Abstract
Background and objective
While several mobility measures exist, there is large variability across measures in how mobility is conceptualized, the source of information and the measurement properties making it challenging to select relevant mobility measures for individuals with acquired brain injury (ABI). Therefore, the objective was to conduct a comprehensive synthesis of existing evidence on the measurement properties, the interpretability and the feasibility of mobility measures from various sources of information (patients, clinicians, technology) using an umbrella review of published systematic reviews among individuals with ABI.
Methods
Ovid MEDLINE, CINHAL, Cochrane Library and EMBASE electronic databases were searched from 2000 to March 2020. Two independent reviewers appraised the methodological quality of the systematic reviews using the Joanna Briggs Institute critical appraisal checklist. Measurement properties and quality of evidence were applied according to COnsensus-based Standards for the Selection of Health Measurement Instrument (COSMIN) guidelines. Mobility measures were categorized using international standards with the international classification of functioning, disability and health (ICF).
Results
Thirty-five systematic reviews were included covering 147 mobility measures, of which 85% were mapped to the ICF Activity and Participation component. Results showed an acceptable overall "sufficient" rating for reliability, construct validity and responsiveness for 132 (90%), 127 (86%) and 76 (52%) of the measures, respectively; however, among these measures, ≤ 25% of the methods for evaluating these properties were rated as ‘high’ quality of evidence. Also, there was limited information that supports measure feasibility and scoring interpretability.
Conclusions
Future systematic reviews should report measures’ content validity to support the use of the measure in clinical care and research. More evaluations of the minimal important difference and floor and ceiling effects are needed to help guide clinical interpretation.
Registration information
International Prospective Register of Systematic Reviews (PROSPERO); ID: CRD42018100068.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Summary
This umbrella review presents a comprehensive synthesis of measurement properties, interpretability and feasibility of mobility measures, from various sources of information (patients, clinicians, technology) among individuals with acquired brain injury including stroke and traumatic brain injury. Both researchers and clinicians search for measures that can be used to evaluate the impact of interventions and monitor changes in patients’ health. Mobility is a key focus of rehabilitation and essential to prepare individuals to return to the community. Evaluating the interplay between the determinants that influence mobility is essential to better understand what influences each patient’s mobility and tailor interventions to meet their needs. This review comprised 147 mobility measures. Results showed an acceptable overall "sufficient" rating for reliability, construct validity and responsiveness for 132 (90%), 127 (86%) and 76 (52%) of the measures, respectively; however, among these measures ≤ 25% of the methods for evaluating these properties were rated as ‘high' quality of evidence. Using different sources of information to measure mobility among individuals with acquired brain injury provides complementary information allowing us to incorporate the self-administered questionnaires, clinical data and data from technology measures to evaluate factors that cannot be readily reported, to support decision making in rehabilitation care. Thus, this review presents the characteristics, application, measurement properties, interpretability and feasibility of mobility measures.
Introduction
Acquired brain injury (ABI), including traumatic brain injury (TBI) and stroke, is the leading cause of disability worldwide [1,2,3]. According to the World Health Organization, the global incidence of all-severity TBI is estimated at 69 million people, while 15 million people suffer a stroke worldwide each year [4,5,6]. Statistics Canada indicates that 100,000 Canadians will experience a stroke (59%) or a TBI (71%) each year [5]. Among the 1.5 million Canadians with ABI that go through the care continuum annually; over 60% report ongoing restrictions in mobility and participation in societal roles [5]. Webber et al. [7] defined mobility broadly as the ability to move oneself within environments that expand from one's home to the neighbourhood and regions beyond. It identifies five "key" interrelated determinants of mobility: cognitive, psychosocial, physical, environmental and financial influences. The multidimensionality and complexity of all domains that encompass mobility are also reflected in the international classification, functioning, disability and health Framework (ICF) mobility core set [8]. The ICF classifies mobility under body function including motion of all body bones and joints. In the activities and participation section, mobility is given an entire chapter and it is about moving by changing body position or location; or by transferring from one place to another, by using the upper extremity in carrying, moving or manipulating objects, by walking, running, or climbing, and by using various forms of transportation. In the environmental factors section, mobility is classified as products, devices, domesticated animals and services used for transportation [8].
Appropriate outcome measures are critical to accurately characterize and monitor changes in mobility during rehabilitation interventions for individuals with ABI [9]. However, selection of the best measure is difficult given the vast number of measures available, and the often unclear distinctions between them. While published guidelines recommend the use of valid, reliable and responsive assessment tools [10,11,12,13], guidance does not extend to which outcome measures are optimal for particular evaluative needs [14,15,16,17,18]. Researchers and clinicians also need to consider the content of measures and whether the domains evaluated match research and clinical objectives. A comparative examination of mobility measures will provide researchers and clinicians with the information needed to select the best outcome measure(s) to address the impairments, activity limitations and participation restrictions experienced by individuals with ABI. The ICF framework can be used to systematically classify the different domains of available outcome measures and, therefore, provide an additional basis for selection of a measure, based on comparison of the content [8].
There are also different sources of information of mobility measures. Outcomes that can only be assessed by asking the person directly are termed patient-reported outcomes (PROs) while clinician-reported outcomes (ClinROs) involve clinical judgement. Performance-reported outcomes (PerfOs) require patient cooperation and motivation [19]. Technology-based outcomes (TechOs) include sensors or assistive technologies to capture community mobility [20]. Self-reported outcomes (SROs) are not the same as PROs because SROs are outcomes that can be reported by the person with ABI but also observed and scored by someone else [22, 24, 25]. Most existing reviews on measuring mobility among individuals with ABI are limited to physical aspects and do not account for an expanded definition of mobility that encompasses mobility determinants [21,22,23,24,25,26,27]. Many walking measures are available and provide an index of what an individual can do or believes they can do, but the extent to which they indicate actual performance in the home environment is limited [28]. Life-space measures attempt to capture broader mobility, including mobility inside and outside the home, within the neighbourhood and beyond [29]. However, life-space measures do not capture transportation patterns or community engagement directly. To date, reviews have indicated that no measure evaluates mobility holistically among individuals with ABI.
Without considering the multidimensional nature of mobility, evaluations will inadequately prepare individuals to return to the community post-rehabilitation and limit our ability to correctly identify interventions which target factors that influence mobility in a given context. Clinicians require information on the content of measures to select comprehensive measures of mobility, as well as on measurement properties to ensure the minimum decision criteria to personalize care and deliver high-quality rehabilitation.
