. Also, the authors are grateful to Dr. Shayesteh Asadollahi for their help.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Safaei-Ghomi, J., Shahbazi-Alavi, H. & Ziarati, A. A comparative screening of the catalytic activity of nanocrystalline MIIZr4(PO4)6 ceramics in the one-pot synthesis of 1,6-diamino-4-aryl-2-oxo-1,2-dihydropyridine-3,5-dicarbonitrile derivatives. Res Chem Intermed 43, 91–101 (2017). https://doi.org/10.1007/s11164-016-2608-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11164-016-2608-6