Abstract
Purpose
To support the use of quality of life (QOL) assessment tools for older adults, we developed knowledge translation (KT) resources tailored for four audiences: (1) older adults and their family caregivers (micro), (2) healthcare providers (micro), (3) healthcare managers and leaders (meso), and (4) government leaders and decision-makers (macro). Our objectives were to (1) describe knowledge gaps and resources and (2) develop corresponding tailored KT resources to support use of QOL assessment tools by each of the micro-, meso-, and macro-audiences.
Methods
Data were collected in two phases through semi-structured interviews/focus groups with the four audiences in Canada. Data were analyzed using qualitative description analysis. KT resources were iteratively refined through formative evaluation.
Results
Older adults and family caregivers (N = 12) wanted basic knowledge about what “QOL assessment” meant and how it could improve their care. Healthcare providers (N = 13) needed practical solutions on how to integrate QOL assessment tools in their practice. Healthcare managers and leaders (N = 14) desired information about using patient-reported outcome measures (PROMs) and patient-reported experience measures (PREMs) in healthcare programs and quality improvement. Government leaders and decision-makers (N = 11) needed to know how to access, use, and interpret PROM and PREM information for decision-making purposes. Based on these insights and evidence-based sources, we developed KT resources to introduce QOL assessment through 8 infographic brochures, 1 whiteboard animation, 1 live-action video, and a webpage.
Conclusion
Our study affirms the need to tailor KT resources on QOL assessment for different audiences. Our KT resources are available: www.healthyqol.com/older-adults.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Plain English summary
Older adults living at home facing frailty, and their family, often have challenges with quality of life. Tools for assessing quality of life can help make the priorities of patients and families visible to healthcare providers and leaders. But there is a lack of resources to support the use of these quality of life tools by patients, families, healthcare providers, healthcare managers, and government leaders. Our goal was to (1) identify gaps in knowledge, and (2) develop “tailored” resources (e.g., videos, written materials.) to support the use of tools to assess quality of life. We found there was a need for simple, introductory resources to address the knowledge gaps of different audiences. With participants’ input, we developed and tailored resources to introduce quality of life assessment through 8 infographic brochures, 1 whiteboard animation, 1 live-action video, and a webpage. Our findings confirm that different audiences have different needs for resources to support their use of quality of life tools. Our tailored resources are now freely available at www.healthyqol.com/older-adults.
Introduction
Older adults living at home with frailty often have complex problems that not only affect their ability to function, but also their quality of life (QOL) [1, 2]. Similarly, their family caregivers' QOL can be affected as they often assume primary responsibility for coordinating and providing care, which may create or impact upon their own health issues [3,4,5]. QOL has been defined by the World Health Organization as “individuals’ perception of their position in life in the context of the culture and value systems in which they live and in relation to their goals, expectations, standards and concerns” [6]. QOL assessments can help to ensure that healthcare for patients and family caregivers is informed by their experiences [7]. QOL assessment tools can facilitate such assessments by asking patients and family caregivers to respond to questions for measuring their QOL, healthcare experiences, physical, mental, and social health. These tools include health-related QOL measures, patient-reported outcome measures (PROMs), or patient-reported experience measures (PREMs). Our review identified 65 PROMs or PREMs used with older adults living at home, and their family caregivers [8]. This project seeks to support the use of QOL assessment tools for older adults by developing introductory and readily available (www.healthyqol.com/older-adults) knowledge translation (KT) resources tailored for different users.
Globally, as the population is aging due to longer life expectancies and decreasing fertility [9], there is increasing requirement for home-based care to support the needs of older adults [10]. In Canada, where our work is located, in 2013, 1.8 million people received publicly-funded homecare, 70% of whom were older adults [11]. There is increasing interest in using QOL assessment tools to support a patient-, person- or people-centered approach to care [12, 13]. These tools are used by different people across micro- (patients, caregivers, and healthcare providers), meso- (healthcare organization managers and leaders), and macro-levels (government) of healthcare [8, 14,15,16,17]. At the micro-level, QOL assessments in clinical practice can improve healthcare provider-patient communication, raise awareness of problems that would otherwise be unidentified, improve care plans, and improve multidisciplinary collaboration [18,19,20,21]. At the meso-level, healthcare managers and leaders increasingly advocate for the routine use of PROMs and PREMs for patient-/person-centered program evaluation and quality improvement purposes [14, 22]. At the macro-level, use of PROMs and PREMs by government leaders is gaining momentum [23, 24].
Despite decades of research and systematic reviews on using QOL assessment tools, there is a dearth of resources that translate this evidence to support the use of QOL assessment tools by different people across micro-, meso-, and macro-levels of healthcare [25,26,27]. To support routine use of QOL assessment tools, it is important that all users, including older adults, family caregivers, healthcare providers, and decision-makers, are knowledgeable about what these tools are and how they can be used to improve healthcare. A few resources have been developed to support use of QOL assessment tools, with a primary focus on use by clinicians in practice [28,29,30,31,32,33]. However, none of the resources focus on contexts of older adult care, nor have they been tailored to address different perspectives and needs of knowledge user audiences at micro-, meso-, or macro-levels [27]. Uptake and evidence-based use of QOL assessment tools requires KT resources be tailored to address the different knowledge needs [34, 35]. This project aimed to address this gap by learning from four audiences about their needs for tailored, evidence-informed KT resources regarding use of QOL assessment tools, including (1) older adults and their family caregivers (micro), (2) healthcare providers (micro), (3) healthcare managers and leaders (meso), and (4) government leaders and decision-makers (macro). Our two objectives were to (1) describe knowledge gaps and resources and (2) develop corresponding tailored KT resources to support use of QOL assessment tools by each of the micro-, meso-, and macro-audiences.
