Abstract
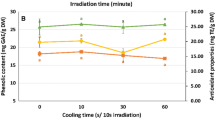
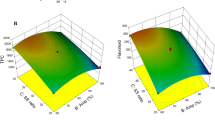
Two environmentally friendly innovative extraction techniques - subcritical water (SWE) and microwave-assisted extraction (MAE) were applied for the extraction of phenolics from pomegranate peel. The impact of process conditions (SWE: temperature 100–220 °C, extraction time 5–30 min; MAE: solvent water and 50% ethanol, irradiation power 470 and 800 W) on the quality of extracts in terms of the content of total phenolics, total flavonoids, major phenolic constituents (gallic acid, ellagic acid, punicalin, punicalagin), as well as 5-hydroxymethylfurfural(HMF) amount was investigated. For SWE, temperature of 130 °C and 20 min extraction time were found optimal for obtaining high content of bioactive compounds and minimizing the yield of HMF. During MAE, phenolic compounds were effectively extracted by using lower microwave power and 50% ethanol. Comparing two techniques, MAE is more efficient than SWE for the extraction of phenolics from pomegranate peel while obtaining a HMF-free extracts.


Similar content being viewed by others
References
Akhtar S, Ismail T, Fraternale D, Sestili P (2015) Pomegranate peel and peel extracts: chemistry and food features. Food Chem 174:417–425. https://doi.org/10.1016/j.foodchem.2014.11.035
Sood A, Gupta M (2015) Extraction process optimization for bioactive compounds in pomegranate peel. Food Biosci 12:100–106. https://doi.org/10.1016/j.fbio.2015.09.004
Fischer UA, Carle R, Kammerer DR (2011) Identification and quantification of phenolic compounds from pomegranate (Punica granatum L.) peel, mesocarp, aril and differently produced juices by HPLC-DAD–ESI/MSn. Food Chem 127:807–821. https://doi.org/10.1016/j.foodchem.2010.12.156
Li J, He X, Li M, Zhao W, Liu L, Kong X (2015) Chemical fingerprint and quantitative analysis for quality control of polyphenols extracted from pomegranate peel by HPLC. Food Chem 176:7–11. https://doi.org/10.1016/j.foodchem.2014.12.040
Salgado JM, Ferreira TRB, de Oliveira BF, dos Santos Dias CT (2012) Increased antioxidant content in juice enriched with dried extract of pomegranate (Punica granatum) peel. Plant Foods Hum Nutr 67(1):39–43. https://doi.org/10.1007/s11130-011-0264-y
Pocan P, Bahcegul E, Oztop MH, Hamamci H (2018) Enzymatic hydrolysis of fruit peels and other lignocellulosic biomass as a source of sugar. Waste Biomass Valori 9:929–937. https://doi.org/10.1007/s12649-017-9875-3
Hasnaoui N, Wathelet B, Jiménez-Araujo A (2014) Valorization of pomegranate peel from 12 cultivars: dietary fibre composition, antioxidant capacity and functional properties. Food Chem 160:196–203. https://doi.org/10.1016/j.foodchem.2014.03.089
Khoddami A, Wilkes MA, Roberts TH (2013) Techniques for analysis of plant phenolic compounds. Molecules 18:2328–2375. https://doi.org/10.3390/molecules18022328
Diamanti AC, Igoumenidis PE, Mourtzinos I, Yannakopoulou K, Karathanos VT (2017) Green extraction of polyphenols from whole pomegranate fruit using cyclodextrins. Food Chem 214:61–66. https://doi.org/10.1016/j.foodchem.2016.07.072
Herrero M, Castro-Puyana M, Mendiola JA, Ibañez E (2013) Compressed fluids for the extraction of bioactive compounds. TrAC Trend Anal Chem 43:67–83. https://doi.org/10.1016/j.trac.2012.12.008
Teo CC, Tan SN, Yong JWH, Hew CS, Ong ES (2010) Pressurized hot water extraction (PHWE). J Chromatogr A 1217:2484–2494. https://doi.org/10.1016/j.chroma.2009.12.050
Metaxas AA, Meredith RJ (1983) Industrial microwave heating (no. 4). IET
Ameer K, Shahbaz HM, Kwon JH (2017) Green extraction methods for polyphenols from plant matrices and their byproducts: a review. Compr Rev Food Sci Food Saf 16:295–315. https://doi.org/10.1111/1541-4337.12253
Delazar A, Nahar L, Hamedeyazdan S, Sarker SD (2012)Microwave-assisted extraction in natural products isolation. In: Natural products isolation, Humana Press, pp 89–115
Wang L, Weller CL (2006) Recent advances in extraction of nutraceuticals from plants. Trends Food Sci Technol 17(6):300–312. https://doi.org/10.1016/j.tifs.2005.12.004
Ameur LA, Trystram G, Birlouez-Aragon I (2006) Accumulation of 5-hydroxymethyl-2-furfural in cookies during the backing process: validation of an extraction method. Food Chem 98:790–796. https://doi.org/10.1016/j.foodchem.2005.07.038
Capuano E, Fogliano V (2011) Acrylamide and 5-hydroxymethylfurfural (HMF): a review on metabolism, toxicity, occurrence in food and mitigation strategies. LWT-Food Sci Technol 44:793–810. https://doi.org/10.1016/j.lwt.2010.11.002
