Abstract
Purpose
System science offers a unique set of tools, including causal loop diagrams (CLDs), for stakeholders to better grasp the complexity of factors surrounding quality of life. Because the health-related quality of life (HRQoL) of cancer immunotherapy patients exists within an intricate system affected by and affecting many factors across multiple dimensions, the development of a systems-level model can provide a powerful framework to aid the understanding of this complexity. We developed a CLD for HRQoL of cancer immunotherapy patients.
Methods
We first applied a literature-based approach to construct a CLD for patients following immunotherapy. We then iteratively reviewed and enhanced the CLD through interviews with subject matter experts.
Results
Based on the reviewed literature and subject matter expert input, we produced a CLD representing the system surrounding cancer immunotherapy patients’ HRQoL. Several feedback loops are identified that span clinical experiences, oncology teams’ perceptions about immunotherapy, social support structures, and further research and development in cancer immunotherapy, in addition to other components. The CLD enables visualization of thought experiments regarding how a change anywhere in the system can ultimately worsen or improve patients’ HRQoL.
Conclusion
The CLD illustrates the valuable contribution of a systems perspective to quality-of-life research. This systems-based qualitative representation gives insight on strategies to inhibit harmful effects, enhance beneficial effects, and inherent tradeoffs within the system. The CLD identifies gaps in the literature and offers a communication tool for diverse stakeholders. Our research method provides an example for studying the complexities of quality of life in other health domains.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
System science tools such as causal loop diagrams (CLDs) offer a powerful framework to understand the complex interplay of factors influencing a phenomenon of interest [1]. While they have been widely used in medicine and public health [2,3,4,5], their applications in studying quality of life has thus far been limited, e.g., [6]. We demonstrate the usefulness of CLDs in a case study of applying the framework to the system surrounding health-related quality of life in cancer immunotherapy, particularly by integrating findings from prior research with insights from experts. While this report focuses on the case of cancer immunotherapy, it provides an example of expanding the application of CLDs to analyzing the underlying dynamics of quality of life in other health problems. The cancer immunotherapy clinical setting is rapidly evolving and linked with external influences [7]. Strategies to improve outcomes, and which may be mentally modeled using the CLD, include but are not limited to: new treatment options; digital tools; remote monitoring; patients’ choices and contributions; clinical trials; treatment delivered at home; and patients’ advocacy groups.
Immunotherapy is defined generally as medicinal strategies that harness the immune system to prevent or treat disease [8]. Immunotherapy used in the context of modern cancer care has been developed to stimulate and enhance the immune system’s response to tumors [9]. Before the introduction of immunotherapy, options for treating cancer fell into four main categories: radiotherapy, chemotherapy, surgery, and other targeted treatments [9]. Immunotherapy is thus often cited as the fifth pillar of cancer care [10].
Immunotherapy applied as cancer treatment includes the following broad classes: cell-based immunotherapies, immunomodulators, vaccines, antibody-based targeted therapies, and oncolytic viruses [11]. Ipilimumab, an immune checkpoint inhibitor targeting CTLA4 was first approved by European Medicines Agency (EMA) and the U.S. Food and Drug Administration (FDA) in 2011, representing the first major modern immunotherapy treatment for cancer [12, 13]. Immunotherapies for cancer have since received approvals for use in patients with multiple types of cancer. Immunotherapy cancer treatment represents a major innovation in the field of medicine, and has been shown to offer patients favorable outcomes in terms of clinical measures such as overall survival, progression-free survival, and tumor size reduction [14, 15]. Nivolumab and pembrolizumab, anti-programmed death 1 (PD-1) checkpoint inhibitors, were approved by the FDA in 2015. These two checkpoint inhibitors have played a major role in cancer immunotherapy, as they have shown great success in treating melanoma, lymphoma, and non-small cell lung cancer [16] and have demonstrated improvements to health-related quality of life (HRQoL) [17].
Defining health outcomes with purely objective clinical measures such as blood test results, tumor size, and prognosis (i.e., expected survival) overlooks many of the important elements that characterize overall patient well-being. The concept of HRQoL, however, attends to important but non-clinical aspects of health such as social and emotional wellness to measure patients’ overall well-being. A few definitions for HRQoL exist in the literature; the primary one we will employ is “how well a person functions in their life and his or her perceived well-being in physical, mental, and social domains of health” [18]. Accounting for HRQoL is critical in the evaluation of cancer patients’ health because they exhibit many diverse symptoms, including losses of functional ability that are not observable with a laboratory test or imaging procedures [19]. HRQoL outcomes measures are invaluable inputs to guide shared clinical decision-making on choices of therapy [20].