Methods
Study design
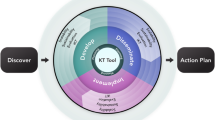
Our research was guided by qualitative description methodology [36, 37] and the Knowledge-to-Action Framework [34, 35] (See Fig. 1). There were two phases: (1) “Initial Consultation” involved recruitment, data collection, and analysis focused on objective 1 as the basis for initial development of KT resources using input from micro-, meso-, and macro-audiences and drawing from evidence-based resources regarding the use of QOL assessment tools. (2) “Formative Evaluation” involved ongoing refinement and iterative evaluation of the KT resources with study participants (objective 2). Our KT team was established to ensure representation of different audiences by including clinicians, healthcare leaders, patients, family caregivers, members from patient advocacy groups and non-profit organizations, and researchers with expertise in QOL assessment and person-centered care.
Study Design Guided by Knowledge-to-Action Frameworka. The Knowledge-to-Action Framework can be regarded as a cycle of integrated knowledge translation (KT), denoted by the circular arrows. In the Initial Consultation (Objective 1/Phase 1), we identified the problem, drafted KT resources for the local context, and ensured they addressed both knowledge gaps and supports. In the Formative Evaluation (Objective 2/Phase 2), we then tailored and revised the KT resources after the 2nd round of interviews with participants. Future work is needed for summative evaluation. aGuided by Graham ID, Logan J, Harrison MB, Straus SE, Tetroe J, Caswell W et al. Lost in knowledge translation: time for a map? The Journal of Continuing Education in the Health Professions. 2006;26(1):13–24. https://doi.org/10.1002/chp.47
Settings and sample
We created the following four participant groups at micro-, meso-, and macro-levels sampled from healthcare across Canada: (1) adults ≥ 65 years old, receiving home care for chronic conditions associated with frailty [1, 2] (e.g., cancer, cardiac, respiratory, endocrine, musculoskeletal conditions, and co-morbidities), and their family caregivers (micro); (2) healthcare providers of older adults living at home (micro); (3) healthcare managers and leaders responsible for providing care to older adults (meso); and (4) provincial government leaders and decision-makers of healthcare organizations (macro) responsible for home health services for older adults.
We used purposive and snowball sampling to obtain a diversity of perspectives (albeit limited to people conversant in English) by inviting older adults and family caregivers with different health conditions, genders, and other participants with various roles and responsibilities. All patients, caregivers, and many healthcare providers (micro) were recruited via telephone or e-mail from an ongoing project on QOL assessments in home care for older adults [38]. Within that project, they had experience with an electronic QOL assessment and practice support system (“QPSS”). Healthcare managers and leaders (meso), as well as government leaders and decision-makers (macro) were recruited via KT team members’ networks, and invited by e-mail. Twenty-seven people declined participation; no one withdrew.
Data collection
Data were collected via 37 interviews and 2 focus groups for the Initial Consultation phase (Nov. 2018–Mar. 2019), and via 23 interviews for the Formative Evaluation phase (Aug. 2019–Oct. 2019). Interviews/focus groups were conducted by the study co-lead (KSM) and five trainees (one nurse, three nursing students, one anthropologist), in homes (n = 20, with older adults or family caregivers), at workplaces (n = 7), or on the phone (n = 35), lasting 45–90 min. There were no prior relationships between interviewers and participants. Debriefing meetings between interviewers and study leads facilitated trainee interview training, ensured data quality, enhanced reflexivity, and oriented the interviewers to the data.
We developed semi-structured interview/focus group guides with open-ended questions for each participant group and the project’s two phases. During the Initial Consultation, we asked participants to first discuss their experience and familiarity with QOL assessment tools, and then more specifically to discuss knowledge gaps, opportunities, and challenges regarding use of QOL assessment tools, as well as supports, benefits, and recommendations to facilitate their use of QOL assessment tools. For the Formative Evaluation, we invited participants to review and provide feedback on the KT resources. The guides went through iterative rounds of development with the KT team. Interviews/focus groups were recorded and transcribed.
Data analysis methods for objective 1
Guided by qualitative description analysis [36, 37], data were coded and categorized to describe knowledge gaps and resources to support the use of QOL assessment tools. Rather than define knowledge gaps as deficits, we viewed them as “a fruitful site for the development of knowledge and practice capacity” (p. 179) [39]. Supportive resources were considered in relation to self (micro), patient-care (micro), program delivery/quality improvement (meso), or policy development (macro), and included recommendations for tailored KT resource development.
