Abstract
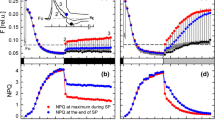
Nonphotochemical quenching acts as a frontline response to prevent excitation energy from reaching the photochemical reaction center of photosystem II before photodamage occurs. Strong fluorescence quenching after merely one multi-turnover saturating light pulse characterizes a unique feature of nonphotochemical quenching in red algae. Several mechanisms underlying red algal nonphotochemical quenching have been proposed, yet which process(es) dominantly account for the strong fluorescence quenching is still under discussion. Here we assessed multiple nonphotochemical quenching processes in the extremophilic red alga Cyanidioschyzon merolae under light pulse and continuous illumination conditions. To assess the nonphotochemical quenching processes that might display different kinetics, fluorescence emission spectra at 77 K were measured after different periods of light treatments, and external fluorophores were added for normalization of the fluorescence level. The phycobilisome- and photosystem II-related nonphotochemical quenching processes were distinguished by light preferentially absorbed by phycobilisomes and photosystems, respectively. Multiple nonphotochemical quenching processes, including the energetic decoupling of phycobilisomes from photosystem II, the energy spillover from phycobilisomes to photosystem I and from photosystem II to photosystem I, were identified along with the previously identified intrinsic quenching within photosystem II. The ability to use multiple nonphotochemical quenching processes appears to maximize the light harvesting efficiency for photochemistry and to provide the flexibility of the energy redistribution between photosystem II and photosystem I. The effect of the various ionophores on the nonphotochemical quenching level suggests that nonphotochemical quenching is modulated by transmembrane gradients of protons and other cations.








Similar content being viewed by others
References
Acuña AM, Snellenburg JJ, Gwizdala M, Kirilovsky D, van Grondelle R, van Stokkum IH (2016) Resolving the contribution of the uncoupled phycobilisomes to cyanobacterial pulse-amplitude modulated (PAM) fluorometry signals. Photosynth Res 127(1):91–102. https://doi.org/10.1007/s11120-015-0141-x
Avenson TJ, Kanazawa A, Cruz JA, Takizawa K, Ettinger WE, Kramer DM (2005) Integrating the proton circuit into photosynthesis: progress and challenges. Plant Cell Environ 28(1):97–109. https://doi.org/10.1111/j.1365-3040.2005.01294.x
Baker NR (2008) Chlorophyll fluorescence: a probe of photosynthesis in vivo. Annu Rev Plant Biol 59(1):89–113. https://doi.org/10.1146/annurev.arplant.59.032607.092759
Belgio E, Kapitonova E, Chmeliov J, Duffy CDP, Ungerer P, Valkunas L, Ruban AV (2014) Economic photoprotection in photosystem II that retains a complete light-harvesting system with slow energy traps. Nat Commun 5(1):4433. https://doi.org/10.1038/ncomms5433
Bhatti AF, Choubeh RR, Kirilovsky D, Wientjes E, van Amerongen H (2020) State transitions in cyanobacteria studied with picosecond fluorescence at room temperature. Biochim Biophys Acta Bioenerg 1861(10):148255. https://doi.org/10.1016/j.bbabio.2020.148255
Bhatti AF, Kirilovsky D, van Amerongen H, Wientjes E (2021) State transitions and photosystems spatially resolved in individual cells of the cyanobacterium Synechococcus elongatus. Plant Physiol 186(1):569–580. https://doi.org/10.1093/plphys/kiab063
Busch A, Nield J, Hippler M (2010) The composition and structure of photosystem I-associated antenna from Cyanidioschyzon merolae. Plant J 62(5):886–897. https://doi.org/10.1111/j.1365-313X.2010.04202.x
Calzadilla PI, Kirilovsky D (2020) Revisiting cyanobacterial state transitions. Photochem Photobiol Sci. https://doi.org/10.1039/C9PP00451C
Calzadilla PI, Zhan J, Sétif P, Lemaire C, Solymosi D, Battchikova N, Wang Q, Kirilovsky D (2019) The cytochrome b6f complex Is not involved in cyanobacterial state transitions. Plant Cell 31(4):911–931. https://doi.org/10.1105/tpc.18.00916
de Wijn R, van Gorkom HJ (2001) Kinetics of electron transfer from QA to QB in photosystem II. Biochemistry 40(39):11912–11922. https://doi.org/10.1021/bi010852r