Despite the importance of HRQoL of patients following immunotherapy, it has only received some limited, recent research attention [21, 22]. One noticeable gap is that less HRQoL-focused research has been performed regarding cancer immunotherapy purely due to the treatment’s relative novelty. Research has focused instead primarily on establishing clinical safety and effectiveness, and attention to patient-reported outcomes used to measure HRQoL is under-explored. The current HRQoL research in cancer immunotherapy tends to focus on primary data collection that produces analyses of specific clinical trial observations in terms of a quality-of-life outcome [23], the symptoms and determinants of HRQoL in cancer immunotherapy patients [24], and tools used to measure HRQoL [19], which are actively evolving to suit the specific needs of the population. Further, existing HRQoL instruments were developed mainly for chemotherapy outcomes and not specifically tailored to measure immunotherapy outcomes [25]. Chemotherapy is distinct from immunotherapy as it directly targets tumors, while immunotherapy treats patients by acting on their immune system and can boost the immune response to teach the immune system how to identify and destroy cancer cells. Acute and delayed toxicities dramatically differ between chemo- and immunotherapy [26]. The way that cancer immunotherapy patients’ HRQoL is analyzed in the literature is incomplete in terms of inattention to dynamic interconnections among various individual and social determinants within a broader system. A qualitative systemic understanding can offer practical advantages by generating insight about how factors interconnect and enabling proactive, impactful decision-making.
To address this research gap, we set an objective to develop a CLD that identifies the system surrounding this patient group’s HRQoL qualitatively by mapping out factors that interact around HRQoL following cancer immunotherapy.
Methods
The research method consists of: (1) a review of the literature to develop the initial CLD, and (2) interviews with clinical experts to enhance and finalize the CLD. These two steps are elaborated below.
A semi-systematic review approach was employed to gather variables related to HRQoL and their potential interactions. A search in PubMed in August of 2020 applied the query (((immunotherapy[Title/Abstract]) OR ("cancer treatment"[Title/Abstract])) OR ("cancer patient*"[Title/Abstract])) AND ("quality of life"[Title/Abstract]). The search strategy was intentionally broad to capture any research that focused on quality of life with respect to cancer treatment. Results were sorted by most to least relevant and research assistants independently checked article titles and abstracts to verify overall relevancy and collected variables connected to cancer patients’ quality of life from the articles’ full text. The research assistants are undergraduate students who are knowledgeable in the fundamentals of public health and system dynamics. Initial findings were reviewed by a third researcher to calibrate understanding before proceeding with the variable extraction process. We identified potential factors relevant to HRQoL until we observed a saturation point of factors already extracted, such that continuing to extract information from the sorted list of studies was no longer producing unique new variables. Then, we explored the literature-based evidence supporting causal relationships.
Once we had constructed an internal first draft of the CLD based upon literature, we conducted semi-structured interviews with eight subject matter experts knowledgeable in cancer immunotherapy treatment. All experts were medical doctors and their expertise spans a wide range of topics, including but not limited to epidemiology, gastrointestinal oncology, melanoma, hemato-oncology, CART-cell therapy, and immune checkpoint inhibitors. Countries represented by experts include France, Netherlands, Portugal, Spain, U.K., and U.S. In each interview, we presented the CLD, explained the meaning of each variable, and walked through each feedback loop step by step. We then asked if the representation correlated with their experience and understanding of the system, whether any variable represented was not relevant, and if any relevant variables were absent. Finally, we asked if the links (arrows) and polarities (± signs on the arrows) were consistent with their belief about causality. From this structured point, the conversations would vary depending on the feedback and we would ask follow-up questions depending on specific points brought up by the expert. We applied an iterative approach by implementing feedback to the CLD draft after each interview and presenting the revised version to the next expert. The CLD version following the final interview was returned to the interviewees for additional feedback. These interviews provide essential inputs in our analysis because: (1) individual studies might not take a broad and systematic perspective and thus study only a subset of factors or connections among factors; (2) limited research in this space may overlook important factors. The CLD is thus reflective of evidence found in the literature as well as critical inputs from all eight experts interviewed.
The CLD includes reinforcing (R) and balancing (B) feedback loops. In a reinforcing feedback loop, an increase (or decrease) in one variable feeds through effects in other variables represented in the loop, and through those mechanisms ends up increasing (or decreasing) the initial variable even more. Meanwhile in a balancing loop, an increase (or decrease) in one variable produces feedback effects which decrease (or increase) the starting variable. In other words, reinforcing loops cause changes in the same direction as the initial variable change direction, and balancing loops cause changes in the inverse direction as the initial variable change direction.
