Abstract
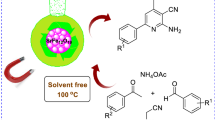
This paper reports an efficient heterogeneous catalyst based on sulfonic acid functionalization of magnetic nanoparticles. This new catalyst was prepared using the reaction between magnetic nanoparticles and sodium 2-bromoethane-1-sulfonate. Magnetic nanoparticles functionalized ethane sulfonic acid (MNESA) was found as efficient catalyst for the synthesis of coumarin derivatives using Pechmann condensation under mild condition. This reaction was catalyzed by MNESA under solvent-free condition at 90 °C, to give the corresponding products in excellent yields. The catalyst is easily separated from the reaction condition and can be reused for several times with consistence in the activity.









Similar content being viewed by others
References
J.A. Melero, J. Iglesiasa, G. Morales, Green Chem. 11, 1285 (2009)
A.F. Lee, J.A. Bennett, J.C. Manayil, K. Wilson, Chem. Soc. Rev. 43, 7887 (2014)
R. Ghanbaripour, I. Mohammadpoor-Baltork, M. Moghadam, A.R. Khosropour, S. Tangestaninejad, V. Mirkhani, Polyhedron 31, 721 (2012)
R. Ghanbaripour, I. Mohammadpoor-Baltork, M. Moghadam, A.R. Khosropour, S. Tangestaninejad, V. Mirkhani, J. Iran. Chem. Soc. 9, 791 (2012)
R. Mrówczyński, A. Nan, J. Liebscher, RSC Adv. 4, 5927 (2014)
M.B. Gawande, P.S. Brancoa, R.S. Varma, Chem. Soc. Rev. 42, 3371 (2013)
R. Ghanbaripour, M. Samadizadeh, G. Honarpisheh, M. Abdolmohammad, Synlett 26, 2117 (2015)
V. Polshettiwar, R. Luque, A. Fihri, H. Zhu, M. Bouhrara, J.M. Basset, Chem. Rev. 111, 3036 (2011)
H. Naeimi, S. Mohamadabadi, Dalton Trans. 43, 12967 (2014)
C.S. Gill, B.A. Price, C.W. Jones, J. Catal. 251, 145 (2007)
H. Mahmoudi, A.A. Jafari, ChemCatChem 5, 3743 (2013)
A. Mobaraki, B. Movassagh, B. Karimi, ACS Comb. Sci. 16, 352 (2014)
B. Movassagh, L. Tahershamsi, A. Mobaraki, Tetrahedron Lett. 56, 1851 (2015)
A. Khalafi-Nezhad, S. Mohammadi, ACS Comb. Sci. 15, 512 (2013)
V.H. Pechmann, C. Duisberg, Chem. Ber. 17, 929 (1884)
D.S. Bose, A.P. Rudradas, M.H. Babu, Tetrahedron Lett. 43, 9195 (2002)
M.S. Manhas, S.N. Ganguly, S. Mukherjee, A.K. Jian, A.K. Bose, Tetrahedron Lett. 47, 2423 (2006)
F. Rajabi, A. Feiz, R. Luque, Catal. Lett. 145, 1621 (2015)
N.G. Khaligh, Catal. Sci. Technol. 2, 1633 (2012)
H. Wang, Monatsh. Chem. 144, 411 (2013)
Y. Zhang, A. Zhu, Q. Li, L. Li, Y. Zhao, J. Wang, RSC Adv. 4, 22946 (2014)
M.S. Peng, J. Cai, Dyes Pigm. 79, 270 (2008)
M. Roussaki, C.A. Kontogiorgis, D. Hadjipavlou-Litina, S. Hamilakis, A. Detsi, Bioorg. Med. Chem. Lett. 20, 3889 (2010)
B. Tyagi, M.K. Mishra, R.V. Jasra, J. Mol. Catal. A: Chem. 286, 41 (2008)
V.O. Iaroshenko, S. Ali, T.M. Babar, S. Dudkin, S. Mkrtchyan, N.H. Rama, A. Villinger, P. Langer, Tetrahedron Lett. 52, 373 (2011)
J.R. Jonhnson, Org. React. 1, 210 (1942)
F. Jones, O. Piermatti, F. Pizzo, Heterocycles 43, 1257 (1996)
R.L. Shirner, Org. React. 1, 1 (1942)
I. Yavari, R. Hekmat-Shoar, A. Zonouzi, Tetrahedron Lett. 39, 2391 (1998)