The transcripts from the first interviews were re-read to generate the initial codebook, which was iteratively refined to arrive at categories of knowledge gaps and supportive resources for the use of QOL assessment tools. N-Vivotm software was used to facilitate collaborative coding. Data from the four participant groups were analyzed separately, and then compared, linked, and contrasted [40] to inform the development of tailored KT resources. One experienced qualitative analyst and two trainees coded the data under the supervision of the lead researchers (RS, KSM). Differences in coding were discussed with trainees and supervisors, and decisions were made through dialogue and consensus. Saturation was achieved [41, 42] when no new categories were identified and when participants’ descriptions became repetitive.
Data analysis methods for objective 2
We followed integrated-KT principles [34, 35] to design and tailor KT resources for each participant group using learnings from the Initial Consultation while drawing upon evidence-based sources about using QOL assessment tools. The KT resources were collaboratively developed through several rounds of revision to keep the resources consistent, easy to read (for the intended audience), and relevant. Our KT team worked with a content editor to ensure consistency of language and messages and a graphic designer to create a simple, harmonious design across all resources.
The KT resources were refined through a formative evaluation process [43] based on participant feedback obtained via the interviews. Before the interviews, participants viewed the KT resources through various means: online, through a recorded webinar, or on a tablet with a KT team member prior to the interview. Participants shared their perspectives and provided input to ensure the resources were easy to understand, appealing, relevant, appropriate, and facilitated understanding of QOL assessments. The same qualitative description analysis methods as for objective 1 were followed to describe the participant’s evaluative feedback to tailor KT resources for micro-, meso-, macro-audiences.
Rigor
To attend to trustworthiness, we drew upon qualitative principles of credibility, transferability, and confirmability [40, 44]. Credibility was upheld through longitudinal engagement, iterative cycles of engagement, and member-checking with the four groups of participants and members of our KT team. Field notes written after each interview included reporting of context, and interviews were discussed during team meetings. Confirmability was established through a detailed audit trail ensuring that records were kept of design and analytic decisions. Preliminary analysis and category development were presented to the full KT team on 6 occasions through video conference or email, and their feedback was incorporated into the refinement of both the codebook and ongoing analysis. These team discussions facilitated reflexivity and were critical to the integrity of the project. The Consolidated Criteria for Reporting Qualitative Research guidelines were followed [45].
Results
Phase 1: Description of knowledge gaps and supportive resources
Forty-seven participants consented to participate including 7 patients, 4 family caregivers, 13 healthcare providers, 14 healthcare managers and leaders, and 11 government leaders and decision-makers (Table 1). Participants in all groups were predominantly female and of European or Canadian descent.
The Initial Consultation facilitated description of the knowledge gaps and resources to support use of QOL assessment tools. While knowledge gaps were primarily related to barriers to knowledge use, these were interconnected with pragmatic, organizational, or systems barriers experienced by participant groups. Findings from the Initial Consultation informed preliminary development of tailored KT resources. See Table 2 for details with objectives, categories, and exemplar quotes.
Each of the four participant groups had unique needs regarding use of QOL assessment tools. Older adults and family caregivers desired basic knowledge to improve their understanding of what “QOL assessment” meant and how information about their QOL could be used to improve care. They wanted to know how their responses could actively be used to inform their self-care or the healthcare they received. As one older adult said, What are you going to do with information like that? They recommended that resources could include videos or pamphlets, and that these resources address how to answer questions in QOL assessment tools.
Healthcare providers wanted practical, didactic information about how to implement QOL assessment tools without adding to their workload, and how to use QOL information to inform care decisions with patients and caregivers. Providers stressed the importance of integrating the QOL assessments within existing work structures and health records. One said, How will clinicians be able to process this information in a way that kind of makes it fun, makes them think they’re really changing-up how they practice, but that doesn’t add workload? Healthcare providers recommended that KT resources should sustain their interest, be concise, provide examples, and pragmatically support their use of QOL assessments in practice regardless of the mode (i.e., electronic or paper).
At meso- and macro-levels, healthcare and government leader participants used the terms PROMs and PREMS to refer to QOL assessment tools. Healthcare managers and leaders recommended that KT resources show them how PROMs and PREMs could be used in organizational decision-making at a meso-level. They needed information to understand how these tools could be used in their practice overseeing healthcare programs, including quality improvement. One leader asked, How are we thinking provincially on a bigger scale? What are we actually using this information for? How are we then translating that down to the people who need what information? They also felt constrained by a lack of time, lack of training, and other structural barriers related to integration with electronic health systems. They specifically recommended resources on implementation of QOL assessment tools and interpretation of results.
Government leaders and decision-makers needed knowledge about how to access, use, and interpret PROM and PREM information in their decision-making. One government leader said, The dream for me is that it is useful for being able to direct resources to those areas that patients are telling us is most needed, rather than something for clinicians or administrators to think they know where the resources should go. But the dream is that we’re making these kinds of decisions not just from behind the desk but in partnership with patients. They expressed concern regarding the use of different tools across organizations, costs or resources, and time-lags between collection and reporting. Government leaders and decision-makers recommended that the KT resources offer examples of use in decision-making, support interpretation of data, be concise but also come alive through stories, and be tailored to their scope of practice.
Phase 2: Development of tailored KT resources
Twenty-three participants took part in the Formative Evaluation including 4 patients, 5 family caregivers, 4 healthcare providers, 6 healthcare managers and leaders, and 4 government leaders and decision-makers. All but 3 of them also participated in the Initial Consultation. (See Table 1 for participant characteristics.)