Results
Forty articles were reviewed. The extracted factors that potentially influence HRQoL are reported in Table 1. Three articles ([27,28,29]) contributed an initial basis of many factors and additional articles often supported relevance of the same variables. Additional factors identified through interviews are also reported in Table 1.
Feedback loops
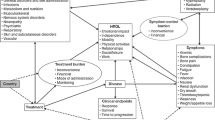
We highlight six reinforcing feedback loops and two balancing feedback loops in Fig. 1. We describe these feedback effects briefly below.
Reinforcing loop R1
Consider reinforcing loop R1. The oncology team’s perceived effectiveness represents the clinicians’ overall impression about the quality of treatment and confidence that the treatment option is the option most likely to produce clinical success for their patients. Perceived effectiveness directly impacts oncology team’s likelihood to recommend immunotherapy. Recommendation by the patient’s immediate team is a foremost deciding factor in the patients’ decision to undergo the treatment and results in receipt of immunotherapy. As shown in effectiveness studies, patients’ HRQoL improves as a result of the receipt of immunotherapy such that this central variable increases in the responding patients [63]. In aggregate over time, patients’ positive experience in terms of improved HRQoL flows up to community-level perceptions about the treatment thus elevates again the first variable: oncology team’s perceived effectiveness.
Reinforcing loop R2
Reinforcing loop R2 begins with a change in the volume of patients receiving immunotherapy (receipt of immunotherapy). For example, this might increase due to new favorable clinical evidence. The increase signals the market and research community to focus resources on research and development activities in cancer immunotherapy, offering further data and refining evidence that helps build confidence in oncologists’ treatment decisions.
Reinforcing loop R3
Another reinforcing mechanism relates to the link between patients’ HRQoL and social aspects of health. Consider when HRQoL decreases due to, for instance, adverse side effects occurring during treatment. Social connection then decreases due to factors such as feeling of alienation (i.e., a shift in identify as a cancer patient as being different and separate from the healthy population that can lead to self-isolation) and a decreased ability to engage in normal work and leisure activities. When social connection decreases, social support may also decrease by way of stigma experienced by patients against communicating the need for support. Decreased social support begets lower psychological well-being and thus an overall decrease in HRQoL. See the appendix figure for a more detailed view of the mechanisms in this loop.
Reinforcing loops R4 and R5
To understand reinforcing loops R4 and R5, consider an increase in incentives to invest in immunotherapy R&D. Over time, investment in research activities produce improvements in the effectiveness of immunotherapy treatment, leading to improved physical well-being (via R4) and psychological well-being (via R5) for immunotherapy patients. When either or both aspects of well-being increase, so does patients’ health-related quality of life. The remaining pieces of the loop—oncology team’s perceived effectiveness through incentives to invest in immunotherapy R&D—have already been described above within loops R1 and R2.
Reinforcing loop R6
R6 illustrates how advances in effectiveness of immunotherapy treatment produced by research activities (incentives to invest in immunotherapy R&D) directly affects the oncology team’s perceived effectiveness of the treatment. Again, the remaining pieces of the loop (oncology team’s perceived effectiveness through incentives to invest in immunotherapy R&D) have been described above. The link between effectiveness and perceived effectiveness represents an independent reinforcing feedback effect.
Balancing loops B1 and B2
Within balancing loops B1 and B2, receipt of immunotherapy often results in adverse side effects due to immunotherapy. Adverse side effects have detrimental effects on both physical well-being (B1) and psychological well-being (B2) which negatively impact the central variable patients’ health-related quality of life. When the central variable decreases, oncology team’s perceived effectiveness also decreases, as does the oncology team’s likelihood to recommend immunotherapy. Lower likelihood for the oncologists to recommend immunotherapy to patients decreases receipt of immunotherapy.
Discussion
The CLD provides a systematic perspective and offers an aid to explain phenomena that have the potential to bolster or deteriorate HRQoL. The literature and expert-based approach allowed identification of a complex interplay of factors with multiple feedback mechanisms present in the system. A wide set of variables was identified. Mechanisms at play in the system ranged from factors at the individual patient level up to dynamics operating at the greater community level. The expanded version of the diagram, shown in the supplement as Figure S-1, delineates these levels and presents a discussion of their components.
The manner that component parts relate to one another and not only how they act separately produces the overall system behavior. A shift within just one feedback loop has the potential to alter the resulting levels of variables throughout the system. Feedback mechanisms thus act as important leverage points. We use the diagram to detect patterns in behavior within this system and identify opportunities to disrupt harmful feedback mechanisms that reduce patients’ HRQoL, strengthen feedback mechanisms that operate to improve patients’ HRQoL, and understand systemic tradeoffs.
