Abstract
This study introduces the concept of developing a functional hemp drink enriched with γ-Aminobutyric acid (GABA) to enhance its nutritional value and functional properties utilizing Solid-State (SSF) co-Fermentation by Lactobacillus casei and Bacillus subtilis and germination bioprocesses. Bioprocesses may offer an alternative solution to challenges in hemp milk, such as product instability and the use of additives. Notably, the hemp milk produced through the germination for three days or co-fermentation processes yielded the highest GABA content of 79.84 and 102.45 mg/100 mL, respectively, compared to the untreated milk. These bioactive milk samples exhibited higher zeta potential and soluble protein content and also reduced solid particle sedimentation and droplet sizes (D4,3 and D3,2) compared to the untreated milk. Furthermore, the peptide, total phenolic content, and antioxidant activity of the produced GABA-enriched kinds of milk surpassed those of the untreated milk. Overall, the SSF and germination processes present a promising alternative for producing stable milk analogs with enhanced health-boosting properties.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
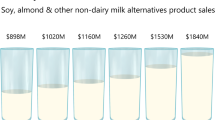
Nutraceutical and functional components, such as γ-aminobutyric acid (GABA), are experiencing rapid growth in the food industry. This growth is especially prevalent in the beverage sector, where bioactive compounds are incorporated into products [1]. This trend has led to the expansion of non-dairy milk substitutes like soy, hemp, almond, and coconut milk in the global milk alternatives market. The popularity of non-dairy milk substitutes, which is expected to reach US$123.1 billion by 2030, can be attributed to various factors, including ethical, environmental, and animal welfare concerns [2]. γ-aminobutyric acid is a naturally occurring compound that can positively impact mood, cognitive function, relaxation, and sleep quality [3, 4]. Natural GABA can be produced through microbial fermentation by probiotic strains and germination, offering a safer and more environmentally friendly alternative to synthetic GABA production [5]. While many studies have used single strains of lactic acid bacteria (LAB) to produce GABA, there are limited reports on co-culturing for GABA production, indicating the potential of this approach [6, 7].
Hemp seeds, sourced from Cannabis sativa L., are a rich source of essential nutrients (25% protein and 30% oil), amino acids, and beneficial fatty acids, making them a promising alternative to traditional dairy, soy, and nut-based milk substitutes [8]. Recent research has revealed various health benefits of hemp seed consumption, including immune system regulation, neuroprotection, and cardiovascular benefits [9]. Hemp seeds also contain compounds like cannabidiolic acid (CBDA) and arginine, which may protect against chronic diseases [10]. Additionally, the high glutamate content in hemp seeds makes them suitable for increasing GABA levels through fermentation and germination.
Hemp milk is a popular and convenient plant-based beverage with a nutrient profile similar to cow’s milk but without cholesterol, gluten, or lactose. Despite its benefits, hemp milk faces stability challenges related to its oil-in-water emulsion [11]. Bioprocesses, including solid-state fermentation and germination, have emerged as promising alternatives to synthetic stabilizers, offering cost-effectiveness, environmental friendliness, safety, sustainability, and ease of implementation [12, 13]. Hemp seeds contain prebiotic ingredients such as oligosaccharides, fibers, polyphenols, and isoflavones. Solid-state fermentation aids in the partial breakdown of organic substances, such as complex protein and protein-phenol structures, yielding smaller, soluble forms [11]. The primary objectives of this study were to compare the impact of fermentation utilizing probiotic strains, including Lactobacillus casei and Bacillus subtilis, and their co-cultures and germination on GABA and antioxidant production, evaluate the effect of solid-state co-fermentation, and investigate the influence of bioprocesses on the physical properties of hemp milk (Fig. 1a). This is the first attempt to examine the GABA content in hemp seeds and their fermented and germinated derivates.
(a) The schematic diagram of hemp milk production from germinated, solid-state fermented, and untreated seeds and (b) summary of the GABA biosynthesis pathway. U: untreated; G3: germinated for 3 days; FBL: solid state fermented by co-cultures of. L. casei and B. subtilis, GABA: γ-aminobutyric acid; GAD: glutamic acid decarboxylase; GDH: glutamic acid dehydrogenase; TCA: tricarboxylic acid; SSADH: succinate-semialdehyde decarboxylase; GABA-AT: GABA transaminase
Materials and Methods
The section on materials and methods is shown in Supplementary File S1.