Khajavi SH, Kimura Y, Oomori T, Matsuno R, Adachi S (2005) Degradation kinetics of monosaccharides in subcritical water. J Food Eng 68:309–313. https://doi.org/10.1016/j.jfoodeng.2004.06.004
Lee CH, Chen KT, Lin JA, Chen YT, Chen YA, Wu JT, Hsieh CW (2019) Recent advances in processing technology to reduce 5-hydroxymethylfurfural in foods. Trends Food Sci Technol 93:271–280. https://doi.org/10.1016/j.tifs.2019.09.021
Plaza M, Turner C (2015) Pressurized hot water extraction of bioactives. TrAC Trend Anal Chem 71:39–54. https://doi.org/10.1016/j.trac.2015.02.022
Yan L, Cao Y, Zheng G (2017) Optimization of subcritical water extraction of phenolic antioxidants from pomegranate (Punica granatum L.) peel by response surface methodology. Anal Methods 9:4647–4656. https://doi.org/10.1039/C7AY01475A
Çam M, Hışıl Y (2010) Pressurised water extraction of polyphenols from pomegranate peels. Food Chem 123:878–885. https://doi.org/10.1016/j.foodchem.2010.05.011
Kanmaz EÖ (2018)5-Hydroxymethylfurfural(HMF) formation during subcritical water extraction. Food Sci Biotechnol 27:981–986. https://doi.org/10.1007/s10068-018-0328-y
Narita Y, Inouye K (2012) High antioxidant activity of coffee silverskin extracts obtained by the treatment of coffee silverskin with subcritical water. Food Chem 135:943–949. https://doi.org/10.1016/j.foodchem.2012.05.078
Erşan S, Üstündağ ÖG, Carle R, Schweiggert RM (2018) Subcritical water extraction of phenolic and antioxidant constituents from pistachio (Pistacia vera L.) hulls. Food Chem 253:46–54. https://doi.org/10.1016/j.foodchem.2018.01.116
Abid M, Renard CM, Watrelot AA, Fendri I, Attia H, Ayadi MA (2016) Yield and composition of pectin extracted from Tunisian pomegranate peel. Int J Biol Macromol 93:186–194. https://doi.org/10.1016/j.ijbiomac.2016.08.033
Dafny-Yalin M, Glazer I, Bar-Ilan I, Kerem Z, Holland D, Amir R (2010) Color, sugars and organic acids composition in aril juices and peel homogenates prepared from different pomegranate accessions. J Agric Food Chem 58:4342–4352. https://doi.org/10.1021/jf904337t
Huang J, He W, Yan X, Sh X (2017) Microwave assisted extraction of flavonoids from pomegranate peel and its antioxidant activity. BIO Web of Conferences 8:03008. https://doi.org/10.1051/bioconf/20170803008
Kaderides K, Papaoikonomou L, Serafim M, Goula AM (2019)Microwave-assisted extraction of phenolics from pomegranate peels: optimization, kinetics, and comparison with ultrasounds extraction. Chem Eng Process 137:1–11. https://doi.org/10.1016/j.cep.2019.01.006
Galan AM, Calinescu I, Trifan A, Winkworth-Smith C, Calvo-Carrascal M, Dodds C, Binner E (2017) New insights into the role of selective and volumetric heating during microwave extraction: investigation of the extraction of polyphenolic compounds from Sea buckthorn leaves using microwave-assisted extraction and conventional solvent extraction. Chem Eng Process 116:29–39. https://doi.org/10.1016/j.cep.2017.03.006
Zheng X, Xu X, Liu C, Sun Y, Lin Z, Liu H (2013) Extraction characteristics and optimal parameters of anthocyanin from blueberry powder under microwave-assisted extraction conditions. Sep Purif Technol 104:17–25. https://doi.org/10.1016/j.seppur.2012.11.011
Simić VM, Rajković KM, Stojičević SS, Veličković DT, Nikolić NČ, Lazić ML, Karabegović IT (2016) Optimization of microwave-assisted extraction of total polyphenolic compounds from chokeberries by response surface methodology and artificial neural network. Sep Purif Technol 160:89–97. https://doi.org/10.1016/j.seppur.2016.01.019
Zheng X, Liu B, Li L, Zhu X (2011)Microwave-assisted extraction and antioxidant activity of total phenolic compounds from pomegranate peel. J Med Plant Res 5:1004–1011
Zou T, Wu H, Li H, Jia Q, Song G (2013) Comparison of microwave-assisted and conventional extraction of mangiferin from mango (Mangifera indica L.) leaves. J Sep Sci 36:3457–3462. https://doi.org/10.1002/jssc.201300518
Bartnik DD, Mohler CM, Houlihan M (2006) Methods for the production of food grade extracts. US Patent 20060088627, 27 April
Acknowledgements
This work was supported by the Ministry of Education, Science and Technological Development of the Republic of Serbia, contract number 451-03-68/2020-14/ 200003.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(DOCX 18 kb)
Rights and permissions
About this article
Cite this article
Vladić, J., Janković, T., Živković, J. et al. Comparative Study of Subcritical Water and Microwave-Assisted Extraction Techniques Impact on the Phenolic Compounds and 5-Hydroxymethylfurfural Content in Pomegranate Peel. Plant Foods Hum Nutr 75, 553–560 (2020). https://doi.org/10.1007/s11130-020-00848-6
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11130-020-00848-6