Disrupting harmful effects
Consider first feedback mechanisms that have potentially severe detrimental effects if left unchecked. R3 in Fig. 1 shows an example. For HRQoL to thrive in the system, psychological well-being must be upheld. Social support is a direct determinant of psychological well-being, which is in jeopardy of deteriorating precipitously should social connection drop to activate a negative reinforcing mechanism. The loop can produce a downward spiral and severely worsen HRQoL. The strength of exogenous variables influencing R3 component variables such as quality and quantity of social support structures and availability of resources for social support e.g., patient support groups/smart apps serve as proactive protectors inhibitive of the potentially harmful effects.
COVID-19 effects are not represented in the diagram, as it was designed as a generic representation not specific to particular years. However, the pandemic is relevant to mention as it pertains to the dynamics discussed above. Cancer patients, in particular patients receiving some types of immunotherapy such as CD19-directed chimeric antigen receptor T-cells [64, 65], are highly vulnerable to SARS-CoV-2 infection and development of severe symptoms [66]. They have thus been socially distancing in extreme measure in recent years, making the social support structures in place for immunotherapy patients all the more important in light of the dynamics highlighted by the CLD.
Enhancing beneficial effects
Here we identify a beneficial feedback cycle scenario. Notice the R1 and R2 loops in Fig. 1, which consist of components described in the above section. Patients’ receipt of immunotherapy connects to the variable for investment in research and development. The research activities produce evidence that flows into oncology team’s likelihood to recommend immunotherapy and back into the individual patient level (receipt of immunotherapy) which then bolsters HRQoL.
The implication is a need to enable amplification of this cycle while maintaining integrity in the evidence upon which decisions are based. Opportunities to amplify R2 include direct investment in research by public and/or private sectors and timely inclusion of evidence-based findings pertaining to immunotherapy treatments where appropriate.
Awareness of tradeoffs
When receipt of immunotherapy increases, the change creates opposing effects within the system via balancing loops (B1 and B2) and reinforcing loops (R1 and R2). The dueling effects triggered by an increase in receipt of immunotherapy highlights a tradeoff within the system. More patients receiving immunotherapy will at once activate mechanisms that would push it in the direction of increasing even more through the reinforcing loops. At the same time, it also activates a balancing effect that pulls receipt of immunotherapy downward. The dynamics are described in greater detail in the appendix.
The effects that will win over directionally in terms of the net effect on receipt of immunotherapy and ultimately on patients’ health-related quality of life depend on the systemic timing and strength of flows between variables. Circumstances that produce a net positive effect cannot be answered with certainty in our qualitative representation. The tradeoff dynamics noticeable in the CLD would be a direction for investigation of a future quantified model. However, in the CLD we can still hypothesize strategies to mitigate the potentially harmful balancing loop effects and stack the deck in favor of a net positive effect on HRQoL. One such strategy would be strengthening the immediate connections level variable effective management of side effects by health care system. Managing side effects better has the potential to interrupt B1 by mitigating the harmful effects from treatment side effects on physical well-being, psychological well-being, and patients’ health-related quality of life. In addition to management, it is also equally important for health care systems to better scrutinize and report potential side effects.
Limitations
Among limitations to this study, we identify the two we consider most critical in the context of HRQoL systems science. First, the process of constructing the CLD has been dependent on limited published research of HRQoL in patients receiving the relatively novel treatment of cancer immunotherapy. Current research on this topic is active but does not yet fully characterize all aspects of the CLD. Second, the CLD is limited by the perspectives of eight subject matter experts. While each interview was extensive and detailed, and experts’ feedback contributed insight from a diverse set of experiences related to cancer immunotherapy treatment, time and availability constraints limited in the number of interview subjects.
Despite these limitations, the qualitative CLD helps highlight the complexity of the systems of various factors that may affect and be affected by HRQoL following immunotherapy. The flow of relationships among factors allows visualization of feedback mechanisms that are intuitive when laid out in the diagram yet not obvious when thinking about many factors abstractly, i.e., without a visual aid. Additionally, the CLD offers a powerful communication tool that allows expansion of the scope of consideration to a diverse set of policymakers and stakeholders involved in immunotherapy care. It can act as a vehicle to simplify understanding of the complexity so that separate actors, who are sometimes confined to just one part of the problem, can better collaborate to achieve the common goal of improved outcomes.