Moreover, Clinicians and clinical researchers may be unfamiliar with how to interpret the score of the mobility measure. They may not understand or have reference to the usual distribution of scores of a particular measure in a clinical or general population. Distribution of scores constitutes the absence of a problem, or meaningful changes in scores are needed for clinicians to know what cut-points of scoring indicate an action is warranted. Without reference values from a comparable population, researchers will not know whether an observed difference between two groups is meaningful, and whether a given change within or between groups is important [30]. In addition, the feasibility of using a measure (i.e. the time, cost required, length of the instrument, type and ease of administration) is another important aspect for a well-considered selection of the most appropriate measure [30, 31]. Thus, this study aimed to address these gaps by conducting a comprehensive synthesis of existing evidence on the measurement properties, the interpretability and the feasibility of mobility measures using an umbrella review [32] of published systematic reviews among individuals with ABI.
Objective
An umbrella review of published systematic reviews among individuals with ABI was used to conduct a comprehensive synthesis of existing evidence on the measurement properties, the interpretability and the feasibility of mobility measures from various sources of information (patients, clinicians, technology).
Methods
This umbrella review was reported according to both the Joanna Briggs Institute (JBI) guidelines for conducting an umbrella review [33] and the COnsensus-based Standards for the Selection of Health Measurement Instrument (COSMIN) guidelines for systematic reviews of outcome measures [31] (Fig. 1). The reason for conducting a JBI umbrella review was to summarize evidence from existing research syntheses on the properties of mobility measures, making use of the work already completed in this area [33]. Given that the JBI umbrella review guidelines did not focus on providing a rigorous methodology to assess the measurement properties and describe the interpretability and the feasibility of an instrument, the COSMIN guidelines were used [31].
Eligibility criteria
The inclusion criteria were systematic reviews published in peer-reviewed journals. Systematic reviews were included if they met all of the following criteria: (1) individuals with ABI (Stroke, TBI) ≥ 18 years; (2) report a clear objective to identify measures of mobility; and (3) evaluate at least one measurement property of the measures. The exclusion criteria were reviews investigating the effectiveness of interventions, monitoring recovery, focusing on diagnostic screening, clinical commentaries, case reports, non-structured reviews, qualitative reviews, non-human studies and grey literature.
Search strategy
A search of the literature was performed using electronic databases of Ovid MEDLINE, CINHAL, Cochrane Library and EMBASE. The search was conducted in collaboration with a health sciences librarian to ensure that the review included the appropriate and necessary keywords. The initial search strategy was constructed for Ovid MEDLINE (SI. 1) and adapted to other databases. A combination of Medical Subject Headings (MeSH) terms, subject headings and/or key words was used. Three groups of terms were generated describing the following: (1) the population ‘acquired brain injury’ AND, (2) the outcome measure ‘mobility’ AND and (3) the psychometric properties using a sensitive validated search filter [34]. Terms within each group were combined with the Boolean operator ‘OR’. Because the search included different types of studies, the search was narrowed by filtering the search specifying the type of studies including systematic review, review and meta-analyses. This filter has been used to avoid missing important information related to mobility measures. Searches were run in July 2019 (n = 32) with an updated search in March 2020 (n = 35).
Study selection
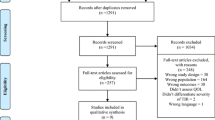
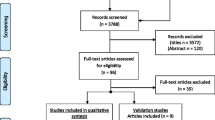
All identified systematic reviews were uploaded into ENDNote X9.1 (Clarivate Analytics, PA, USA) and duplicates were removed. Two independent reviewers screened titles and abstracts of each systematic review against the eligibility criteria. Then, full text of the included systematic reviews was retrieved and evaluated for eligibility. Disagreements were resolved by discussion and consensus. The reference list of the articles included for the full-text screening was also hand-searched for additional identification of relevant systematic reviews. The Preferred Reporting Items for Systematic reviews and Meta-Analyses (PRISMA) flow diagram [35] were used to guide the selection process.
Data extraction
Two reviewers independently extracted descriptive data from the included systematic reviews based on both JBI data extraction tool for Systematic Reviews and Research Syntheses [33] and COSMIN guidelines [31]. We extracted the characteristics of each systematic review, characteristics of mobility measures, healthcare settings or recovery phase where the mobility measure was used (if possible), results on the measurement properties, the interpretability of the scores of the measure and the feasibility of the measure. Extracted outcome measures were categorized according to the study population, sources of information and settings.
Linking to the ICF
Each extracted mobility measure was linked to the ICF according to a set of linking rules [36] (Fig. 2). A measure can be linked to one or more ICF components (body functions and structures, activity and participation and contextual factors), depending on the number of constructs contained in each measure.
Appraising methodological quality
The JBI critical appraisal checklist for systematic reviews and research syntheses quality assessment tool that includes 11 items was used to evaluate the quality of the systematic reviews [33]. In addition, the 4-point COSMIN rating scale was used to evaluate the methodological quality for evaluating the measurement properties of each study included in a given systematic review. The checklist consists of 10 measurement properties, each with their own quality criteria, which form three domains (content validity, internal structure and remaining measurement properties) [31, 37]. Each study was rated as very good, adequate, doubtful, or inadequate quality. Two independent reviewers evaluated the methodological quality followed by discussions and consensus [31, 37].
Levels of evidence appraisal
Based on COSMIN guidelines [31], for each study in a systematic review, the estimate of the measurement property was rated against the updated criteria for good measurement properties [38]. Each estimate was rated as sufficient (+), insufficient (−), or indeterminate (?). A level-of-evidence appraisal was undertaken to determine the overall quality of each measurement property for a given measure across all studies reported in the systematic reviews. The appraisal produced a final rating for each measure for each of the measurement properties. All available information was synthesized, combining the results quantitatively into one overall category of the different studies for each measure. The overall rating for the summarized results was then rated as sufficient (+), insufficient (−), inconsistent (±), or indeterminate (?) [31, 39]. The quality of the evidence was graded by using the modified-Grading of Recommendations Assessment, Development and Evaluation (modified-GRADE) approach and the quality of the evidence was graded as high, moderate, low, or very low [31]. Two independent reviewers completed the evaluation before consensus discussions.