Delphin E, Duval J-C, Etienne A-L, Kirilovsky D (1996) State transitions or ΔpH-dependent quenching of photosystem II fluorescence in red algae. Biochemistry 35(29):9435–9445. https://doi.org/10.1021/bi960528+
Delphin E, Duval J-C, Etienne A-L, Kirilovsky D (1998) ΔpH-dependent photosystem II fluorescence quenching induced by saturating, multiturnover pulses in red algae. Plant Physiol 118(1):103–113. https://doi.org/10.1104/pp.118.1.103
Derks A, Schaven K, Bruce D (2015) Diverse mechanisms for photoprotection in photosynthesis: dynamic regulation of photosystem II excitation in response to rapid environmental change. Biochim Biophys Acta Bioenerg 1847(4):468–485. https://doi.org/10.1016/j.bbabio.2015.02.008
Fang Y, Liu D, Jiang J, He A, Zhu R, Tian L (2022) Photoprotective energy quenching in the red alga Porphyridium purpureum occurs at the core antenna of the photosystem II but not at its reaction center. J Biol Chem 298(4):101783. https://doi.org/10.1016/j.jbc.2022.101783
Farooq S, Chmeliov J, Wientjes E, Koehorst R, Bader A, Valkunas L, Trinkunas G, van Amerongen H (2018) Dynamic feedback of the photosystem II reaction centre on photoprotection in plants. Nat Plants 4(4):225–231. https://doi.org/10.1038/s41477-018-0127-8
Gardian Z, Bumba L, Schrofel A, Herbstova M, Nebesarova J, Vacha F (2007) Organisation of photosystem I and photosystem II in red alga Cyanidium caldarium: encounter of cyanobacterial and higher plant concepts. Biochim Biophys Acta Bioenerg 1767(6):725–731. https://doi.org/10.1016/j.bbabio.2007.01.021
Kaňa R, Govindjee (2016) Role of ions in the regulation of light-harvesting. Front Plant Sci 7:1849–1849. https://doi.org/10.3389/fpls.2016.01849
Kaňa R, Kotabová E, Lukeš M, Papáček Š, Matonoha C, Liu L-N, Prášil O, Mullineaux CW (2014) Phycobilisome mobility and its role in the regulation of light harvesting in red algae. Plant Physiol 165(4):1618–1631. https://doi.org/10.1104/pp.114.236075
Klughammer C, Siebke K, Schreiber U (2013) Continuous ECS-indicated recording of the proton-motive charge flux in leaves. Photosynth Res 117(1):471–487. https://doi.org/10.1007/s11120-013-9884-4
Kowalczyk N, Rappaport F, Boyen C, Wollman FA, Collen J, Joliot P (2013) Photosynthesis in Chondrus crispus: the contribution of energy spill-over in the regulation of excitonic flux. Biochim Biophys Acta Bioenerg 1827(7):834–842. https://doi.org/10.1016/j.bbabio.2013.04.004
Krupnik T, Kotabova E, van Bezouwen LS, Mazur R, Garstka M, Nixon PJ, Barber J, Kana R, Boekema EJ, Kargul J (2013) A reaction center-dependent photoprotection mechanism in a highly robust photosystem II from an extremophilic red alga, Cyanidioschyzon Merolae. J Biol Chem 288(32):23529–23542. https://doi.org/10.1074/jbc.M113.484659
Ley AC, Butler WL (1977) Energy transfer from photosystem II to photosystem I in Porphyridium cruentum. Biochim Biophys Acta Bioenerg 462(2):290–294. https://doi.org/10.1016/0005-2728(77)90127-X
Liu L-N, Elmalk AT, Aartsma TJ, Thomas J-C, Lamers GEM, Zhou B-C, Zhang Y-Z (2008) Light-induced energetic decoupling as a mechanism for phycobilisome-related energy dissipation in red algae: a single molecule study. PLoS ONE 3(9):e3134. https://doi.org/10.1371/journal.pone.0003134
Liu L-N, Aartsma TJ, Thomas J-C, Zhou B-C, Zhang Y-Z (2009) FRAP analysis on red alga reveals the fluorescence recovery is ascribed to intrinsic photoprocesses of phycobilisomes than large-scale diffusion. PLoS ONE 4(4):e5295. https://doi.org/10.1371/journal.pone.0005295
Malnoë A (2018) Photoinhibition or photoprotection of photosynthesis? Update on the (newly termed) sustained quenching component qH. Environ Exp Bot. https://doi.org/10.1016/j.envexpbot.2018.05.005
McConnell MD, Koop R, Vasil’ev S, Bruce D (2002) Regulation of the distribution of chlorophyll and phycobilin-absorbed excitation energy in cyanobacteria: a structure-based model for the light state transition. Plant Physiol 130(3):1201–1212. https://doi.org/10.1104/pp.009845
Minoda A, Sakagami R, Yagisawa F, Kuroiwa T, Tanaka K (2004) Improvement of culture conditions and evidence for nuclear transformation by homologous recombination in a red alga, Cyanidioschyzon merolae 10D. Plant Cell Physiol 45(6):667–671. https://doi.org/10.1093/pcp/pch087