This work might be used in the future to create quantified simulation models. Such models would then estimate and project the impact of interventions on specific outcomes of interest. They would also enable comparisons of outcomes resulting from a portfolio of interventions and allow decision-makers to optimally allocate resources. Availability of data will determine how the development of such models can proceed. Changes in HRQoL can be quantified using patient-reported outcomes (PRO) instruments. PRO instruments frequently used in cancer research include the generic EQ-5D and the cancer-specific EORTC QLQ-C30 questionnaires [67]. Novel, immunotherapy-specific instruments are also an area of active development among ongoing efforts continue to collect HRQoL data from patients, including in our large study in several European countries; see the project website for more information [68].
References
Sterman, J. D. (2006). Learning from evidence in a complex world. American Journal of Public Health, 96(3), 505–514. https://doi.org/10.2105/ajph.2005.066043
Darabi, N., & Hosseinichimeh, N. (2020). System dynamics modeling in health and medicine: A systematic literature review. System Dynamics Review, 36(1), 29–73. https://doi.org/10.1002/sdr.1646
Jalali, M. S., & Kaiser, J. P. (2018). Cybersecurity in hospitals: A systematic, organizational perspective. Journal of Medicine and Internet Research, 20(5), 159. https://doi.org/10.2196/10059
Jalali, M. S., Rahmandad, H., Bullock, S. L., & Ammerman, A. (2017). Dynamics of implementation and maintenance of organizational health interventions. International Journal of Environmental Research and Public Health, 14(8), 917.
Sajid, Z., Andersen, M., & Ottesen, J. T. (2020). System dynamics of cancer in erythropoiesis with multiple EPO feedbacks. System Dynamics Review, 36(4), 447–466. https://doi.org/10.1002/sdr.1670
Lounsbury, D. W., Hirsch, G. B., Vega, C., & Schwartz, C. E. (2014). Understanding social forces involved in diabetes outcomes: A systems science approach to quality-of-life research. Quality of Life Research, 23(3), 959–969. https://doi.org/10.1007/s11136-013-0532-4.
Emens, L. A., et al. (2017). Cancer immunotherapy: Opportunities and challenges in the rapidly evolving clinical landscape. European Journal of Cancer, 81, 116–129. https://doi.org/10.1016/j.ejca.2017.01.035
Dobosz, P., & Dzieciątkowski, T. (2019). The intriguing history of cancer immunotherapy. Frontiers in Immunology, 10, 2965–2965. https://doi.org/10.3389/fimmu.2019.02965
Kaufman, H. L., et al. (2019). The promise of Immuno-oncology: Implications for defining the value of cancer treatment. Journal for Immunotherapy of Cancer, 7(1), 129–129. https://doi.org/10.1186/s40425-019-0594-0
Oiseth, S. J., & Aziz, M. S. (2017). Cancer immunotherapy: A brief review of the history, possibilities, and challenges ahead. Journal of Cancer Metastasis and Treatment, 3, 250–261. https://doi.org/10.20517/2394-4722.2017.41
Cancer Research Institute. "Immunotherapy Treatment Types. Retrieved March 5, 2021 from https://www.cancerresearch.org/immunotherapy/treatment-types.
Cameron, F., Whiteside, G., & Perry, C. (2011). Ipilimumab: First global approval,. Drugs, 71(8), 1093–1104. https://doi.org/10.2165/11594010-000000000-00000
Eno, J. (2017). Immunotherapy through the years. Journal of Advanced Practise Oncology, 8(7), 747–753.
Ventola, C. L. (2017). Cancer Immunotherapy, Part 2: Efficacy, Safety, and Other Clinical Considerations. P T, 42(7), 452–463.
Anagnostou, V., et al. (2017). Immuno-oncology Trial Endpoints: Capturing Clinically Meaningful Activity. Clinical Cancer Research, 23(17), 4959. https://doi.org/10.1158/1078-0432.CCR-16-3065
Morgensztern, D., & Herbst, R. S. (2016). Nivolumab and pembrolizumab for non-small cell lung cancer. Clinical Cancer Research, 22(15), 3713. https://doi.org/10.1158/1078-0432.CCR-15-2998
Abdel-Rahman, O., Oweira, H., & Giryes, A. (2018). Health-related quality of life in cancer patients treated with PD-(L)1 inhibitors: A systematic review. Expert Review of Anticancer Therapy, 18(12), 1231–1239. https://doi.org/10.1080/14737140.2018.1528146
Karimi, M., & Brazier, J. (2016). Health, health-related quality of life, and quality of life: what is the difference? PharmacoEconomics, 34(7), 645–649. https://doi.org/10.1007/s40273-016-0389-9
Osoba, D. (2011). Health-related quality of life and cancer clinical trials. Ther Adv Med Oncol, 3(2), 57–71. https://doi.org/10.1177/1758834010395342
Committee on Improving the Quality of Cancer Care: Addressing the Challenges of an Aging Population, Board on Health Care Services, and Institute of Medicine, "Patient-Centered Communication and Shared Decision Making," in Delivering High-Quality Cancer Care: Charting a New Course for a System in Crisis., L. Levit, E. Balogh, S. Nass, and P. A. Ganz Eds. Washington (DC): National Academies Press, 2013.