Measurement properties
The psychometric results reported in the systematic reviews were described and categorized into the following COSMIN measurement properties including content/structural validity, internal structure, reliability, measurement error, construct validity and responsiveness. Table 1 presents the updated criteria for good measurement properties based on COSMIN guidelines [31].
Evaluate content validity
Content validity is defined as ‘the degree to which the content of the outcome measure is an adequate reflection of the construct to be measured’ and is considered the most important measurement property [40]. In the COSMIN guidelines, Terwee et al. [41] describe three aspects of content validity, including relevance, comprehensiveness and comprehensibility.
Evaluate internal structure
Internal structure refers to the relation among different items in the outcome measure. The evaluation of the internal structure includes an evaluation of the following:
Structural validity is defined as ‘the degree to which the scores of the outcome measures are an adequate reflection of the dimensionality of the construct to be measured’ [31].
Internal consistency is defined as ‘the degree of interrelatedness among the items’ [31].
Cross-cultural validity is defined as ‘the degree to which the performance of the items on a translated or culturally adapted outcome measure is comparable with the performance of the original version of the outcome measure’ [31].
Evaluate the remaining measurement properties
Reliability is defined as ‘the degree to which the measurement is free from measurement error’ [31].
Measurement error is defined as ‘the systematic and random error of a patient’s score that is not attributed to true changes in the construct to be measured’ [31].
Construct validity is defined as ‘the degree to which the scores of an instrument are consistent with hypotheses (for instance, with regard to internal relationships, relationships to scores of other instruments, or differences between relevant groups) based on the assumption that the instrument validly measures the construct to be measured’ [31].
Responsiveness is defined as ‘the ability of an instrument to detect change over time in the construct to be measured’ [31].
Describe interpretability and feasibility
Interpretability and feasibility are not measurement properties, because they do not refer to the quality of the outcome measure. However, they are considered important aspects for a well-considered selection of the outcome measure.
Interpretability is defined as ‘the degree to which one can assign qualitative meaning, that is, clinical or commonly understood connotations to an instrument’s quantitative scores or change in scores‘. It includes distribution of the scores in the study population, floor and ceiling effect, minimal important change and minimal important difference [31].
Feasibility is defined as ‘the ease of application of the measure in its intended setting, given constraints such as time or money‘. It includes type and ease of administration, length of the instrument, completion time, ease of score calculation, cost of the instruments and required equipment available in different settings [31].
Results
Search results
The search strategy yielded a total of 35 systematic reviews. Figure 3 in supplementary presents the PRISMA flow diagram including the selection process and reasons for exclusion.
Characteristics of the systematic reviews
The 35 systematic reviews were published between 2004 and 2019 in peer-reviewed journals. Nine reviews focused mainly on ClinRO/PerfO, 7 on PRO/SRO; 2 on TechO and 17 reports mixed sources of information. Twenty-six reviews targeted individuals with stroke [16,17,18, 22, 24, 26, 42,43,44,45,46,47,48,49,50,51,52,53,54,55,56,57,58,59,60,61], three targeted both stroke and TBI [21, 62, 63], one targeted TBI [64] and five incorporated stroke and TBI as part of a wider population search [23, 25, 27, 28, 65]. 320 mobility measures were extracted from the systematic reviews. After removing the duplicates, 147 measures were identified; some measures were used in multiple healthcare settings. The included systematic reviews did not specify the recovery phase for individuals with TBI (SI. 2).
Linking to the ICF
The 147 mobility measures covered the component of activities and participation (85%), followed by body functions (30%) (Table 2).
Methodological quality
Based on JBI guidelines checklist, nine (26%) systematic reviews used a standardized methodology, either PRISMA guidelines [21, 49, 52,53,54, 57] or standardized accepted guidelines from previously published work [25, 46, 63]. Although the quality of evidence of the literature search and evaluation of measurement properties of the review was generally acceptable, a minority of systematic reviews (17%) [21, 26, 49, 52,53,54] used the COSMIN Risk of Bias checklist, which resulted in low quality of evidence (SI. 3). We have applied the 4-point COSMIN rating scale to evaluate the quality of studies in each included systematic review. Among the 147 mobility measures, we found that the quality for content/structural validity was rated as adequate or higher for 16 (11%), internal consistency for 45 (30%), reliability for 54 (36%), construct validity for 101 (68%) and responsiveness for 46 (67%) of measures (SI. 4). Many measurement properties were not reported, and there was inconsistent reporting between studies. None of the included systematic reviews reported cross-cultural validity, or criterion validity.
Levels of evidence of measurement properties
Table 3 presents the level of evidence for the 147 mobility measures among individuals with ABI, categorized by population, sources of information and settings (For more information about the analysis, please refer to SI. 5, 6 & 7). Here, we present the overall level of evidence on measurement properties among all mobility measures across all settings, ABI population and source of information (n = 185).
Content validity was reported for 57 measures, with overall rating of sufficient for 55 and insufficient for 2 of measures. Of the 57, 20 were rated as high, 10 as moderate and 27 as low quality. Content validity was not reported for most of the measures, given that there was no evidence was indicated.
Internal consistency was reported for 50 measures, with overall rating of sufficient for 48 and insufficient for 2 of measures. Of the 50, 13 were rated as high, 13 as moderate and 24 as low quality. Internal consistency was not reported for most of the measures, as no evidence was indicated.
Reliability was reported for 144 measures, with overall rating of sufficient for 132, insufficient for 11 and indeterminate for 1 of measures. Of the 144, 36 were rated as high, 39 as moderate and 69 as low quality.
Measurement error was reported for 13 measures, with overall rating of sufficient for 12 and indeterminate for 1 of measures. Of the 13, 3 were rated as high, 3 as moderate and 7 as low quality. Measurement error was not reported for most of the measures, given that there was no evidence was indicated.
Construct validity was reported for 148 measures, with overall rating of sufficient for 127 and insufficient for 21 of measures. Of the 148, 32 were rated as high, 39 as moderate and 77 as low quality.
Responsiveness was reported for 90 measures, with overall rating of sufficient for 76 and insufficient for 14 of measures. Of the 90, 19 were rated as high, 25 as moderate and 46 as low quality.
Description of interpretability and feasibility
Fifty-seven (39%) of mobility measures (9 ClinROs, 29 PerfOs, 10 PROs, 8 SROs, a TechO) met the standards and criteria for interpretability and feasibility. For most of the measures, evaluating the distribution of scores in the study population, the availability of scores and change scores for relevant groups and the minimal important change or minimal important difference was limited. Information about floor and ceiling effects was limited and only reported in 7 (5%) of measures (Table 4 in Supplementary).