Results and Discussion
GABA Content
In solid-state fermentation using L. casei, B. subtilis, and their co-cultures on hemp seeds, higher GABA contents were observed compared to germinated seeds for up to 3 days (Table 1- see in S2). The FBL seeds that co-fermented with L. casei and B. subtilis displayed the highest GABA content at 175.98 mg/100 g dry weight (dw), surpassing untreated seeds by 10.28 times. FB seeds had a GABA content of 152.38 mg/100 g dw, and FL seeds had a GABA content of 128.51 mg/100 g dw, representing 8.90 and 7.51 times higher GABA levels than untreated seeds, respectively. Through solid-state fermentation using single and co-cultures of L. casei and B. subtilis, as well as germination for up to 3 days, the GABA content in hemp milk formulations ranged from 14.08 to 102.45 mg/100 mL (Table 1- see in S2). As seen in Fig. 1b, GABA synthesis from L-glutamate is facilitated by the GAD enzyme, which various microorganisms, including LAB, yeasts, and fungi, can produce. The synthesis of GABA occurs via the enzymatic decarboxylation of L-glutamate facilitated by glutamic acid decarboxylase (GAD). The GAD is formed from α-ketoglutaric acid in the tricarboxylic acid (TCA) cycle through the action of glutamic acid dehydrogenase (GDH) [14,15,16]. The amount of glutamic acid in the raw products influences the GABA content in sprouts. The increased GABA content observed during seed germination can be attributed to the breakdown of seed-storage compounds and the synthesis of structural proteins and other cellular components necessary for plant growth. In higher plants, GABA biosynthesis primarily occurs through the GABA shunt pathway, with the polyamine degradation pathway playing a secondary role. The GABA shunt pathway involves the decarboxylation of glutamate to produce GABA, catalyzed by GAD. GABA is subsequently converted to succinate semialdehyde by GABA transaminase (GABA-AT). Succinate semialdehyde is further oxidized by succinate semialdehyde decarboxylase (SSADH) to form succinic acid, which enters the tricarboxylic acid cycle (TCA cycle) to provide a carbon skeleton for ATP synthesis in plants [20]. The synthesis of GABA from L-glutamate in metabolism is facilitated by the GAD enzyme, which can be produced by various microorganisms such as LAB, yeasts, and fungi [34]. Previous studies have reported GABA production levels of 50.4 mg/kg for L. casei, and 15.40 mg/mL for B. subtilis [17, 18]. In the co-culture fermentation of L. casei and B. subtilis, there appears to be a synergistic enhancement in GABA bioconversion compared to their individual fermentations. Initially, B. subtilis dominated the fermentation of hemp seeds. Still, as conditions became acidic and anaerobic, L. casei became the predominant microorganism, a condition known to increase GABA production in species like L. brevis and L. plantarum. Thus, the anaerobic conditions and a limited carbon source likely facilitated L-glutamate conversion to GABA [19]. Recent studies on co-culture fermentation and GABA production support these findings, suggesting that co-culture solid-state fermentation holds promise for GABA generation [20,21,22].
Zeta Potential and Droplet Sizes
Figure 2a and b show the zeta potential and droplet sizes of hemp milk samples. The FBL milk exhibited the highest zeta potential values among the milk samples, negatively increasing to - 42.68 mV. Compared to the untreated milk sample (-29.31 mV), the G3 (-35.2 mV) and FBL milk samples had zeta potentials that were 17 and 31% higher, respectively. Zeta potential values exceeding ±25 mV commonly indicate improved colloidal stability. High values suggest electrostatic solid repulsion between particles, effectively preventing their aggregation and sedimentation within the colloidal suspension [23]. Zeta potential measurements aligned with particle size distribution data, showing that both D3,2 and D4,3 values in the G3 and FBL samples were lower than in the untreated hemp milk. The FBL sample had the lowest D3,2 and D4,3 values, with a reduction of 9.4% and 7.1% compared to the untreated sample. Hemp milk’s high-fat macromolecules and globular edestin protein with high crystallinity contribute to low solubility and protein-protein aggregation [24]. Solid-state fermentation and germination processes enzymatically break down these macromolecules, forming more minor, more soluble subunits [25].