Description of initial KT resources
There was a need for simple, introductory didactic resources on QOL assessments tailored specifically to the context and needs for each group. In various ways, each group asked, “what’s in it for me?” Table 3 outlines the KT resources that addressed this question, developed collaboratively with members of our KT team, while drawing on evidence-based sources.
Brochures were tailored to address the knowledge gaps of each audience. For older adults and family caregivers, brochures offered information about the use of QOL assessment tools to live their best life possible. Brochures for healthcare providers were designed not only to help them incorporate QOL assessments into their interactions with older adults and family caregivers, but also to address common myths and misconceptions about the use of QOL assessment tools. For healthcare managers and leaders, the brochures were designed to support discussions about the importance and use of QOL assessments in their organizations for person-centered care and quality improvement. The brochures for government leaders and decision-makers included a “fact-sheet” and “frequently asked questions” focused on the use of evidence-based QOL assessment tools and data to monitor performance, improve quality, and make policy and budget decisions regarding healthcare for older adults and their family caregivers.
Several resources were created that spanned participant groups. One whiteboard animation (2 min) introduced QOL assessment tools and use of this information in micro-level decision-making. A live-action video (6 min) depicted a case study of a father and a son who is a caregiver, using QOL assessment tools to inform their relationship and improve decision-making about care. Statements in each KT resource were referenced with evidence-based sources.
Formative evaluation to refine KT resources
Formative evaluation of the KT resources identified an overall positive response by all four participant groups, and all offered suggestions for refinement. See Table 4 for participants’ comments, suggested changes (with exemplar quotes) to each KT resource, and revisions made.
Looking across the groups, participants discussed potential benefits, perceived relevance, and usefulness of the KT resources. For example, one family caregiver appreciated the encouragement: I like the repeating of the phrase ‘living your best life possible,’ because some days that doesn’t look very good for a lot of people. Healthcare providers saw the linkage between completion of QOL assessment tools and follow-up discussions which may include the family. One explained: It’s not focusing just on the client, but it’s also pulling in the family caregiver or the primary caregiver, which I think is really important if we’re going to be looking at a sort of patient-centred, family-centred care. A healthcare leader perceived benefits of the KT resources at a meso-level: Yes, I think it is speaking to us in the language that we understand around performance, accountability, all those things. I think that makes sense to me. A government leader also perceived the value of QOL assessment data alongside other data used in decision-making: I think you’re getting at kind of the critical ones [information], which is ‘what is the value of-’ or ‘not the value of-’, and this kind of data ‘holds up’ next to administrative-level data or clinical-level data, which I think is very often kind of dismissed.
Participants described what they liked about the KT resources, including ease of use, multiple modalities, and comprehensiveness. One family caregiver said, It’s standard English, and there were no large words, ‘onomatopoeia’ or something like that! Many participants across groups appreciated the multi-media approaches. A healthcare provider explained, You’ve got a webpage, you’ve got some pamphlets, and you’ve got a variety of methods. And I think that’s really important, so I do really appreciate that. Because we know that when we’re trying to reach out and embed some new ways of working in our organization, that you need a lot of different resources…to support that. I appreciate the fact there’s sort of a multimodal approach. Many participants commented on the simplicity of the messages. One government leader said, Very clear, very crisp. I like the tagline, the mnemonic. I think having a fact sheet and a Q&A are pretty complementary, so I think that’s all there.
Participants also offered recommendations for refinement. Older adults and family asked that the reading level be lowered and messages be simplified. As one older adult said, I’m trying to put myself in the shoes…of somebody who has a Grade 8 reading level. Some words/phrases like “enhance,” “healthcare team,” or “support system” were too academic, and were clarified. Both healthcare providers and leaders emphasized the need for references to evidence-based sources, and suggested that less is more, meaning that content be condensed so they could read/skim it quickly. One healthcare manager said, I think they could be ‘conciser’…what I find is the more that you go ‘up’ the organization, the simpler things have to get because the–what do you call it–the ‘bandwidth’ is very narrow. Government leaders and decision-makers recommended various edits to avoid healthcare jargon and abstract language. One said, Have one more layer on ‘how.’ It’s still a bit theoretical, still a bit academic. A few participants found the layout or colors not appealing, visuals not sufficient, or the font size too small. Participants suggested revisions to the whiteboard and live-action video. Refinements included slowing down the pace of both videos, adding tag lines or images to help with flow, and reducing the length. The KT resources were subsequently refined based on the formative feedback and final versions were made available to all participants and freely online at www.healthyqol.com/older-adults.
Discussion
This study was motivated by a need for KT resources that addressed knowledge gaps of micro-, meso-, and macro-audiences regarding the use of QOL assessment tools for older adults and family caregivers. Our results confirmed the need for introductory resources, and the importance of tailoring these resources to specifically address the knowledge gaps of different audiences. Older adults and family caregivers wanted to understand how their QOL information could be collected, reported, and used to improve their care. Healthcare providers emphasized the need for practical information on how to integrate and use QOL assessment tools in their practice. Healthcare managers and leaders focused on information about using standardized tools, like PROMs and PREMs, for care decisions and quality improvement in their organizations. Government leaders and decision-makers required evidence-based information on using PROMs and PREMs for macro-level purposes to monitor performance, improve quality of care, and make budget decision in healthcare systems. Our project serves as an exemplar of how such information can be used to develop introductory KT resources tailored for different audiences.