Summary of evidence
Results identified several mobility measures that were rated as ‘sufficient’ for most measurement properties as well as interpretability and feasibility, including Rivermead Mobility Index (RMI), six-minute walking test (6MWT), ten-metre walking test (10MWT), Barthel Index (BI), Berg Balance Scale (BBS), Frenchay Activity Index (FAI) and Stroke Impact Scale (SIS) among individuals with stroke and RMI and 6MWT among individuals with TBI.
Discussion
This umbrella review aimed to synthesize the measurement properties, the interpretability and the feasibility of mobility measures evaluated using clinician, patient and technology-derived information among individuals with ABI. Additionally, unified results from several reviews can provide a larger body of evidence and strengthen the recommendations based on these findings. In this review, 85% of 147 mobility measures among 35 systematic reviews were mapped mainly to the ICF component of Activity and Participation. This finding is consistent with previous studies that mapped the construct of mobility measures into the component of activity and participation [23, 26, 27, 46, 48, 56, 66, 67]. Also, our results showed that current mobility measures lack information on environmental factors. Identifying environmental factors a physical and societal levels, as a potential determinant that influences mobility, is crucial for maintaining independent mobility and fully integrates the patient's perspectives, experiences and needs into every phase of medical consultation, evaluation, treatment and follow-up. For example, participation of individuals with disabilities in society is dependent on the use of accessible designs to remove physical environmental barriers in public and private facilities [68]. Therefore, we recommend increasing the coverage of environmental factors when evaluating mobility, especially as evidence accumulates about how to tailor interventions to specific individual profiles [67].
Without published guidelines for umbrella reviews for measurement properties, we applied the COSMIN guidelines for systematic reviews of outcome measures to guide the methodology of this review [31]. This facilitated comparing the evidence supporting the measures' measurement properties across systematic reviews, identifying strengths and limitations of mobility measures and supporting the selection of outcome measures for a specific purpose. Our findings showed that the systematic reviews' methodological quality using the JBI critical appraisal tool was relatively low as 83% of systematic reviews did not apply COSMIN Risk of Bias checklist. The use of clear, unified criteria for the evaluation of measurement properties enable a reasonable comparison between the findings and are recommended for future systematic reviews.
Although content validity is considered the most important measurement property [31], only 11% of measures were evaluated as ‘adequate‘. High-quality content validity systematic reviews include studies with representative samples of target users who could attest to the relevance, comprehensiveness and comprehensibility of the measurement tool [31]. Future systematic reviews should report measures’ content validity, as the appropriate content as perceived by target users imperative to support the use of the measure in clinical care and research. None of the included systematic reviews reported cross-cultural validity, meaning it is unknown if the tool's content validity is maintained at a conceptual level across cultures and languages. Also, criterion validity was not reported in any study due to lack of a ‘gold standard’, according to the COSMIN definition [31]. Therefore, future systematic reviews should include cross-cultural validity and criterion validity when evidence is available according to COSMIN guidelines [31].
Results showed an acceptable overall "sufficient" rating for reliability, construct validity and responsiveness for 132 (90%), 127 (86%) and 76 (52%) of the measures, respectively; however, among these measures ≤ 25% of the methods for evaluating these properties were rated as 'high; quality of evidence. One reason was related to the sample size, as the majority of systematic reviews included studies with sample size either < 50 or unreported. Recruiting an adequate sample size to detect modest but important effect sizes is a challenge in the current state of training and funding in rehabilitation research [69]. The synthesis of the sample size used to evaluate the measurement properties of each measure in this review can be used to inform the sample size that is ideal for future evaluation of mobility measures.
Only 39% of mobility measures contained information on interpretability and feasibility. For each source of information, there are different reasons for lack of feasibility which should be reported in the future studies. For ClinRO/PerfO, feasibility is primarily expressed as the proportion of missing data for participants that cannot be assessed [66]. For PRO/SRO, whether participants required assistance is considered while evaluating feasibility [70]; and for TechO, the complexity of tracking motion while carrying out daily activities may influence feasibility [71]. Less information was provided in terms of scoring interpretability. Future studies should evaluate the minimal important difference or minimal important change, and floor and ceiling effects to help guide clinical interpretation.
Results identified several mobility measures that were rated as "sufficient" for most measurement properties as well as interpretability and feasibility. RMI and 6MWT have been used across the continuum of care; SIS and 10MWT were used in both sub-acute and chronic settings; and FAI, BI and BBS were used at both acute and chronic settings. These widely used measures, however, have limitations in certain contexts; for example, a patient with cognitive impairment or unable to change body position. Decisions for selection of a mobility measure need to consider applicability to all patients and clinical contexts [72].
Few reviews of mobility measures focused on TBI as compared to stroke. Many of the outcome measures that were developed for individuals with TBI are either related to injury severity (e.g. Glasgow Coma Scale) or reflect global outcome (e.g. Disability Rating Scale). Multidimensional tools reflecting complex ranges of factors affecting TBI outcomes may be required for assessment across the continuum of care depending on the level of recovery and context of practice and the need to evaluate community activities.
Terminologies for sources of information were used interchangeably with no distinctions if patients or clinicians reported on a domain in a measure. For example, in a systematic review of PRO measures for functional performance [21] in the lower limb, they did not distinguish between SROs and PROs. Distinction between different sources of information is important as, in addition to the items and scale, the respondent influences the interpretation of the scores. Thus, a common language for the sources of information needs to be standardized to facilitate the selection of measures ensuring that evaluations of change within and between patients can be compared. In this review, we used sources of information definitions published by Mayo et al. [19].
Moreover, to capture the quality of movement, technological measures are required. For example, accelerometry provides kinematic data that can provide an opportunity to extend the quality and accuracy of measurement, filling the gaps not covered by the ClinRO, PRO and SRO scales. However, we found variations in evaluation of measurement properties between the different technologies. Two systematic reviews [22, 45] incorporated technology measures, without a standardized evaluation of the measurement properties. Standardization of how TechO measurement properties are tested is needed to increase applicability of rapidly emerging technologies in research and clinical care.