(a) Zeta potential, (b) droplet sizes (D3,2, D4,3), (c) solid particle sedimentation, (d) brix, (e) free -SH group content (µmol/g protein), and (f) soluble protein content (%) of hemp milk samples. U: untreated; G3: germinated for 3 days; FBL: solid state fermented by co-cultures of. L. casei and B. subtilis
Solid Particle Sedimentation and ºBrix
Figure 2c demonstrates a slight reduction in solid particle sedimentation in the FBL and G3 milk samples compared to the untreated milk, leading to improved physical stability. Bioprocessing methods effectively mitigate the tendency of macroaggregates to coalesce, resulting in a reduction in particle size. This leads to the formation of smaller protein aggregates with surface-active properties, enhancing the development of a more efficient film layer around oil droplets and, consequently, bolstering emulsion stability [26]. The noticeable increase in the ºBrix measurement of the FBL sample, compared to both the untreated and G3 milk samples, may be attributed to improved solubility (Fig. 2d). The presence of exopolysaccharides generated through bacterial fermentation and carbohydrates decomposed during the germination process may contribute to increased viscosity in hemp milk, resulting to a more stable product. Consequently, bioprocessing methods are a viable and cost-effective alternative to synthetic stabilizers.
Free Sulfhydryl (-SH) Group Content
Thiol, which contains free -SH functional groups, is an essential antioxidant in preventing oxidative stress-induced damage and protects cells against oxidative stress [19]. As illustrated in Fig. 2e, the FBL milk samples exhibited a higher concentration of free -SH groups than the untreated and G3 samples. The observed phenomenon can be attributed to the activation of endogenous proteases during the fermentation and germination process. These proteases, secreted by microorganisms, enzymatically cleave the peptide bonds within the protein, resulting in the unfolding of the polypeptide chain and subsequent disruption of the disulfide bonds associated with the active sulfhydryl groups situated within the protein molecule [27]. These processes lead to an increase in the abundance of free sulfhydryl groups.
Soluble Protein Content
Figure 2f shows the influence of the SSF and germination bioprocesses on the protein solubility of hemp milk samples. As depicted, the FBL and G3 milk samples exhibited higher protein solubility of 3.00 and 2.25 g/100 mL, respectively, a 38 and 25% increase compared to the untreated milk sample. Biotechnological processes reduced particle size, as seen in Fig. 2b, leading to increased surface charge on proteins, including hydrophilic regions. This enhanced surface charge can promote converting insoluble protein forms into soluble forms through hydrolysis and partial denaturation [28]. Amino acid transformation by de novo synthesis and increased hydrophilic amino acid content [27], such as lysine, during bioprocesses, can improve the water affinity of protein isolates, leading to increased solubility.
Peptide Content
The peptide content of hemp milk samples is depicted in Fig. 3a. The bioprocesses of the SSF and germination resulted in increased peptide content within the milk samples. Specifically, the G3 and FBL milk samples exhibited peptide content that was 9.3 and 11.7 times higher, respectively, compared to the untreated milk. Germination is a physiological process that stimulates the disintegration of plant cell structures and the hydrolysis of proteins through activating enzymes, including hydrolases, proteases, and peptidases [27]. This intricate process entails the mobilization of storage proteins via endopeptidases and carboxypeptidases, leading to the breakdown of proteins into amino acids to serve as an energy source [26]. Consequently, the proteins undergo hydrolysis, forming low molecular weight amino acids and peptides, thereby augmenting protein solubility. Likewise, bacterial proteolytic enzymes from co-cultures of L. casei and B. subtilis during solid-state fermentation facilitate the degradation of large protein aggregates into peptides. Both germination and fermentation processes involve the activation of hydrolytic enzymes to facilitate protein degradation and release smaller peptide constituents [29, 30]. The proteolytic activity exhibited by Lactobacillus spp. enables the enzymatic degradation of proteins into specific bioactive peptides that display inhibitory properties against α-amylase and α-glucosidase [31]. Numerous studies have reported on the antioxidant and anti-hypertensive attributes of hemp seed peptides and protein hydrolysate [32]. Therefore, the field of bioprocess engineering is experiencing a surge in interest regarding biologically active peptides derived from proteins.
