Abstract
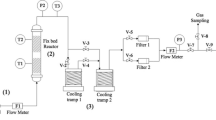
Borax and its calcined derivatives were used in pyrolytic valorization of tire tube rubber to produce high value oil and gases. This work involved the preparation and characterization of catalysts and optimization of the reaction parameters for the catalytic and non-catalytic pyrolysis of waste tire tube rubber. The derivatives of borax catalyst were prepared by calcining it at 600 °C, 800 °C and 1000 °C. The borax derivatives were labeled as borax-600, borax-800 and borax-1000 and used for the pyrolysis of waste rubber. The outcomes of the catalytic and thermal pyrolysis processes were compared to check the effectiveness of the borax derivatives as a catalyst. Both processes produced different quantities of oil, gas and char specifically in case of borax catalyzed and borax-1000 catalyzed reactions. The oil product of thermal pyrolysis was composed of 15 compounds while borax catalyzed pyrolysis produced oil with 14 compounds. On the other hand, all borax derivatives produced oil having nine compounds. The oil contained aliphatic and cyclic hydrocarbons along with small quantities of sulfur and phosphorous containing organic compounds. The results of distillation showed that thermal pyrolysis produced 83.80% volatile oil while borax catalyzed pyrolysis produced 83.84% volatile oil. The oil content decreased to 66.34% with an increase in borax calcination temperature. Similarly, thermal pyrolysis produced 12.56% residue while borax catalyzed reaction produced 16.10% residue. The residue decreased slightly with a rise in calcination temperature.




Similar content being viewed by others
Data availability
The data used in the paper is available from the corresponding author on request.
References
Nuzaimah M, Sapuan SM, Nadlene R, Jawaid M (2018) Recycling of waste rubber as fillers: a review. Mater Sci Eng 368:012016. https://doi.org/10.1088/1757-899X/368/1/012016
Ding K, Zhong Z, Zhang B, Wang J, Min A, Ruan R (2016) Catalytic pyrolysis of waste tire to produce valuable aromatic hydrocarbons: an analytical Py-GC/MS study. J Anal Appl Pyrolysis 122:55–63. https://doi.org/10.1016/j.jaap.2016.10.023
Rathsack P, Riedewald F, Sousa-Gallagher M (2015) Analysis of pyrolysis liquid obtained from whole tyre pyrolysis with molten zinc as the heat transfer media using comprehensive gas chromatography mass spectrometry. J Anal Appl Pyrolysis 116:49–57. https://doi.org/10.1016/j.jaap.2015.10.007
Yang Q, Yu S, Zhong H, Liu T, Yao E, Zhang Y, Zou H, Du W (2021) Gas products generation mechanism during co-pyrolysis of styrene–butadiene rubber and natural rubber. J Hazard Mater 401:123302. https://doi.org/10.1016/j.jhazmat.2020.123302
Milani G, Milani F (2018) Quasi-analytical kinetic model for natural rubber and polybutadiene rubber blends. Reac Kinet Mech Cat 123:351–365
Isahak WNRW, Hisham MW, Yarmo MA, Hin TYY (2012) A review on bio-oil production from biomass by using pyrolysis method. Renew Sustain Energy Rev 16(8):5910–5923. https://doi.org/10.1016/j.rser.2012.05.039
Luo S, Feng Y (2017) The production of fuel oil and combustible gas by catalytic pyrolysis of waste tire using waste heat of blast-furnace slag. Energy Convers Manag 136:27–35. https://doi.org/10.1016/j.enconman.2016.12.076