Taarnhøj, G. A., Johansen, C., Lindberg, H., Basch, E., Dueck, A., & Pappot, H. (2020). Patient reported symptoms associated with quality of life during chemo- or immunotherapy for bladder cancer patients with advanced disease. Cancer Medicine, 9(9), 3078–3087. https://doi.org/10.1002/cam4.2958
Voon, P. J., Cella, D., & Hansen, A. R. (2021). Health-related quality-of-life assessment of patients with solid tumors on immuno-oncology therapies. Cancer. https://doi.org/10.1002/cncr.33457
O’Reilly, A., et al. (2020). An immunotherapy survivor population: health-related quality of life and toxicity in patients with metastatic melanoma treated with immune checkpoint inhibitors. Supportive Care in Cancer, 28(2), 561–570. https://doi.org/10.1007/s00520-019-04818-w
Lluncor, M., et al. (2018). Determinants of health related quality of life (HRQoL) in adult patients with hereditary angioedema due to C1 inhibitor deficiency (C1-INH-HAE). Journal of Allergy and Clinical Immunology, 141(2), 266. https://doi.org/10.1016/j.jaci.2017.12.845
van Roij, J., Fransen, H., van de Poll-Franse, L., Zijlstra, M., & Raijmakers, N. (2018). Measuring health-related quality of life in patients with advanced cancer: a systematic review of self-administered measurement instruments. Quality of Life Research, 27(8), 1937–1955. https://doi.org/10.1007/s11136-018-1809-4
Puzanov, I., et al. (2017). Managing toxicities associated with immune checkpoint inhibitors: consensus recommendations from the Society for Immunotherapy of Cancer (SITC) Toxicity Management Working Group. Journal of Immunotherapy Cancer, 5(1), 95. https://doi.org/10.1186/s40425-017-0300-z
Ashing-Giwa, K. T. (2005). The contextual model of HRQoL: A paradigm for expanding the HRQoL framework). Quality of Life Research, 14(2), 297–307. https://doi.org/10.1007/s11136-004-0729-7
Donovan, K., Sanson-Fisher, R. W., & Redman, S. (1989). Measuring quality of life in cancer patients. Journal of Clinical Oncology, 7(7), 959–968. https://doi.org/10.1200/JCO.1989.7.7.959
Kagawa-Singer, M., Padilla, G. V., & Ashing-Giwa, K. (2010). Health-related quality of life and culture. Seminars in Oncology Nursing, 26(1), 59–67. https://doi.org/10.1016/j.soncn.2009.11.008
Taghian, N. R., Miller, C. L., Jammallo, L. S., O’Toole, J., & Skolny, M. N. (2014). Lymphedema following breast cancer treatment and impact on quality of life: A review. Critical Reviews in Oncology Hematology, 92(3), 227–234. https://doi.org/10.1016/j.critrevonc.2014.06.004
P. F. Nascimento et al., "Women's occupational performance and quality of life during breast cancer treatment," (in eng), BMJ Support Palliat Care, Jul 28 2019, doi: https://doi.org/10.1136/bmjspcare-2019-001788.