Total Phenolic Content and Antioxidant Activity
The total phenolic content of hemp milk samples was measured, and the results showed that the FBL sample exhibited the highest phenolic content, with an impressive increase of over 75% compared to the untreated milk sample (Fig. 3b and c). The G3 milk sample also displayed a substantial increase, with a 53% rise in phenolic content. Additionally, the DPPH reducing activity (%) significantly increased in the FBL and G3 milk samples, with enhancements of 92.97 and 66.40%, respectively, compared to the untreated milk samples. Several factors contribute to the increase in total phenolic content and antioxidant activities during germination and fermentation processes. Bioprocessing techniques like germination and fermentation break down protein-phenolic complexes, converting bound phenolic compounds into free forms through proteolytic activities [33, 34]. Fermentation, driven by β-glucosidase enzymes, further hydrolyzes phenolic-glucosides and phenolic-protein conjugates, increasing free phenolic concentrations [27, 35]. These processes generate essential intermediate compounds for phenolic biosynthesis [36]. Microbial enzymatic activity and metabolic processes can create new phenolic compounds facilitated by changes in pH and distribution of amino acids [25, 36]. Amino acids with antioxidant properties, such as histidine, tyrosine, methionine, and cysteine, correlate positively with increased antioxidant activity. Various enzymatic processes catalyze proteolysis, leading to higher levels of potentially bioactive peptides and active polyphenolic compounds, enhancing antioxidant activity [8].
Conclusions
This study utilized the solid-state fermentation and germination processes to enhance the nutritional value and functionality of hemp milk, resulting in a GABA-enriched product. Despite the challenges related to hemp milk stability and additives, SSF and germination proved to be viable alternatives. The G3 and FBL milk samples exhibited substantial increases in GABA levels, surpassing untreated seeds by 6.6 and 10.8 times, respectively, potentially offering health benefits. These improved samples also showed higher zeta potential and lower droplet sizes, indicating increased stability. Both bioprocesses contributed to higher phenolic, antioxidant, and peptide contents, enhancing the milk’s potential as a health-promoting beverage. Overall, the study demonstrated the potential of GABA-enriched hemp milk as a unique and healthful option.
Data Availability
No datasets were generated or analysed during the current study.
References
Vallath A, Shanmugam A, Rawson A (2022) Prospects of future pulse milk variants from other healthier pulses - as an alternative to soy milk. Trends Food Sci Technol 124:51–62. https://doi.org/10.1016/j.tifs.2022.03.028
Grandview Report (2023) https://www.grandviewresearch.com/industry-analysis/dairy-alternativesmarket
Sahab NRM, Subroto E, Balia RL, Utama GL (2020) γ-Aminobutyric acid found in fermented foods and beverages: current trends. Heliyon 6:e05526. https://doi.org/10.1016/j.heliyon.2020.e05526
Singh A, Bobade H, Sharma S, Singh B, Gupta A (2021) Enhancement of digestibility of nutrients (in vitro), antioxidant potential and functional attributes of wheat flour through grain germination. Plant Foods Hum Nutr 76. https://doi.org/10.1007/s11130-021-00881-z
Garzón AG, Van de Velde F, Drago SR (2020) Gastrointestinal and colonic in vitro bioaccessibility of γ-aminobutiric acid (GABA) and phenolic compounds from novel fermented sorghum food. Lwt 130:109664. https://doi.org/10.1016/j.lwt.2020.109664
Cui Y, Miao K, Niyaphorn S, Qu X (2020) Production of gamma-aminobutyric acid from lactic acid bacteria: a systematic review. Int J Mol Sci 21. https://doi.org/10.3390/ijms21030995
Pannerchelvan S, Rios-Solis L, Faizal Wong FW, Zaidan UH, Wasoh H, Mohamed MS, Tan JS, Mohamad R, Halim M (2023) Strategies for improvement of gamma-aminobutyric acid (GABA) biosynthesis via lactic acid bacteria (LAB) fermentation, Food Funct. 14. https://doi.org/10.1039/d2fo03936b