There are various other evidence-based resources available to support the use of QOL assessment tools in healthcare [28,29,30,31,32,33]. Notable examples include a user’s guide on patient-reported outcomes in clinical practice by the International Society of Quality of Life Research [28, 46, 47], and guidelines for the use of electronic patient-reported outcomes [32, 48]. Additional guidance is provided by a range of theories and systematic reviews on QOL assessment [8, 14, 15, 49,50,51,52]. These resources provide invaluable, detailed, evidence-based information for healthcare providers and organizations motivated to use QOL assessments. Our previous work [26, 27, 53] identified that for those user groups who were not yet convinced of their importance, there was a need to develop KT resources to introduce QOL assessment and “what’s in it for me?” For QOL assessment tools to be used, it is important that users first have a basic understanding of their value and potential use for different purposes. Our project addressed this need by specifically focusing on the development of tailored introductory resources through engagement with patients, clinicians, and decision-makers.
In addition to developing tailored content, it is also important to develop resources that use language and modality that is familiar, supportive, and accessible to the different audiences. To achieve this, working with a language specialist and graphic designer may be required. With respect to language, it is important for reading levels to match the audience. For example, for patients and family caregivers, a Grade 6 reading level (or below) is generally recommended [54,55,56]. In addition, different audiences are familiar with different terminology, designs, and formats. For example, healthcare leaders and decision-makers may not be familiar with the term “QOL assessment” and more commonly use PROMs and PREMs, whereas the terms “QOL” and “assessment” may be more familiar to patients, family caregivers, and clinicians. Government leaders may be more familiar with short briefing notes, whereas conventional continuing education formats (e.g., manuscripts) are more familiar to clinicians. Accessibility to patients and family caregivers could be enhanced by providing materials both in written and audiovisual formats.
While our work was guided by a well-established KT framework [34, 35], there are limitations. First, we had limited diversity among participants, thus various ethnic groups, geographies (e.g., remote), and living arrangements (e.g., multigenerational households and structure) may not be sufficiently attended to in our KT resources. Second, we did not fully address the question of how to develop KT resources. The current project may serve as an exemplar for a further project in this area with specific emphasis on further tailoring of resources for patients, family caregivers, and healthcare providers with diverse backgrounds and other healthcare systems. In so doing, we particularly recommend studies in different languages, healthcare systems, and diverse populations. Third, our work did not entail summative evaluation for end-of-project evaluation of the KT resources, which may be taken up in future work.
Conclusion
Through this study, we offer three important contributions to the field. First, our findings identify that knowledge gaps of micro-, meso-, and macro-audiences regarding QOL assessment are distinct and their needs must be addressed. Second, our results affirm the need to tailor evidence-based KT resources to address knowledge gaps that may hinder different audiences’ use of QOL assessment tools. Third, while evidence-based information is available for those already motivated to use QOL assessment tools, through an integrative KT approach we produced tailored, introductory KT resources for those who may still be asking: “what’s in it for me?” Our study exemplifies engaging patients, family caregivers, clinicians, and decision-makers in developing such resources to address their unique knowledge gaps and support the use of QOL assessment tools for older adults and family caregivers.
Data availability
Ethical approval was not attained for the purposes of public sharing of the data. Scientists who are interested in using the data may contact the corresponding author for further information.
Code availability
Not applicable.
References
Kojima, G., Iliffe, S., Jivraj, S., & Walters, K. (2016). Association between frailty and quality of life among community-dwelling older people: A systematic review and meta-analysis. Journal of Epidemiology & Community Health, 70(7), 716–721.
Crocker, T. F., Brown, L., Clegg, A., Farley, K., Franklin, M., Simpkins, S., & Young, J. (2019). Quality of life is substantially worse for community-dwelling older people living with frailty: Systematic review and meta-analysis. Quality of Life Research, 28(8), 2041–2056.
Schulz, R., & Beach, S. R. (1999). Caregiving as a risk factor for mortality: The caregiver health effects study. Journal of the American Medical Association, 282(23), 2215–2219.
Funk, L., Stajduhar, K. I., Toye, C., Aoun, S., Grande, G. E., & Todd, C. J. (2010). Part 2: Home-based family caregiving at the end of life: A comprehensive review of published qualitative research (1998–2008). Palliative Medicine, 24(6), 594–607.
Stajduhar, K. I., Funk, L., Toye, C., Grande, G. E., Aoun, S., & Todd, C. J. (2010). Part 1: Home-based family caregiving at the end of life: A comprehensive review of published quantitative research (1998–2008). Palliative Medicine, 24(6), 573–593.
The WHO Group. (1995). The World Health Organization quality of life assessment (WHOQOL): Position paper from the World Health Organization. Social Science & Medicine, 41(10), 1403–1409.
Sawatzky, R., & Cohen, S. R. (2020). Quality of life assessment in palliative care. In E. Bruera, I. Higginson, C. F. von Gunten, & T. Morita (Eds.), Textbook of palliative medicine and supportive care (pp. 189–198). CRC Press.