Duan D, Zhang Y, Wang Y, Lei H, Wang Q, Ruan R (2020) Production of renewable jet fuel and gasoline range hydrocarbons from catalytic pyrolysis of soap stock over corn cob-derived activated carbons. Energy 209:118454. https://doi.org/10.1016/j.energy.2020.118454
Shah J, Jan MR, Mabood F (2009) Recovery of value-added products from the catalytic pyrolysis of waste tyre. Energy Convers Manag 50(4):991–994. https://doi.org/10.1016/j.enconman.2008.12.017
Yu J, Liu S, Cardoso A, Han Y, Bikane K, Sun L (2019) Catalytic pyrolysis of rubbers and vulcanized rubbers using modified zeolites and mesoporous catalysts with Zn and Cu. Energy 188:116117. https://doi.org/10.1016/j.energy.2019.116117
Qu B, Li A, Qu Y, Wang T, Zhang Y, Wang X, Gao Y, Fu W, Ji G (2020) Kinetic analysis of waste tire pyrolysis with metal oxide and zeolitic catalysts. J Anal Appl Pyrolysis 152:104949. https://doi.org/10.1016/j.jaap.2020.104949
Hijazi A, Boyadjian C, Ahmad MN, Zeaiter J (2018) Solar pyrolysis of waste rubber tires using photoactive catalysts. Waste Manag 77:10–21. https://doi.org/10.1016/j.wasman.2018.04.044
Singh MV (2020) Conversions of waste tube-tyres (WTT) and waste polypropylene (WPP) into diesel fuel through catalytic pyrolysis using base SrCO3. Eng Sci 13:87–97. https://doi.org/10.30919/es8d1158
Demirbas A, Al-Sasi BO, Nizami AS (2016) Conversion of waste tires to liquid products via sodium carbonate catalytic pyrolysis. Energy Source A 38(16):2487–2493. https://doi.org/10.1080/15567036.2015.1052598
Wang J, Jiang J, Wang X, Liu P, Li J, Liu G, Wang K, Li M, Zhong Z, Xu J, Ragauskas AJ (2019) Catalytic conversion of rubber wastes to produce aromatic hydrocarbons over USY zeolites: effect of SiO2/Al2O3 mole ratio. Energy Convers Manag 197:111857. https://doi.org/10.1016/j.enconman.2019.111857
Salmasi SSZ, Abbas-Abadi MS, Haghighi MN, Abedini H (2015) The effect of different zeolite based catalysts on the pyrolysis of poly butadiene rubber. Fuel 160:544–548. https://doi.org/10.1016/j.fuel.2015.07.091
Elordi G, Olazar M, Aguado R, Lopez G, Arabiourrutia M, Bilbao J (2007) Catalytic pyrolysis of high density polyethylene in a conical spouted bed reactor. J Anal Appl Pyrolysis 79(1–2):450–455. https://doi.org/10.1016/j.jaap.2006.11.010
Tekin K, Akalın MK, Bektaş S, Karagöz S (2013) Hydrothermal wood processing using borax decahydrate and sodium borohydride. J Anal Appl Pyrolysis 104:68–72. https://doi.org/10.1016/j.jaap.2013.09.008
Yüceda E, Durak H (2019) Bio-oil and bio-char from Lactuca serriola: significance of catalyst and temperature for assessing yield and quality of pyrolysis. Energy Source A. https://doi.org/10.1080/15567036.2019.1645765
Wang Z, Qu L, Qian J, He Z, Yi S (2019) Effects of the ultrasound-assisted pretreatments using borax and sodium hydroxide on the physicochemical properties of Chinese fir. Ultrason Sonochem 50:200–207. https://doi.org/10.1016/j.ultsonch.2018.09.017
Durak H (2016) Pyrolysis of Xanthium strumarium in a fixed bed reactor: effects of boron catalysts and pyrolysis parameters on product yields and character. Energy Source A 38(10):1400–1409. https://doi.org/10.1080/15567036.2014.947446
Xu Q, Ma X, Yu Z, Cai Z (2014) A kinetic study on the effects of alkaline earth and alkali metal compounds for catalytic pyrolysis of microalgae using thermogravimetry. Appl Therm Eng 73(1):357–361. https://doi.org/10.1016/j.applthermaleng.2014.07.068