Study limitations
The main strength of this umbrella review is that we have independently applied COSMIN guidelines to synthesize measurement properties, interpretability and feasibility of ABI mobility measures. The main limitations included the following: (1) data on measurement properties relied on what was in the reviews and were not retrieved or evaluated from primary studies; (2) articles before the year 2000 were not included. This decision was based on the rationale that the recommendations for appropriate statistical methods and interpretation of the results changed over time; (3) articles with low methodological quality were not excluded, as this review intended to be a comprehensive review of measures of mobility among individuals with ABI; (4) according to the standards at the time of publication, many studies used different terms and statistical methods to examine measurement properties. Applying modern measurement standards often requires "translation" between the author's terminology and COSMIN terms; (5) systematic reviews of measures that only evaluated determinants were not included to limit the scope of this review. However, some measures included determinants of mobility as part of the content, and these are reported in this review; and (6) this review is still limited in capturing all mobility measures, as we only included systematic reviews reporting measurement properties and used systematic literature searches to enable an unbiased selection of the outcome measures. It is possible that we have missed tools that are used in clinical practice but have not been applied in research. Therefore, we missed studies that mapped mobility measures to the ICF without considering the measurement properties [12, 73,74,75,76,77,78,79,80,81,82]. Some of these domains may become important for a Core Outcome Set for mobility to standardize mobility measures among individuals with ABI.
Conclusion
This study presented a comprehensive synthesis of existing evidence on the measurement properties, the interpretability and the feasibility of mobility measures from various sources of information (patients, clinicians, technology) using an umbrella review of published systematic reviews among individuals with ABI. We expect the results to be a resource for researchers and clinicians to assist them in selecting mobility outcome measure based on the evidence supporting their psychometric properties. RMI, BI, FAI, BBS, 6MWT, 10MWT and SIS had the strongest measurement properties and support for their interpretability and feasibility. However, each measure was limited in evaluating mobility comprehensively. Also, considering tools which comprehensively capture the degree of complexity and variety of deficits experienced by individuals surviving TBI was limited in this review. The included systematic reviews were limited in reporting measures’ content validity. Also, they were limited in evaluating the minimal important difference or minimal important change, and floor and ceiling effects. Reporting these properties are essential to help guide clinical interpretation and to support the use of the measure in clinical care and research. Finally, identifying the most critical domains for mobility based on the ICF is critical to guide the development of the Core Outcome Set among individuals with ABI.
Data availability
Data are available as Supplementary Information.
Abbreviations
- 10MWT:
-
Ten-metre walking test
- 6MWT:
-
Six-minute walking test
- ABI:
-
Acquired brain injury
- BBS:
-
Berg balance scale
- BI:
-
Barthel index
- ClinRO:
-
Clinician-reported outcome
- COSMIN:
-
Consensus-based standards for the selection of health measurement instrument
- FAI:
-
Frenchay activities index
- GRADE:
-
Grading of recommendations assessment, development and evaluation
- ICF:
-
International classification, functioning, disability and health
- JBI:
-
Joanna Briggs institute
- PerfO:
-
Performance-reported outcome
- PRISMA:
-
Preferred reporting items for systematic reviews and meta-analyses
- PRO:
-
Patient-reported outcome
- RMI:
-
Rivermead mobility index
- SI:
-
Supplementary information
- SIS:
-
Stroke impact scale
- SRO:
-
Self-report outcome
- TBI:
-
Traumatic brain injury
- TechO:
-
Technology-reported outcome
References
Maas, A. I., Stocchetti, N., & Bullock, R. (2008). Moderate and severe traumatic brain injury in adults. The Lancet Neurology, 7(8), 728–741.
Patil, M., Gupta, A., Khanna, M., Taly, A. B., Soni, A., Kumar, J. K., & Thennarasu, K. (2017). Cognitive and functional outcomes following inpatient rehabilitation in patients with acquired brain injury: A prospective follow-up study. Journal of neurosciences in rural practice, 8(3), 357.
Nethan, S., Sinha, D., & Mehrotra, R. (2017). Non communicable disease risk factors and their trends in India. Asian Pacific journal of cancer prevention: APJCP, 18(7), 2005.
Dewan, M. C., Rattani, A., Gupta, S., Baticulon, R. E., Hung, Y.-C., Punchak, M., Agrawal, A., Adeleye, A. O., Shrime, M. G., Rubiano, A. M., & Rosenfeld, J. V. (2018). Estimating the global incidence of traumatic brain injury. Journal of Neurosurgery, 130(4), 1080–1097.
Chambers-Richards, T.L.-T.A. (2020). Risk factors trends, health care and general life satisfaction for select neurological conditions among an aging population in Canada. University of Saskatchewan.
Thrift, A. G., Thayabaranathan, T., Howard, G., Howard, V. J., Rothwell, P. M., Feigin, V. L., Norrving, B., Donnan, G. A., & Cadilhac, D. A. (2017). Global stroke statistics. International Journal of Stroke, 12(1), 13–32.
Webber, S. C., Porter, M. M., & Menec, V. H. (2010). Mobility in older adults: A comprehensive framework. The Gerontologist, 50(4), 443–450.
Organization, W. H. (2001). International classification of functioning, disability and health: ICF. World Health Organization.
Murphy, M. A., Resteghini, C., Feys, P., & Lamers, I. (2015). An overview of systematic reviews on upper extremity outcome measures after stroke. BMC Neurology, 15(1), 29.
Otterman, N., Veerbeek, J., Schiemanck, S., van der Wees, P., Nollet, F., & Kwakkel, G. (2017). Selecting relevant and feasible measurement instruments for the revised Dutch clinical practice guideline for physical therapy in patients after stroke. Disability and Rehabilitation, 39(14), 1449–1457.
Sullivan, J. E., Crowner, B. E., Kluding, P. M., Nichols, D., Rose, D. K., Yoshida, R., & Zipp, G. P. (2013). Outcome measures for individuals with stroke: process and recommendations from the American physical therapy association neurology section task force. Physical Therapy, 93(10), 1383–1396.
McCulloch, K. L., De Joya, A. L., Hays, K., Donnelly, E., Johnson, T. K., Nirider, C. D., Roth, H., Saliga, S., & Ward, I. (2016). Outcome measures for persons with moderate to severe traumatic brain injury: Recommendations from the American physical therapy association academy of neurologic physical therapy TBI EDGE task force. Journal of Neurologic Physical Therapy, 40(4), 269–280.