Schick-Makaroff, K., Karimi-Dehkordi, M., Cuthbertson, L., Dixon, D., Cohen, S. R., Hilliard, N., & Sawatzky, R. (2021). Using patient- and family-reported outcome and experience measures across transitions of care for frail older adults living at home: A meta-narrative synthesis. The Gerontologist, 61(3), e23–e38.
World Health Organization. (2015). The growing need for home health care for the elderly Home health care for the elderly as an integral part of primary health care services. World Health Organization, Regional Office for the Eastern Mediterranean.
Johnson, S., Bacsu, J., Abeykoon, H., McIntosh, T., Jeffery, B., & Novik, N. (2018). No place like home: A systematic review of home care for older adults in Canada. Canadian Journal on Aging/La Revue canadienne du vieillissement, 37(4), 400–419.
Canadian Home Care Association. (2013). Portraits of home care in Canada. Canadian Home Care Association.
Hakansson Eklund, J., Holmstrom, I. K., Kumlin, T., Kaminsky, E., Skoglund, K., Hoglander, J., Sundler, A. J., Conden, E., & Summer Meranius, M. (2019). “Same same or different?” A review of reviews of person-centered and patient-centered care. Patient Education and Counseling, 102, 3–11.
Dion, A., Joseph, L., Jimenez, V., Gutierrez, A. C., Ben Ameur, A., Robert, E., & Andersson, N. (2019). Grounding evidence in experience to support people-centered health services. International Journal of Public Health, 64(5), 797–802.
Greenhalgh, J., Dalkin, S., Gibbons, E., Wright, J., Valderas, J. M., Meads, D., & Black, N. (2018). How do aggregated patient-reported outcome measures data stimulate health care improvement? A realist synthesis. Journal of Health Services Research & Policy, 23(1), 57–65.
Greenhalgh, J., Gooding, K., Gibbons, E., Dalkin, S., Wright, J., Valderas, J., & Black, N. (2018). How do patient reported outcome measures (PROMs) support clinician-patient communication and patient care? A realist synthesis. Journal of Patient-Reported Outcomes, 2(1), 42.
Sutherland, H. J., & Till, J. E. (1993). Quality-of-life assessments and levels of decision-making: Differentiating objectives. Quality of Life Research, 2(4), 297–303.
Öhlén, J., Reimer-Kirkham, S., Astle, B., Håkanson, C., Lee, J., Eriksson, M., & Sawatzky, R. (2017). Person-centred care dialectics - Inquired in the context of palliative care. Nursing Philosophy, 18(4), e12177.
Marshall, S., Haywood, K., & Fitzpatrick, R. (2006). Impact of patient-reported outcome measures on routine practice: A structured review. Journal of Evaluation in Clinical Practice, 12(5), 559–568.
Catania, G., Beccaro, M., Costantini, M., Ugolini, D., De Silvestri, A., Bagnasco, A., & Sasso, L. (2015). Effectiveness of complex interventions focused on quality-of-life assessment to improve palliative care patients’ outcomes: A systematic review. Palliative Medicine, 29(1), 5–21.
Bowling, A. (2007). Ageing well - Quality of life in old age.
Haywood, K. L., Garratt, A. M., & Fitzpatrick, R. (2006). Quality of life in older people: A structured review of self-assessed health instruments. Expert Review of Pharmacoeconomics & Outcomes Research, 6(2), 181–194.
Van Der Wees, P. J., Nijhuis-Van Der Sanden, M. W., Ayanian, J. Z., Black, N., Westert, G. P., & Schneider, E. C. (2014). Integrating the use of patient-reported outcomes for both clinical practice and performance measurement: Views of experts from 3 countries. The Milbank Quarterly, 92(4), 754–775.
Black, N., Burke, L., Forrest, C. B., Ravens Sieberer, U. H., Ahmed, S., Valderas, J. M., Bartlett, S. J., & Alonso, J. (2016). Patient-reported outcomes: Pathways to better health, better services, and better societies. Quality of Life Research, 25(5), 1103–1112.
Devlin, N. J., & Appleby, J. (2010). Getting the most out of proms. Putting health outcomes at the heart of NHS decision-making: The King's Fund.
Antunes, B., Harding, R., & Higginson, I. J. (2014). Implementing patient-reported outcome measures in palliative care clinical practice: A systematic review of facilitators and barriers. Palliative Medicine, 28(2), 158–175.
Sawatzky, R., Laforest, E., Schick-Makaroff, K., Stajduhar, K., Reimer-Kirkham, S., Krawczyk, M., Öhlén, J., McLeod, B., Hilliard, N., Tayler, C., & Cohen, S. R. (2018). Design and introduction of a quality of life assessment and practice support system: Perspectives from palliative care settings. Journal of Patient-Reported Outcomes, 2(1), 1–13.
Krawczyk, M., Sawatzky, R., Schick-Makaroff, K., Stajduhar, K., Öhlen, J., Reimer-Kirkham, S., Mercedes Laforest, E., & Cohen, R. (2019). Micro–meso–macro practice tensions in using patient-reported outcome and experience measures in hospital palliative care. Qualitative Health Research, 29(4), 510–521.