Ringdal, G. I., & Ringdal, K. (2000). A follow-up study of the quality of life in cancer patients with different prognoses. Quality of Life Research, 9(1), 65–73. https://doi.org/10.1023/A:1008937724943
Ung, B., Gupta, S., Parikh, K., Liebert, R., Abouzaid, S., & Rosenberg, A. S. (2017). Adherence, quality of life, health care resource use, and functional impairment among patients with oral multiple myeloma (MM) treatments. Journal of Clinical Oncology, 35(15), e19519. https://doi.org/10.1200/JCO.2017.35.15_suppl.e19519
Pfaendler, K. S., Wenzel, L., Mechanic, M. B., & Penner, K. R. (2015). Cervical cancer survivorship: Long-term quality of life and social support. Clinical Therapeutics, 37(1), 39–48. https://doi.org/10.1016/j.clinthera.2014.11.013
Ashing-Giwa, K. T., & Lim, J.-W. (2008). Predicting health-related quality of life: Testing the contextual model using structural equation modeling. Applied Research in Quality of Life, 3(3), 215–230. https://doi.org/10.1007/s11482-009-9057-y
Tyson, M. D., 2nd., & Barocas, D. A. (2018). Quality of life after radical cystectomy. Urologic Clinics of North America, 45(2), 249–256. https://doi.org/10.1016/j.ucl.2017.12.008
Heber, D., & Li, Z. (2016). Nutrition intervention in cancer. Medical Clinics of North America, 100(6), 1329–1340. https://doi.org/10.1016/j.mcna.2016.06.011
Villa, A., & Akintoye, S. O. (2018). Dental management of patients who have undergone oral cancer therapy. Dental Clinics of North America, 62(1), 131–142. https://doi.org/10.1016/j.cden.2017.08.010
Finck, C., Barradas, S., Zenger, M., & Hinz, A. (2018). Quality of life in breast cancer patients: Associations with optimism and social support. International Journal of Clinical Health Psychology, 18(1), 27–34. https://doi.org/10.1016/j.ijchp.2017.11.002
Joseph, R. W., et al. (2020). Health-related quality of life (QoL) in patients with advanced melanoma receiving immunotherapies in real-world clinical practice settings. Quality of Life Research, 29(10), 2651–2660. https://doi.org/10.1007/s11136-020-02520-7
Steensma, D. P., et al. (2014). Disparity in perceptions of disease characteristics, treatment effectiveness, and factors influencing treatment adherence between physicians and patients with myelodysplastic syndromes. Cancer, 120(11), 1670–1676. https://doi.org/10.1002/cncr.28631
Hampson, L. A., Cowan, J. E., Zhao, S., Carroll, P. R., & Cooperberg, M. R. (2015). Impact of age on quality-of-life outcomes after treatment for localized prostate cancer. European Urology, 68(3), 480–486. https://doi.org/10.1016/j.eururo.2015.01.008
Stone, J. B., & DeAngelis, L. M. (2016). Cancer-treatment-induced neurotoxicity—focus on newer treatments. Nature Reviews Clinical Oncology, 13(2), 92–105. https://doi.org/10.1038/nrclinonc.2015.152
Livingstone, A., Agarwal, A., Stockler, M. R., Menzies, A. M., Howard, K., & Morton, R. L. (2020). Preferences for immunotherapy in melanoma: A systematic review. Annals of Surgical Oncology, 27(2), 571–584. https://doi.org/10.1245/s10434-019-07963-y
Wilson, M. K., Karakasis, K., & Oza, A. M. (2015). Outcomes and endpoints in trials of cancer treatment: The past, present, and future). The lancet Oncology, 16(1), e32-42. https://doi.org/10.1016/s1470-2045(14)70375-4
Dilla, T., et al. (2015). Do new cancer drugs offer good value for money? The perspectives of oncologists, health care policy makers, patients, and the general population. Patient Preference and Adherence, 10, 1–7. https://doi.org/10.2147/PPA.S93760
Mu, K. (2019) Study on the influencing factors of quality of life in postoperative patients with breast cancer. In 2019 10th International Conference on Information Technology in Medicine and Education (ITME) (pp. 217–221). https://doi.org/10.1109/ITME.2019.00056
Biglia, N., Zanfagnin, V., Daniele, A., Robba, E., & Bounous, V. E. (2017). Lower body lymphedema in patients with gynecologic cancer. Anticancer Research, 37(8), 4005–4015. https://doi.org/10.21873/anticanres.11785
Feuerstein, M., et al. (2010). Work in cancer survivors: a model for practice and research. Journal of Cancer Survivorship, 4(4), 415–437. https://doi.org/10.1007/s11764-010-0154-6
Henry, D. H., Viswanathan, H. N., Elkin, E. P., Traina, S., Wade, S., & Cella, D. (2008). Symptoms and treatment burden associated with cancer treatment: results from a cross-sectional national survey in the U.S. Supportive Care Cancer, 16(7), 791–801. https://doi.org/10.1007/s00520-007-0380-2
Kane, D., Rajacich, D., & Andary, C. (2020). Experiences of cancer patients’ return to work. Canadian Oncology Nursing Journal, 30(2), 113–118. https://doi.org/10.5737/23688076302113118
Karanth, S., et al. (2019). Race, socioeconomic status, and health-care access disparities in ovarian cancer treatment and mortality: Systematic review and meta-analysis. JNCI Cancer Spectrum. https://doi.org/10.1093/jncics/pkz084