Russell, S. J., & Norvig, P. (2016). Artificial intelligence: A modern approach. Pearson Education Limited.
Miller, E. L., Murray, L., Richards, L., Zorowitz, R. D., Bakas, T., Clark, P., & Billinger, S. A. (2010). Comprehensive overview of nursing and interdisciplinary rehabilitation care of the stroke patient: A scientific statement from the American heart association. Stroke, 41(10), 2402–2448.
Party, I. S. W. (2012). National clinical guideline for stroke. Royal College of Physicians.
Salter, K., Jutai, J., Teasell, R., Foley, N., & Bitensky, J. (2005). Issues for selection of outcome measures in stroke rehabilitation: ICF body functions. Disability and Rehabilitation, 27(4), 191–207.
Salter, K., Jutai, J., Teasell, R., Foley, N., Bitensky, J., & Bayley, M. (2005). Issues for selection of outcome measures in stroke rehabilitation: ICF participation. Disability and Rehabilitation, 27(9), 507–528.
Salter, K., Jutai, J., Teasell, R., Foley, N., Bitensky, J., & Bayley, M. (2005). Issues for selection of outcome measures in stroke rehabilitation: ICF activity. Disability and Rehabilitation, 27(6), 315–340.
Mayo, N. E., Figueiredo, S., Ahmed, S., & Bartlett, S. J. (2017). Montreal accord on patient-reported outcomes (PROs) use series–paper 2: Terminology proposed to measure what matters in health. Journal of clinical epidemiology, 89, 119–124.
Webber, S. C., & Porter, M. M. (2009). Monitoring mobility in older adults using global positioning system (GPS) watches and accelerometers: A feasibility study. Journal of Aging and Physical Activity, 17(4), 455–467.
Ashford, S., Brown, S., & Turner-Stokes, L. (2015). Systematic review of patient-reported outcome measures for functional performance in the lower limb. Journal of Rehabilitation Medicine, 47(1), 9–17.
Fini, N. A., Holland, A. E., Keating, J., Simek, J., & Bernhardt, J. (2015). How is physical activity monitored in people following stroke? Disability and Rehabilitation, 37(19), 1717–1731.
Hong, I., & Bonilha, H. S. (2017). Psychometric properties of upper extremity outcome measures validated by Rasch analysis: A systematic review. International Journal of Rehabilitation Research, 40(1), 1–10.
Rowland, T. J., & Gustafsson, L. (2008). Assessments of upper limb ability following stroke: A review. British Journal of Occupational Therapy, 71(10), 427–437.
Tyson, S., & Connell, L. (2009). The psychometric properties and clinical utility of measures of walking and mobility in neurological conditions: A systematic review. Clinical Rehabilitation, 23(11), 1018–1033.
van Bloemendaal, M., van de Water, A. T., & van de Port, I. G. (2012). Walking tests for stroke survivors: A systematic review of their measurement properties. Disability and Rehabilitation, 34(26), 2207–2221.
Velstra, I.-M., Ballert, C. S., & Cieza, A. (2011). A systematic literature review of outcome measures for upper extremity function using the international classification of functioning, disability, and health as reference. PM&R, 3(9), 846–860.
Pearson, O. R., Busse, M., Van Deursen, R. W. M., & Wiles, C. M. (2004). Quantification of walking mobility in neurological disorders. QJM, 97(8), 463–475.
Peel, C., Baker, P. S., Roth, D. L., Brown, C. J., Bodner, E. V., & Allman, R. M. (2005). Assessing mobility in older adults: The UAB study of aging life-space assessment. Physical Therapy, 85(10), 1008–1019.
Velentgas, P., Dreyer, N. A., & Wu, A. W. (2013). Outcome definition and measurement. Developing a protocol for observational comparative effectiveness research: A user’s guide. Agency for Healthcare Research and Quality (US).
Prinsen, C., Mokkink, L., Bouter, L., Alonso, J., Patrick, D., de Vet, H., & Terwee, B. (2018). COSMIN guideline for systematic reviews of patient-reported outcome measures. Quality of Life Research, 27(5), 1147–1157.
Aromataris, E., Fernandez, R., Godfrey, C. M., Holly, C., Khalil, H., & Tungpunkom, P. (2015). Summarizing systematic reviews: Methodological development, conduct and reporting of an umbrella review approach. International Journal of Evidence-based Healthcare, 13(3), 132–140.
Aromataris, E., Fernandez, R., Godfrey, C., Holly, C., Khalil, H., & Tungpunkom, P. (2014). The Joanna Briggs Institute reviewers’ manual 2014: Methodology for JBI umbrella reviews. University of Adelaide: Joanna Briggs Institute.
Terwee, C. B., Jansma, E. P., Riphagen, I. I., & de Vet, H. C. (2009). Development of a methodological PubMed search filter for finding studies on measurement properties of measurement instruments. Quality of Life Research, 18(8), 1115–1123.
Moher, D., Shamseer, L., Clarke, M., Ghersi, D., Liberati, A., Petticrew, M., et al. (2015). Preferred reporting items for systematic review and meta-analysis protocols (PRISMA-P) 2015 statement. Systematic Reviews, 4(1), 1.
Cieza, A., Fayed, N., Bickenbach, J., & Prodinger, B. (2019). Refinements of the ICF linking rules to strengthen their potential for establishing comparability of health information. Disability and Rehabilitation, 41(5), 574–583.
Mokkink, L. B., De Vet, H. C., Prinsen, C. A., Patrick, D. L., Alonso, J., Bouter, L. M., & Terwee, C. B. (2018). COSMIN risk of bias checklist for systematic reviews of patient-reported outcome measures. Quality of Life Research, 27(5), 1171–1179.
Terwee, C. B., Bot, S. D., de Boer, M. R., van der Windt, D. A., Knol, D. L., Dekker, J., Bouter, M. L., & Henrica, C. W. (2007). Quality criteria were proposed for measurement properties of health status questionnaires. Journal of Clinical Epidemiology, 60(1), 34–42.
Prinsen, C. A., Vohra, S., Rose, M. R., Boers, M., Tugwell, P., Clarke, M., Williamson, P. R., & Terwee, C. B. (2016). How to select outcome measurement instruments for outcomes included in a “core outcome set”–a practical guideline. Trials, 17(1), 1–10.