Snyder, C. F., Aaronson, N. K., Choucair, A. K., Elliott, T. E., Greenhalgh, J., Halyard, M. Y., Hess, R., Miller, D. M., Reeve, B. B., & Santana, M. (2012). Implementing patient-reported outcomes assessment in clinical practice: A review of the options and considerations. Quality of Life Research, 21(8), 1305–1314.
Skovlund, P. C., Ravn, S., Seibaek, L., Thaysen, H. V., Lomborg, K., & Nielsen, B. K. (2020). The development of PROmunication: A training-tool for clinicians using patient-reported outcomes to promote patient-centred communication in clinical cancer settings. Journal of Patient-Reported Outcomes, 4(1), 10.
Snyder, C., Brundage, M., Rivera, Y. M., & Wu, A. W. (2019). A PRO-cision medicine methods toolkit to address the challenges of personalizing cancer care using patient-reported outcomes: Introduction to the supplement. Medical Care, 57, S1–S7.
Wehrlen, L., Krumlauf, M., Ness, E., Maloof, D., & Bevans, M. (2016). Systematic collection of patient reported outcome research data: A checklist for clinical research professionals. Contemporary Clinical Trials, 48, 21–29.
LeRouge, C., Austin, E., Lee, J., Segal, C., Sangameswaran, S., Hartzler, A., Lober, W. B., Heim, J., & Lavallee, D. C. (2020). ePROs in clinical care: Guidelines and tools for health systems. CERTAIN, University of Washington.
Healthcare Improvement Scotland. (2012). PROMs (Patient Reported Outcome Measures) user guide. http://www.healthcareimprovementscotland.org/our_work/person-centred_care/proms_questionnaire_project/proms_user_guide.aspx.
Graham, I. D., Logan, J., Harrison, M. B., Straus, S. E., Tetroe, J., Caswell, W., & Robinson, N. (2006). Lost in knowledge translation: Time for a map? The Journal of Continuing Education in the Health Professions, 26(1), 13–24.
Graham, I. D., & Tetroe, J. M. (2010). The knowledge to action framework. In J. RycroftMalone & T. Bucknall (Eds.), Models and frameworks for implementing evidence-based practice: Linking evidence to action (pp. 207–221). Wiley-Blackwell.
Neergaard, M. A., Olesen, F., Andersen, R. S., & Sondergaard, J. (2009). Qualitative description – The poor cousin of health research? BMC Medical Research Methodology, 9(52), 1–5.
Sandelowski, M. (2000). Whatever happened to qualitative description? Research in Nursing & Health, 23(4), 334–340.
Sawatzky, R. (2016). Quality of life assessment and practice support system in home care services for older adults. Identifier NCT02940951, from https://clinicaltrials.gov/ct2/show/NCT02940951?term=Sawatzky&draw=2&rank=1
Hartrick Doane, G., Reimer-Kirkham, S., Antifeau, E., & Stajduhar, K. (2015). (Re)theorizing integrated knowledge translation: A heuristic for knowledge-as-action. Advances in Nursing Science, 38(3), 175–186.
Morse, J. M., Swanson, J. M., & Kuzel, A. J. (2000). The nature of qualitative evidence. SAGE Publications.
Hennink, M. M., Kaiser, B. N., & Marconi, V. C. (2017). Code saturation versus meaning saturation: How many interviews are enough? Qualitative Health Research, 27(4), 591–608.
Hennink, M. M., Kaiser, B. N., & Weber, M. B. (2019). What influences saturation? Estimating sample sizes in focus group research. Qualitative Health Research, 29(10), 1483–1496.
Stetler, C. B., Legro, M. W., Wallace, C. M., Bowman, C., Guihan, M., Hagedorn, H., Kimmel, B., Sharp, N. D., & Smith, J. L. (2006). The role of formative evaluation in implementation research and the QUERI experience. Journal of General Internal Medicine, 21, S1–S8.
Denzin, N. K., & Lincoln, Y. S. (2018). The SAGE handbook of qualitative research (5th ed.). Sage.
Tong, A., Sainsbury, P., & Craig, J. (2007). Consolidated criteria for reporting qualitative research (COREQ): A 32-item checklist for interviews and focus groups. International Journal for Quality in Health Care, 19(6), 349–357.
International Society of Quality of Life, prepared by Aaronson, N., Elliott, T. E., Greenhalgh, J., Halyard, M. Y., Hess, R., Miller, D. M., Reeve, B., Santana, M., & Snyder, C. (2015). User’s guide to implementing patient-reported outcomes assessment in clinical practice, Version: January 2015. International Society for Quality of Life Research.
International Society for Quality of Life Research, prepared by Chan, E., Edwards, T., Haywood, K., Mikles, S., & Newton, L. (2018). Companion guide to implementing patient reported outcomes assessment in clinical practice, Version: February 2018. International Society for Quality of Life Research.
Tavares, A. P. D. S., Paparelli, C., Kishimoto, C. S., Cortizo, S. A., Ebina, K., Braz, M. S., Mazutti, S. R. G., Arruda, M. J. C., & Antunes, B. (2017). Implementing a patient-centred outcome measure in daily routine in a specialist palliative care inpatient hospital unit: An observational study. Palliative Medicine, 31(3), 275–282.