R.O. Kennedy, R.D. Zhorenes, Coumarins: Biology, Applications and Mode of Action (Wiley, Chichester, 1997)
G.J. Fan, W. Mar, M.K. Park, E. Wook Choi, K. Kim, S. Kim, Bioorg. Med. Chem. Lett. 11, 2361 (2001)
G. Cravotto, G.M. Nano, G. Palmisano, S. Tagliapietra, Tetrahedron Asymmetry 12, 707 (2001)
N.A. Petasis, A.N. Butkevich, J. Organomet. Chem. 694, 1747 (2009)
A. Alizadeh, R. Ghanbaripour, Synlett 25, 2777 (2014)
D. Tejedor, L. Cotos, F. García-Tellado, J. Org. Chem. 78, 8853 (2013)
N. Majumdar, K.A. Korthals, W.D. Wulff, J. Am. Chem. Soc. 134, 1357 (2012)
A. Alizadeh, R. Ghanbaripour, L.G. Zhu, Synlett 25, 1596 (2014)
T. Amanpour, K. Zangger, F. Belaj, A. Bazgir, D. Dallinger, C.O. Kappe, Tetrahedron 71, 7159 (2015)
A. Alizadeh, R. Ghanbaripour, L.G. Zhu, Tetrahedron 70, 2048 (2014)
H. Sharghi, M. Jokar, Heterocycles 71, 12 (2007)
E.C. Horning, Organic Synthesis, vol. III (Wiley, New York, 1955), p. 281
S.K. De, R.A. Gibbs, Synlett 16, 1231 (2005)
G.V.M. Sharma, J.J. Reddy, P.S. Lakshmi, P.R. Krishna, Tetrahedron Lett. 46, 6119 (2005)
M.K. Potdur, S.S. Mohile, M.M. Salunkhe, Tetrahedron Lett. 42, 9285 (2001)
Kandekar, A.C., Khadikar, B.M.: Synlett. 152 (2002)
Y. Gu, J. Zhang, Z. Duan, Y. Deng, Adv. Synth. Catal. 347, 512 (2005)
Hoz, A.D., Andres, M., Vazquez, E., Synlett. 602 (1999)
E.A. Gunnewegh, A.J. Hoefnegal, H. Van Bekkum, J. Mol. Catal. A: Chem. 100, 87 (1995)
S. Frere, V. Thiery, T. Besson, Tetrahedron Lett. 42, 2791 (2001)
T. Li, Z. Zhang, F. Yang, C. Fu, J. Chem. Res. 1, 38 (1998)
J.C. Rodriguez-Dominguez, G. Kirsch, Tetrahedron Lett. 47, 3279 (2006)
F.K. Esfahani, D. Zareyee, R. Yousefi, ChemCatChem 6, 3333 (2014)
B. Karimi, D. Zareyee, Org. Lett. 10, 3989 (2008)
A. Khalafi-Nezhad, S. Mowlazadeh Haghighi, F. Panahi, ACS Sustain Chem. Eng. 1, 1015 (2013)
D. Yuan, Q. Zhang, J. Dou, Catal. Commun. 11, 606 (2010)
Y. Kang, L. Zhou, X. Li, J. Yuvan, J. Mater. Chem 21, 3704 (2011)
S. Vahid Atghia, S. Sarvi Beigbaghlou, J. Nanostructure Chem. 3, 38 (2013)
R. Kizil, J. Irudayaraj, K. Seetharaman, J. Agric. Food Chem. 50, 3912 (2002)
F. M´arquez, T. Campo, M. Cotto, R. Polanco, R. Roque, P. Fierro, J.M. Sanz, E. Elizalde, C. Morant, Soft Nanosci. Lett. 1, 25 (2011)
R.L. Siqueira, I.V.P. Yoshida, L.C. Pardini, M.A. Schiavon, Mater. Res. 10, 147 (2007)
C. Wang, H. Daimon, S. Sun, Nano Lett. 9, 1493 (2009)
Acknowledgments
We are thankful to Islamic Azad University central Tehran branch for partial support of this work.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Samadizadeh, M., Nouri, S. & Kiani Moghadam, F. Magnetic nanoparticles functionalized ethane sulfonic acid (MNESA): as an efficient catalyst in the synthesis of coumarin derivatives using Pechmann condensation under mild condition. Res Chem Intermed 42, 6089–6103 (2016). https://doi.org/10.1007/s11164-016-2447-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11164-016-2447-5