Lin, Y., Wimberly, M. C., Da Rosa, P., Hoover, J., & Athas, W. F. (2018). Geographic access to radiation therapy facilities and disparities of early-stage breast cancer treatment. Geospatial Health. https://doi.org/10.4081/gh.2018.622
Knapp, S., Marziliano, A., & Moyer, A. (2014). Identity threat and stigma in cancer patients. Health Psychol Open, 1(1), 2055102914552281–2055102914552281. https://doi.org/10.1177/2055102914552281
Kang, E., et al. (2021). Impact of family caregivers’ awareness of the prognosis on their quality of life/depression and those of patients with advanced cancer: A prospective cohort study. Supportive Care Cancer, 29(1), 397–407. https://doi.org/10.1007/s00520-020-05489-8
Huber, M. A., & Kraut, N. (2015). Key drivers of biomedical innovation in cancer drug discovery. EMBO Mol Med, 7(1), 12–16. https://doi.org/10.15252/emmm.201404596
Prasad, V., & Mailankody, S. (2017). Research and development spending to bring a single cancer drug to market and revenues after approval. JAMA Internal Med, 177(11), 1569–1575. https://doi.org/10.1001/jamainternmed.2017.3601
Chen, E. Y., Raghunathan, V., & Prasad, V. (2019). An overview of cancer drugs approved by the US Food and Drug Administration based on the surrogate end point of response rate. JAMA Internal Med, 179(7), 915–921. https://doi.org/10.1001/jamainternmed.2019.0583
Cohen, E. E. W., et al. (2019). The Society for Immunotherapy of Cancer consensus statement on immunotherapy for the treatment of squamous cell carcinoma of the head and neck (HNSCC). Journal of ImmunoTherapy Cancer, 7(1), 184. https://doi.org/10.1186/s40425-019-0662-5
Egestad, H. (2013). The significance of fellow patients for head and neck cancer patients in the radiation treatment period. European Journal of Oncology Nursing, 17(5), 618–624. https://doi.org/10.1016/j.ejon.2013.01.001
Hamann, H. A., Ver Hoeve, E. S., Carter-Harris, L., Studts, J. L., & Ostroff, J. S. (2018). Multilevel opportunities to address lung cancer stigma across the cancer control continuum. Journal of Thoracic Oncology, 13(8), 1062–1075. https://doi.org/10.1016/j.jtho.2018.05.014
Vrinten, C., Gallagher, A., Waller, J., & Marlow, L. A. V. (2019). Cancer stigma and cancer screening attendance: A population based survey in England. BMC Cancer, 19(1), 566. https://doi.org/10.1186/s12885-019-5787-x
Laetsch, T. W., et al. (2019). Patient-reported quality of life after tisagenlecleucel infusion in children and young adults with relapsed or refractory B-cell acute lymphoblastic leukaemia: A global, single-arm, phase 2 trial. Lancet Oncology, 20(12), 1710–1718. https://doi.org/10.1016/s1470-2045(19)30493-0
Dai, M., et al. (2020). Patients with cancer appear more vulnerable to SARS-CoV-2: A multicenter study during the COVID-19 outbreak. Cancer Discovery, 10(6), 783–791. https://doi.org/10.1158/2159-8290.Cd-20-0422
Spanjaart, A. M., et al. (2021). Poor outcome of patients with COVID-19 after CAR T-cell therapy for B-cell malignancies: results of a multicenter study on behalf of the European Society for Blood and Marrow Transplantation (EBMT) Infectious Diseases Working Party and the European Hematology Association (EHA) Lymphoma Group. Leukemia, 35(12), 3585–3588. https://doi.org/10.1038/s41375-021-01466-0
Liu, C., Zhao, Y., Okwan-Duodu, D., Basho, R., & Cui, X. (2020). COVID-19 in cancer patients: Risk, clinical features, and management. Cancer Biology & Medicine, 17(3), 519–527. https://doi.org/10.20892/j.issn.2095-3941.2020.0289
Colomer-Lahiguera, S., et al. (2020). Patient-reported outcome instruments used in immune-checkpoint inhibitor clinical trials in oncology: a systematic review. Journal of Patient Reported Outcomes, 4(1), 58. https://doi.org/10.1186/s41687-020-00210-z
QUALITOP. (2021). https://h2020qualitop.liris.cnrs.fr/wordpress/index.php/project/. Retrieved March 22, 2021.
Acknowledgements
The authors thank the individuals who participated in interviews as subject matter experts as well as participants at the International System Dynamics Conference who provided feedback on earlier versions of the causal loop diagram.
Funding
Open Access funding provided by the MIT Libraries. This research was made possible by European Union’s Horizon 2020 research and innovation program under grant agreement No 875171.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflicts of interest to declare.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Beaulieu, E., Spanjaart, A., Roes, A. et al. Health-related quality of life in cancer immunotherapy: a systematic perspective, using causal loop diagrams. Qual Life Res 31, 2357–2366 (2022). https://doi.org/10.1007/s11136-022-03110-5
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11136-022-03110-5