Murata N (1969) Control of excitation transfer in photosynthesis I: light-induced change of chlorophyll a fluoresence in Porphyridium cruentum. Biochim Biophys Acta Bioenerg 172(2):242–251. https://doi.org/10.1016/0005-2728(69)90067-X
Nicol L, Nawrocki WJ, Croce R (2019) Disentangling the sites of non-photochemical quenching in vascular plants. Nat Plants 5(11):1177–1183. https://doi.org/10.1038/s41477-019-0526-5
Ranjbar Choubeh R, Wientjes E, Struik PC, Kirilovsky D, van Amerongen H (2018) State transitions in the cyanobacterium Synechococcus elongatus 7942 involve reversible quenching of the photosystem II core. Biochim Biophys Acta Bioenerg 1859(10):1059–1066. https://doi.org/10.1016/j.bbabio.2018.06.008
Remelli W (1859) Santabarbara S (2018) Excitation and emission wavelength dependence of fluorescence spectra in whole cells of the cyanobacterium Synechocystis sp. PPC6803: Influence on the estimation of photosystem II maximal quantum efficiency. Biochim Biophys Acta Bioenerg 11:1207–1222. https://doi.org/10.1016/j.bbabio.2018.09.366
Ruban AV, Johnson MP (2009) Dynamics of higher plant photosystem cross-section associated with state transitions. Photosynth Res 99(3):173–183. https://doi.org/10.1007/s11120-008-9387-x
Santabarbara S, Villafiorita Monteleone F, Remelli W, Rizzo F, Menin B, Casazza AP (2019) Comparative excitation-emission dependence of the FV/FM ratio in model green algae and cyanobacterial strains. Physiol Plant 166(1):351–364. https://doi.org/10.1111/ppl.12931
Sonoike K (2011) Photoinhibition of photosystem I. Physiol Plant 142(1):56–64. https://doi.org/10.1111/j.1399-3054.2010.01437.x
Teplova VV, Belosludtsev KN, Kruglov AG (2017) Mechanism of triclosan toxicity: mitochondrial dysfunction including complex II inhibition, superoxide release and uncoupling of oxidative phosphorylation. Toxicol Lett 275:108–117. https://doi.org/10.1016/j.toxlet.2017.05.004
Tian L, Nawrocki WJ, Liu X, Polukhina I, van Stokkum IHM, Croce R (2019) pH dependence, kinetics and light-harvesting regulation of nonphotochemical quenching in Chlamydomonas. Proc Natl Acad Sci USA 116(17):8320–8325. https://doi.org/10.1073/pnas.1817796116
Ueno Y, Aikawa S, Kondo A, Akimoto S (2015) Light adaptation of the unicellular red alga, Cyanidioschyzon merolae, probed by time-resolved fluorescence spectroscopy. Photosynth Res 125(1):211–218. https://doi.org/10.1007/s11120-015-0078-0
Ueno Y, Aikawa S, Niwa K, Abe T, Murakami A, Kondo A, Akimoto S (2017) Variety in excitation energy transfer processes from phycobilisomes to photosystems I and II. Photosynth Res 133:1–9. https://doi.org/10.1007/s11120-017-0345-3
Van Etten J, Cho CH, Yoon HS, Bhattacharya D (2022) Extremophilic red algae as models for understanding adaptation to hostile environments and the evolution of eukaryotic life on the early earth. Semin Cell Dev Biol. https://doi.org/10.1016/j.semcdb.2022.03.007
Weatherly LM, Shim J, Hashmi HN, Kennedy RH, Hess ST, Gosse JA (2016) Antimicrobial agent triclosan is a proton ionophore uncoupler of mitochondria in living rat and human mast cells and in primary human keratinocytes. J Appl Toxicol 36(6):777–789. https://doi.org/10.1002/jat.3209
Wilson A, Ajlani G, Verbavatz J-M, Vass I, Kerfeld CA, Kirilovsky D (2006) A soluble carotenoid protein involved in phycobilisome-related energy dissipation in cyanobacteria. Plant Cell 18(4):992–1007. https://doi.org/10.1105/tpc.105.040121
Wollman F-A (2001) State transitions reveal the dynamics and flexibility of the photosynthetic apparatus. EMBO J 20(14):3623–3630. https://doi.org/10.1093/emboj/20.14.3623
Acknowledgements
We thank Shao-Lun Liu at Tunghai University, Taiwan, for his kind gift of the C. merolae 10D strain.
Funding
This work was supported by Ministry of Science and Technology, Taiwan (MOST 108-2311-B-110-001-, 109-2311-B-110-002-, 110-2311-B-110-002-MY3) and by the Higher Education Sprout Project and the Yushan Young Scholar Program, Ministry of Education, Taiwan.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Chiang, YH., Huang, YJ. & Fu, HY. Identification of multiple nonphotochemical quenching processes in the extremophilic red alga Cyanidioschyzon merolae. Photosynth Res 154, 125–141 (2022). https://doi.org/10.1007/s11120-022-00963-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11120-022-00963-2