Mokkink, L. B., Terwee, C. B., Patrick, D. L., Alonso, J., Stratford, P. W., Knol, D. L., Bouter, L. M., & Vet, H. C. (2010). The COSMIN study reached international consensus on taxonomy, terminology, and definitions of measurement properties for health-related patient-reported outcomes. Journal of Clinical Epidemiology, 63(7), 737–745.
Terwee, C. B., Prinsen, C. A., Chiarotto, A., Westerman, M. J., Patrick, D. L., Alonso, J., Bouter, L. M., Vet, H. C., & Mokkink, L. B. (2018). COSMIN methodology for evaluating the content validity of patient-reported outcome measures: A Delphi study. Quality of Life Research, 27(5), 1159–1170.
Baker, K., Cano, S. J., & Playford, E. D. (2011). Outcome measurement in stroke: A scale selection strategy. Stroke, 42(6), 1787–1794.
Barak, S., & Duncan, P. W. (2006). Issues in selecting outcome measures to assess functional recovery after stroke. NeuroRx, 3(4), 505–524.
Croarkin, E., Danoff, J., & Barnes, C. (2004). Evidence-based rating of upper-extremity motor function tests used for people following a stroke. Physical Therapy, 84(1), 62–74.
Gebruers, N., Vanroy, C., Truijen, S., Engelborghs, S., & De Deyn, P. P. (2010). Monitoring of physical activity after stroke: A systematic review of accelerometry-based measures. Archives of Physical Medicine and Rehabilitation, 91(2), 288–297.
Geroin, C., Mazzoleni, S., Smania, N., Gandolfi, M., Bonaiuti, D., Gasperini, G., Sale, P., Munari, D., Waldner, A., Spidalieri, R., Bovolenta, F., Picelli, A., Posteraro, F., Molteni, F., & Franceschini, M. (2013). Systematic review of outcome measures of walking training using electromechanical and robotic devices in patients with stroke. Journal of Rehabilitation Medicine, 45(10), 987–996.
Gor-García-Fogeda, M. D., Molina-Rueda, F., Cuesta-Gómez, A., Carratalá-Tejada, M., Alguacil-Diego, I. M., & Miangolarra-Page, J. C. (2014). Scales to assess gross motor function in stroke patients: A systematic review. Archives of Physical Medicine and Rehabilitation, 95(6), 1174–1183.
Lemmens, R. J., Timmermans, A. A., Janssen-Potten, Y. J., Smeets, R. J., & Seelen, H. A. (2012). Valid and reliable instruments for arm-hand assessment at ICF activity level in persons with hemiplegia: A systematic review. BMC Neurology, 12(1), 21.
Martins, J. C., Aguiar, L. T., Nadeau, S., Scianni, A. A., Teixeira-Salmela, L. F., & Faria, C. D. C. D. M. (2019). Measurement properties of self-report physical activity assessment tools for patients with stroke: A systematic review. Brazilian Journal of Physical Therapy, 23(6), 476–490.
Oczkowski, C., & O’Donnell, M. (2010). Reliability of proxy respondents for patients with stroke: A systematic review. Journal of Stroke and Cerebrovascular Diseases, 19(5), 410–416.
Pollock, C., Eng, J., & Garland, S. (2011). Clinical measurement of walking balance in people post stroke: A systematic review. Clinical Rehabilitation, 25(8), 693–708.
Salbach Kelly, N. M., O’Brien, K. K., Brooks, D., Irvin, E., Martino, R., Takhar, P., Chan, S., & Howe, J.-A. (2017). Considerations for the selection of time-limited walk tests poststroke: a systematic review of test protocols and measurement properties. Journal of Neurologic Physical Therapy, 41(1), 3–17.
Scrivener, K., Sherrington, C., & Schurr, K. (2013). A systematic review of the responsiveness of lower limb physical performance measures in inpatient care after stroke. BMC Neurology, 13(1), 4.
Silva, P. F., Quintino, L. F., Franco, J., & Faria, C. D. (2014). Measurement properties and feasibility of clinical tests to assess sit-to-stand/stand-to-sit tasks in subjects with neurological disease: A systematic review. Brazilian Journal of Physical Therapy, 18(2), 99–110.
Simpson, L. A., & Eng, J. J. (2013). Functional recovery following stroke: Capturing changes in upper-extremity function. Neurorehabilitation and Neural Repair, 27(3), 240–250.
Sivan, M., O’Connor, R. J., Makower, S., Levesley, M., & Bhakta, B. (2011). Systematic review of outcome measures used in the evaluation of robot-assisted upper limb exercise in stroke. Journal of Rehabilitation Medicine, 43(3), 181–189.
Sorrentino, G. S. P., Solaro, C., Rabini, A., Cerri, C., & Ferriero, G. (2018). Clinical measurement tools to assess trunk performance after stroke: a systematic review. European Journal of Physical and Rehabilitation Medicine. https://doi.org/10.2736/S1973-9087.18.05178-X
Teale, E. A., & Young, J. B. (2010). A review of stroke outcome measures valid and reliable for administration by postal survey. Reviews in Clinical Gerontology, 20(4), 338–353.
Tse, T., Douglas, J., Lentin, P., & Carey, L. (2013). Measuring participation after stroke: A review of frequently used tools. Archives of Physical Medicine and Rehabilitation, 94(1), 177–192.
Van Peppen, R. P., Hendriks, H., Van Meeteren, N. L., Helders, P. J., & Kwakkel, G. (2007). The development of a clinical practice stroke guideline for physiotherapists in The Netherlands: A systematic review of available evidence. Disability and Rehabilitation, 29(10), 767–783.
Verheyden, G., Nieuwboer, A., Van de Winckel, A., & De Weerdt, W. (2007). Clinical tools to measure trunk performance after stroke: A systematic review of the literature. Clinical Rehabilitation, 21(5), 387–394.
Ashford, S., Slade, M., Malaprade, F., & Turner-Stokes, L. (2008). Evaluation of functional outcome measures for the hemiparetic upper limb: A systematic review. Journal of Rehabilitation Medicine, 40(10), 787–795.
Connell, L. A., & Tyson, S. F. (2012). Clinical reality of measuring upper-limb ability in neurologic conditions: A systematic review. Archives of Physical Medicine and Rehabilitation, 93(2), 221–228.