Frassinetti S, Moccia E, Caltavuturo L, Gabriele M, Longo V, Bellani L, Giorgi G, Giorgetti L (2018) Nutraceutical potential of hemp (Cannabis sativa L.) seeds and sprouts. Food Chem 262:56–66. https://doi.org/10.1016/j.foodchem.2018.04.078
Paul AA, Kumar S, Kumar V, Sharma R (2020) Milk Analog: Plant based alternatives to conventional milk, production, potential and health concerns. Crit Rev Food Sci Nutr 60:3005–3023. https://doi.org/10.1080/10408398.2019.1674243
Nigro E, Pecoraro MT, Formato M, Piccolella S, Ragucci S, Mallardo M, Russo R, Di Maro A, Daniele A, Pacifico S (2022) Cannabidiolic acid in hemp seed oil table spoon and beyond. Molecules 27. https://doi.org/10.3390/molecules27082566
Wang Q, Jiang J, Xiong YL (2018) High pressure homogenization combined with pH shift treatment: a process to produce physically and oxidatively stable hemp milk. Food Res Int 106:487–494. https://doi.org/10.1016/j.foodres.2018.01.021
Lu Y, Guo X (2020) The effect of light in vitamin c metabolism regulation and accumulation in mung bean (Vigna radiata) germination, Plant Foods Hum. Nutr 75. https://doi.org/10.1007/s11130-019-00787-x
Chavarín-Martínez CD, Gutiérrez-Dorado R, Perales-Sánchez JXK, Cuevas-Rodríguez EO, Milán-Carrillo J (2019) Reyes-Moreno, Germination in optimal conditions as effective strategy to improve nutritional and nutraceutical value of underutilized Mexican blue maize seeds. Plant Foods Hum Nutr 74. https://doi.org/10.1007/s11130-019-00717-x
Ding J, Hou GG, Nemzer BV, Xiong S, Dubat A, Feng H (2018) Effects of controlled germination on selected physicochemical and functional properties of whole-wheat flour and enhanced γ-aminobutyric acid accumulation by ultrasonication. Food Chem 243:214–221. https://doi.org/10.1016/j.foodchem.2017.09.128
Sahin S, Eulenburg V, Kreis W, Villmann C, Pischetsrieder M (2016) Three-step test system for the identification of novel GABAA receptor modulating food plants. Plant Foods Hum Nutr 71. https://doi.org/10.1007/s11130-016-0566-1
Sharma S, Saxena DC, Riar CS (2016) Isolation of functional components β-glucan and γ-amino butyric acid from raw and germinated barnyard millet (Echinochloa Frumentaceae) and their characterization. Plant Foods Hum Nutr 71. https://doi.org/10.1007/s11130-016-0545-6
Coda R, Rizzello CG, Gobbetti M (2010) Use of sourdough fermentation and pseudo-cereals and leguminous flours for the making of a functional bread enriched of γ-aminobutyric acid (GABA). Int J Food Microbiol 137. https://doi.org/10.1016/j.ijfoodmicro.2009.12.010
Suwanmanon K, Hsieh PC (2014) Isolating Bacillus subtilis and optimizing its fermentative medium for GABA and nattokinase production. CYTA - J Food 12. https://doi.org/10.1080/19476337.2013.848472
Zhang Q, Sun Q, Tan X, Zhang S, Zeng L, Tang J, Xiang W (2020) Characterization of γ-aminobutyric acid (GABA)-producing Saccharomyces cerevisiae and coculture with Lactobacillus plantarum for mulberry beverage brewing. J Biosci Bioeng 129:447–453. https://doi.org/10.1016/j.jbiosc.2019.10.001
Kwon SY, Garcia CV, Song YC, Lee SP (2016) GABA-enriched water dropwort produced by co-fermentation with Leuconostoc mesenteroides SM and Lactobacillus plantarum K154. LWT 73. https://doi.org/10.1016/j.lwt.2016.06.002
Zhang D, Wei X, Liu Z, Wu X, Bao C, Sun Y, Su N, Cui J (2021) Transcriptome analysis reveals the molecular mechanism of GABA Accumulation during Quinoa (Chenopodium quinoa Willd.) Germination. J Agric Food Chem 69:12171–12186. https://doi.org/10.1021/acs.jafc.1c02933
Lee MJ, Lee HJ, Lee Y, Yang JY, Song JS, Woo SY, Kim HY, Song SY, Seo WD, Son YJ (2023) Il Park, Cold plasma treatment increases bioactive metabolites in oat (Avena sativa L.) sprouts and enhances in vitro osteogenic activity of their extracts. Plant Foods Hum Nutr 78. https://doi.org/10.1007/s11130-022-01029-3
Salopek B, Krasic D, Filipovic S (1992) Measurement and application of zeta-potential. Rud Zb 4