Greenhalgh, J. (2009). The applications of PROs in clinical practice: What are they, do they work, and why? Quality of Life Research, 18(1), 115–123.
Valderas, J. M., & Alonso, J. (2008). Patient reported outcome measures: A model-based classification system for research and clinical practice. Quality of Life Research, 17(9), 1125–1135.
Santana, M.-J., & Feeny, D. (2014). Framework to assess the effects of using patient-reported outcome measures in chronic care management. Quality of Life Research, 23(5), 1505–1513.
Porter, I., Goncalves-Bradley, D., Ricci-Cabello, I., Gibbons, C., Gangannagaripalli, J., Fitzpatrick, R., Black, N., Greenhalgh, J., & Valderas, J. M. (2016). Framework and guidance for implementing patient-reported outcomes in clinical practice: Evidence, challenges and opportunities. Journal of Comparative Effectiveness Research, 5(5), 507–519.
Schick-Makaroff, K., & Sawatzky, R. (2020). Divergent perspectives on the use of the Edmonton Symptom Assessment System (Revised) in palliative care. Journal of Hospice and Palliative Nursing, 22(1), 75–81.
van Ballegooie, C., & Hoang, P. (2021). Assessment of the readability of online patient education material from major geriatric associations. Journal of the American Geriatrics Society, 69(4), 1051–1056.
Wittink, H., & Oosterhaven, J. (2018). Patient education and health literacy. Musculoskeletal Science & Practice, 38, 120–127.
Parnell, T. A. (2015). Health literacy in nursing: Providing person-centered care: Springer
Acknowledgements
The team acknowledges contributions to the project by Shelly Cory, Bruce Forde, Neil Hilliard, Melissa Kuo, Esther Laforest, Mary Luk (Patient Partner), and Alies Maybee (Patient Partner). Specifically, the team acknowledges Danielle Judd, Theo Fletcher, and Chung Liu for facilitating participant recruiting, and/or supporting data analysis and report writing. We also acknowledge Laurie Culshaw (designbylkc.com) and Nancy Burgess (nancyburgess.net) for their contributions to design and copyediting of the KT resources.
Funding
Open access funding provided by University of Gothenburg. This work was funded by Canadian Frailty Network, 2018 Catalyst Program. (Grant # CAT2018-17), with matching funds provided by Fraser Health Authority, Cambian Business Services, Office of Patient-Centred Measurement of the BC Ministry of Health, University of Alberta, and Trinity Western University. KSM is a New Investigator funded by CIHR Can-SOLVE CKD – KRESCENT (Canadians Seeking Solutions & Innovations to Overcome Chronic Kidney Disease; Kidney Research Scientist Core Education & National Training Program). This research was undertaken, in part, thanks to funding from the Canada Research Chairs (CRC) Program supporting Dr. Sawatzky’s CRC in Person-Centred Outcomes at Trinity Western University.
Author information
Authors and Affiliations
Contributions
RS, KS-M, and LC co-designed and co-lead the study. RS and KS-M co-lead the analysis, drafting, and revisions of the manuscript. AB, DD, and NS supported data analysis. All authors contributed to aspects of study implementation, interpretation of the analysis, draft writing, and revisions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors LC, JO, MKD, KS, LT, AW, and RC declare that they have no conflicts of interest. KSM declares that this research was undertaken, in part, thanks to her New Investigator funding from by CIHR Can-SOLVE CKD—KRESCENT (Canadians Seeking Solutions & Innovations to Overcome Chronic Kidney Disease; Kidney Research Scientist Core Education & National Training Program) from 2016 to 2019. RS declares that this research was undertaken, in part, thanks to funding from the Canada Research Chairs Program supporting Sawatzky’s Chair in Person-Centred Outcomes held at Trinity Western University. RS and KSM declare that Cambian Business Services (www.cambian.com), Fraser Health Authority, and the British Columbia Office of Patient- Centred Measurement were project partners who may benefit from results supporting the use of QOL assessment tools. AB and DD declare that they received payment as Canadian Frailty Network Trainees named on the project. AB declares that she received payment as a research assistant for helping to write and review the manuscript. DD declares that she was employed as a nurse by the same organization and health authority as some of the healthcare managers and leaders who were participants in the study. NS declares that she completed her contract on this project in January 2020, and hence worked as an independent contractor after office hours and during the weekends to work on the manuscript tables.
Ethical approval
Ethics approval for the study was obtained from Trinity Western University [TWU18F08]; University of Alberta [PRO00085430]; Harmonized ethical review by University of British Columbia, Providence Health Care, and Fraser Health [H18-02191]; and the McGill University Institutional Review Board [#A04-E31-19B].
Consent to participate
Informed consent was obtained from all individual participants included in this study.
Consent to publication
Participants’ informed consents included publication of their data as long as they could not be identified.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Schick-Makaroff, K., Sawatzky, R., Cuthbertson, L. et al. Knowledge translation resources to support the use of quality of life assessment tools for the care of older adults living at home and their family caregivers. Qual Life Res 31, 1727–1747 (2022). https://doi.org/10.1007/s11136-021-03011-z
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11136-021-03011-z