Jeong K, Jeong HJ, Lee G, Kim SH, Kim KH, Yoo CG (2020) Catalytic effect of alkali and alkaline earth metals in lignin pyrolysis: a density functional theory study. Energy Fuels 34(8):9734–9740. https://doi.org/10.1021/acs.energyfuels.0c01897
Giudicianni P, Gargiulo V, Grottola CM, Alfè M, Ragucci R (2018) Effect of alkali metal ions presence on the products of xylan steam assisted slow pyrolysis. Fuel 216:36–43. https://doi.org/10.1016/j.fuel.2017.11.150
Waclawska I (1995) Thermal decomposition of borax. J Therm Anal Calorim 43(1):261–269. https://doi.org/10.1007/BF02635993
He L, Szopinski D, Wu Y, Luinstra GA, Theato P (2015) Toward self-healing hydrogels using one-pot thiol–ene click and borax-diol chemistry. ACS Macro Lett 4(7):673–678. https://doi.org/10.1021/acsmacrolett.5b00336
Molla A, Hussain S (2014) Borax catalyzed domino reactions: synthesis of highly functionalised pyridines, dienes, anilines and dihydropyrano [3, 2-c] chromenes. RSC Adv 4(56):29750–29758. https://doi.org/10.1039/C4RA03627A
Sahin O, Bulutcu AN (2003) Evaluation of thermal decomposition kinetics of borax pentahydrate using genetic algorithm method by isothermal analysis. Turk J Chem 27:197–207
Mkhize NM, Van der Gryp P, Danon B, Görgens JF (2016) Effect of temperature and heating rate on limonene production from waste tyre pyrolysis. J Anal Appl Pyrolysis 120:314–320. https://doi.org/10.1016/j.jaap.2016.04.019
Barbooti MM, Mohamed TJ, Hussain AA, Abas FO (2014) Optimization of pyrolysis conditions of scrap tires under inert gas atmosphere. J Anal Appl Pyrolysis 72(1):165–170. https://doi.org/10.1016/j.jaap.2004.05.001
Miranda M, Pinto F, Gulyurtlu I, Cabrita I (2013) Pyrolysis of rubber tyre wastes: a kinetic study. Fuel 103:542–552. https://doi.org/10.1016/j.fuel.2012.06.114
Shaaban A, Se SM, Dimin MF, Juoi JM, Husin MHM, Mitan NMM (2014) Influence of heating temperature and holding time on biochars derived from rubber wood sawdust via slow pyrolysis. J Anal Appl Pyrolysis 107:31–39. https://doi.org/10.1016/j.jaap.2014.01.021
Inguanzo M, Domınguez A, Menéndez JA, Blanco CG, Pis JJ (2002) On the pyrolysis of sewage sludge: the influence of pyrolysis conditions on solid, liquid and gas fractions. J Anal Appl Pyrolysis 63(1):209–222. https://doi.org/10.1016/S0165-2370(01)00155-3
Mohamed BA, Ellis N, Kim CS, Bi X (2019) Microwave-assisted catalytic biomass pyrolysis: effects of catalyst mixtures. Appl Catal B 253:226–234. https://doi.org/10.1016/j.apcatb.2019.04.058
López A, De Marco I, Caballero BM, Laresgoiti MF, Adrados A, Aranzabal A (2011) Catalytic pyrolysis of plastic wastes with two different types of catalysts: ZSM-5 zeolite and Red Mud. Appl Catal B 104(3–4):211–219. https://doi.org/10.1016/j.apcatb.2011.03.030
Acknowledgements
The authors would also like to acknowledge the efforts of King Khalid University, Saudi Arabia (Deanship of Scientific Research) for support through the Research Groups Project under the Grant Number (R.G.P.2/169/42).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Authors declare no conflict of interest regarding publication of this article.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Khan, A., Hussain, Z., Hussain, K. et al. Evaluation of the catalytic activity of borax and its calcined derivatives for pyrolytic valorization of waste tire tube rubber for production of oil and gases. Reac Kinet Mech Cat 134, 883–901 (2021). https://doi.org/10.1007/s11144-021-02108-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11144-021-02108-3