Karabulut G, Feng H, Yemiş O (2022) Physicochemical and antioxidant properties of industrial hemp seed protein isolate treated by high-intensity ultrasound. Plant Foods Hum Nutr 77:577–583. https://doi.org/10.1007/s11130-022-01017-7
Merenkova S, Fatkullin R, Kalinina I (2022) Effect of fermentation on the biochemical parameters antioxidant capacity and dispersed composition of plant beverages based on barley and hemp seeds. Fermentation 8. https://doi.org/10.3390/fermentation8080384
Hu M, Du X, Liu G, Zhang S, Wu H, Li Y (2022) Germination improves the functional properties of soybean and enhances soymilk quality. Int J Food Sci Technol 57. https://doi.org/10.1111/ijfs.15461
Maleki S, Razavi SH (2020) Pulses’ germination and fermentation: two bioprocessing against hypertension by releasing ACE inhibitory peptides. Crit Rev Food Sci Nutr 1–18. https://doi.org/10.1080/10408398.2020.1789551
Ma Z, Boye JI, Hu X (2018) Nutritional quality and techno-functional changes in raw, germinated and fermented yellow field pea (Pisum sativum L.) upon pasteurization, LWT. 92. https://doi.org/10.1016/j.lwt.2018.02.018
López-Barrios L, Antunes-Ricardo M, Gutiérrez-Uribe JA (2016) Changes in antioxidant and antiinflammatory activity of black bean (Phaseolus vulgaris L.) protein isolates due to germination and enzymatic digestion. Food Chem 203:417–424. https://doi.org/10.1016/j.foodchem.2016.02.048
Dos V, Salgado SCN, Zago L, Antunes AEC, Miyahira RF (2022) Chia (Salvia hispanica L.) seed germination: a brief review. Plant Foods Hum Nutr 77. https://doi.org/10.1007/s11130-022-01011-z
Sandoval-Sicairos ES, Milán-Noris AK, Luna-Vital DA, Milán-Carrillo J, Montoya-Rodríguez A (2021) Anti-inflammatory and antioxidant effects of peptides released from germinated amaranth during in vitro simulated gastrointestinal digestion. Food Chem 343:128394. https://doi.org/10.1016/j.foodchem.2020.128394
Teh SS, Bekhit AEDA, Carne A, Birch J (2016) Antioxidant and ACE-inhibitory activities of hemp (Cannabis sativa L.) protein hydrolysates produced by the proteases AFP, HT, Pro-G, actinidin and zingibain. Food Chem 203:199–206. https://doi.org/10.1016/j.foodchem.2016.02.057
Liu F, Chen Z, Shao J, Wang C, Zhan C (2017) Effect of fermentation on the peptide content, phenolics and antioxidant activity of defatted wheat germ. Food Biosci 20:141–148. https://doi.org/10.1016/j.fbio.2017.10.002
Gómez-Favela MA, Gutiérrez-Dorado R, Cuevas-Rodríguez EO, Canizalez-Román VA, del Rosario León-Sicairos C, Milán-Carrillo J, Reyes-Moreno C (2017) Improvement of Chia seeds with antioxidant activity, GABA, essential amino acids, and dietary fiber by controlled germination bioprocess. Plant Foods Hum Nutr 72. https://doi.org/10.1007/s11130-017-0631-4
Miyahira RF, de Lopes J, Antunes AEC (2021) The use of sprouts to improve the nutritional value of food products: a brief review. Plant Foods Hum Nutr 76. https://doi.org/10.1007/s11130-021-00888-6
Ha KS, Jo SH, Mannam V, Kwon YI, Apostolidis E (2016) Stimulation of phenolics, antioxidant and α-glucosidase inhibitory activities during barley (Hordeum vulgare L.) seed germination. Plant Foods Hum Nutr 71. https://doi.org/10.1007/s11130-016-0549-2
Funding
Not Applicable.
Open access funding provided by the Carolinas Consortium.
Author information
Authors and Affiliations
Contributions
G. K. wrote the main manuscript text, methodology, and data analysis. B.V.N. methodology and data analysis. H. F. funding acquisition, supervision, and editing. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Ethics Approval
This article does not include any studies with human or animals’ subjects.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic Supplementary Material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Karabulut, G., Nemzer, B.V. & Feng, H. γ-Aminobutyric Acid (GABA)-enriched Hemp Milk by Solid-state Co-fermentation and Germination Bioprocesses. Plant Foods Hum Nutr 79, 322–329 (2024). https://doi.org/10.1007/s11130-024-01187-6
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11130-024-01187-6