Wilde, E. A., Whiteneck, G. G., Bogner, J., Bushnik, T., Cifu, D. X., Dikmen, S., French, L., Giacino, J. T., Hart, T., Malec, J. F., Millis, S. R., Novack, T. A., Sherer, M., Tulsky, D. S., Vanderploeg, R. D., & Steinbuechel, N. (2010). Recommendations for the use of common outcome measures in traumatic brain injury research. Archives of Physical Medicine and Rehabilitation, 91(11), 1650–16617.
Stevens, P. M. (2010). Clinimetric properties of timed walking events among patient populations commonly encountered in orthotic and prosthetic rehabilitation. JPO: Journal of Prosthetics and Orthotics, 22(1), 62–74.
Mortenson, W. B., Miller, W. C., & Auger, C. (2008). Issues for the selection of wheelchair-specific activity and participation outcome measures: A review. Archives of Physical Medicine and Rehabilitation, 89(6), 1177–1186.
Menezes, K. V. R. S., Auger, C., de Souza Menezes, W. R., & Guerra, R. O. (2017). Instruments to evaluate mobility capacity of older adults during hospitalization: A systematic review. Archives of Gerontology and Geriatrics, 72, 67–79.
Evans, D. W., Lucas, N., & Kerry, R. (2017). The form of causation in health, disease and intervention: Biopsychosocial dispositionalism, conserved quantity transfers and dualist mechanistic chains. Medicine, Health Care and Philosophy, 20(3), 353–363.
Asano, M., Dawes, D., Arafah, A., Moriello, C., & Mayo, N. (2009). What does a structured review of the effectiveness of exercise interventions for persons with multiple sclerosis tell us about the challenges of designing trials? Multiple Sclerosis Journal, 15(4), 412–421.
Auger, C., Demers, L., & Swaine, B. (2006). Making sense of pragmatic criteria for the selection of geriatric rehabilitation measurement tools. Archives of gerontology and geriatrics, 43(1), 65–83.
Maceira-Elvira, P., Popa, T., Schmid, A.-C., & Hummel, F. C. (2019). Wearable technology in stroke rehabilitation: Towards improved diagnosis and treatment of upper-limb motor impairment. Journal of Neuroengineering and Rehabilitation, 16(1), 142.
Auger, C., Demers, L., Desrosiers, J., Giroux, F., Ska, B., & Wolfson, C. (2007). Applicability of a toolkit for geriatric rehabilitation outcomes. Disability and Rehabilitation, 29(2), 97–109.
Geyh, S., Kurt, T., Brockow, T., Cieza, A., Ewert, T., Omar, Z., & Resch, K. L. (2004). Identifying the concepts contained in outcome measures of clinical trials on stroke using the international classification of functioning, disability and health as a reference. Journal of Rehabilitation Medicine, 36, 56–62.
Lord, S. E., & Rochester, L. (2005). Measurement of community ambulation after stroke: Current status and future developments. Stroke, 36(7), 1457–1461.
McCabe, P., Lippert, C., Weiser, M., Hilditch, M., Hartridge, C., & Villamere, J. (2007). Community reintegration following acquired brain injury. Brain Injury, 21(2), 231–257.
Nichol, A. D., Higgins, A., Gabbe, B., Murray, L., Cooper, D., & Cameron, P. (2011). Measuring functional and quality of life outcomes following major head injury: Common scales and checklists. Injury, 42(3), 281–287.
Steins, D., Dawes, H., Esser, P., & Collett, J. (2014). Wearable accelerometry-based technology capable of assessing functional activities in neurological populations in community settings: A systematic review. Journal of Neuroengineering and Rehabilitation, 11(1), 1–13.
Verceles, A. C., & Hager, E. R. (2015). Use of accelerometry to monitor physical activity in critically ill subjects: A systematic review. Respiratory Care, 60(9), 1330–1336.
Wang, Q., Markopoulos, P., Yu, B., Chen, W., & Timmermans, A. (2017). Interactive wearable systems for upper body rehabilitation: A systematic review. Journal of Neuroengineering and Rehabilitation, 14(1), 1–21.
Wang, S., Hsu, C. J., Trent, L., Ryan, T., Kearns, N. T., Civillico, E. F., & Kontson, K. L. (2018). Evaluation of performance-based outcome measures for the upper limb: A comprehensive narrative review. PM&R, 10(9), 951-962. e953.
Williams, G., Robertson, V., & Greenwood, K. (2004). Measuring high-level mobility after traumatic brain injury. American Journal of Physical Medicine & Rehabilitation, 83(12), 910–920.
Zheng, H., Black, N. D., & Harris, N. D. (2005). Position-sensing technologies for movement analysis in stroke rehabilitation. Medical and Biological Engineering and Computing, 43(4), 413–420.
Richards, C. L., Malouin, F., & Nadeau, S. (2015). Stroke rehabilitation: Clinical picture, assessment, and therapeutic challenge. Progress in Brain Research, 218, 253–280.
García-Rudolph, A., Sánchez-Pinsach, D., Salleras, E. O., & Tormos, J. M. (2019). Subacute stroke physical rehabilitation evidence in activities of daily living outcomes: A systematic review of meta-analyses of randomized controlled trials. Medicine. https://doi.org/10.1097/MD.0000000000014501
Acknowledgements
Rehab Alhasani is supported by Princess Nourah bint Abdulrahman University, Riyadh, Saudi Arabia. Dr. Sara Ahmed and Dr. Claudine Auger are supported by career awards from the Fonds de la recherche du Québec-santé. The funding agency had no role in the design of the study, in the collection and analysis of the data, or in the decision to publish the results.
Funding
1. Initiatives pour le développement de Nouvelles technologies et Pratiques en Réadaptation (INSPIRE) Lindsay Foundation 2. the Canadian Foundation of Innovation Funding for the Biomedical Research and Informatics Living Laboratory for Innovative Advances of New Technologies in Community Mobility Rehabilitation (BRILLIANT) (https://www.brilliant-cfi).
Author information
Authors and Affiliations
Contributions
All authors contributed to the design of this study, provided critical insights and contributed to the final written manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors have not disclosed any conflict of interest.
Ethical approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
This manuscript has not been published and is not under consideration for publication elsewhere.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Alhasani, R., Auger, C., Paiva Azevedo, M. et al. Quality of mobility measures among individuals with acquired brain injury: an umbrella review. Qual Life Res 31, 2567–2599 (2022). https://doi.org/10.1007/s11136-022-03103-4
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11136-022-03103-4