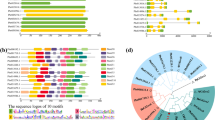
Abstract
Chloroplast functional genomics, in particular understanding the chloroplast transcriptional response is of immense importance mainly due to its role in oxygenic photosynthesis. As a photosynthetic unit, its efficiency and transcriptional activity is directly regulated by reactive oxygen species during abiotic and biotic stress and subsequently affects carbon assimilation, and plant biomass. In crops, understanding photosynthesis is crucial for crop domestication by identifying the traits that could be exploited for crop improvement. Transcriptionally and translationally active chloroplast plays a key role by regulating the PSI and PSII photo-reaction centres, which ubiquitously affects the light harvesting. Using a comparative transcriptomics mapping approach, we identified differential regulation of key chloroplast genes during salt stress across Triticeae members with potential genes involved in photosynthesis and electron transport system such as CytB6f. Apart from differentially regulated genes involved in PSI and PSII, we found widespread evidence of intron splicing events, specifically uniquely spliced petB and petD in Triticum aestivum and high proportion of RNA editing in ndh genes across the Triticeae members during salt stress. We also highlight the role and differential regulation of ATP synthase as member of CF0CF1 and also revealed the effect of salt stress on the water-splitting complex under salt stress. It is worthwhile to mention that the observed conserved down-regulation of psbJ across the Triticeae is limiting the assembly of water-splitting complexes and thus making the BEP clade Triticeae members more vulnerable to high light during the salt stress. Comparative understanding of the chloroplast transcriptional dynamics and photosynthetic regulation will improve the approaches for improved crop domestication.




Similar content being viewed by others
References
Allakhverdiev SI (2011) Recent progress in the studies of structure and function of photosystem II. Elsevier, Amsterdam
Allakhverdiev SI, Murata N (2008) Salt stress inhibits photosystems II and I in cyanobacteria. Photosynth Res 98:529–539
Allakhverdiev SI, Sakamoto A, Nishiyama Y, Inaba M, Murata N (2000) Ionic and osmotic effects of NaCl-induced inactivation of photosystems I and II in Synechococcus sp. Plant Physiol 123:1047–1056
Allakhverdiev SI, Nishiyama Y, Miyairi S, Yamamoto H, Inagaki N, Kanesaki Y, Murata N (2002) Salt stress inhibits the repair of photodamaged photosystem II by suppressing the transcription and translation of psbAGenes in Synechocystis. Plant Physiol 130:1443–1453
Araus J, Slafer G, Reynolds M, Royo C (2002) Plant breeding and drought in C3 cereals: what should we breed for? Ann Bot 89:925–940
Bennett J (1977) Phosphorylation of chloroplast membrane polypeptides. Nature 269:344–346
Bolger AM, Lohse M, Usadel B (2014) Trimmomatic: a flexible trimmer for Illumina sequence data. Bioinformatics 30:2114–2120
Börner GV, Mörl M, Wissinger B, Brennicke A, Schmelzer C (1995) RNA editing of a group II intron in Oenothera as a prerequisite for splicing. Mol Gen Genet 246:739–744
Brestic M, Zivcak M, Kalaji HM, Carpentier R, Allakhverdiev SI (2012) Photosystem II thermostability in situ: environmentally induced acclimation and genotype-specific reactions in Triticum aestivum L. Plant Physiol Biochem 57:93–105
Castandet B, Hotto AM, Strickler SR, Stern DB (2016) ChloroSeq, an optimized chloroplast RNA-Seq bioinformatic pipeline, reveals remodeling of the organellar transcriptome under heat stress. G3 Genes Genom Genet 6:2817–2827
Dinh SN, Sai TZT, Nawaz G, Lee K, Kang H (2016) Abiotic stresses affect differently the intron splicing and expression of chloroplast genes in coffee plants (Coffea arabica) and rice (Oryza sativa). J Plant Physiol 201:85–94
Guo R et al (2015) Comparative metabolic responses and adaptive strategies of wheat (Triticum aestivum) to salt and alkali stress. BMC Plant Biol 15:170
Hager M, Hermann M, Biehler K, Krieger-Liszkay A, Bock R (2002) Lack of the small plastid-encoded PsbJ polypeptide results in a defective water-splitting apparatus of photosystem II, reduced photosystem I levels, and hypersensitivity to light. J Biol Chem 277:14031–14039
He Y et al. (2015) Increasing cyclic electron flow is related to Na + sequestration into vacuoles for salt tolerance in soybean. J Exp Bot 66:6877–6889
Hein A, Polsakiewicz M, Knoop V (2016) Frequent chloroplast RNA editing in early-branching flowering plants: pilot studies on angiosperm-wide coexistence of editing sites and their nuclear specificity factors. BMC Evol Biol 16:23
Ichinose M, Sugita M (2016) RNA editing and its molecular mechanism in plant organelles. Genes 8:5
Kim D, Pertea G, Trapnell C, Pimentel H, Kelley R, Salzberg SL (2013) TopHat2: accurate alignment of transcriptomes in the presence of insertions, deletions and gene fusions. Genome Biol 14:R36
Kotera E, Tasaka M, Shikanai T (2005) A pentatricopeptide repeat protein is essential for RNA editing in chloroplasts. Nature 433:326
Kwon K-C, Verma D, Jin S, Singh ND, Daniell H (2013) Release of proteins from intact chloroplasts induced by reactive oxygen species during biotic and abiotic stress. PLoS ONE 8:e67106
Leister D, Wang L, Kleine T (2017) Organellar gene expression and acclimation of plants to environmental stress. Front Plant Sci 8:387
Lgloi G, Kössel H (1992) The transcriptional apparatus of chloroplasts. Crit Rev Plant Sci 10:525–558
Li J, Hu L, Zhang L, Pan X, Hu X (2015) Exogenous spermidine is enhancing tomato tolerance to salinity–alkalinity stress by regulating chloroplast antioxidant system and chlorophyll metabolism. BMC Plant Biol 15:303
Loll B, Kern J, Saenger W, Zouni A, Biesiadka J (2007) Lipids in photosystem II: interactions with protein and cofactors. Biochim Biophys Acta 1767:509–519
Lu K, Yang Y, He Y, Jiang D (2008) Induction of cyclic electron flow around photosystem 1 and state transition are correlated with salt tolerance in soybean. Photosynthetica 46:10–16
Meng F et al. (2016) Physiological and proteomic responses to salt stress in chloroplasts of diploid and tetraploid black locust (Robinia pseudoacacia L.). Sci Rep. https://doi.org/10.1038/srep23098
Mohanty P, Allakhverdiev SI, Murata N (2007) Application of low temperatures during photoinhibition allows characterization of individual steps in photodamage and the repair of photosystem II. Photosynth Res 94:217–224
Monde RA, Zito F, Olive J, Wollman FA, Stern DB (2000) Post-transcriptional defects in tobacco chloroplast mutants lacking the cytochrome b6/f complex. Plant J 21:61–72
Munekage Y, Hashimoto M, Miyake C, Tomizawa K-I, Endo T, Tasaka M, Shikanai T (2004) Cyclic electron flow around photosystem I is essential for photosynthesis. Nature 429:579–582
Munns R (2013) Hoagland’s nutrient solution. http://prometheuswiki.publish.csiro.au/tiki-index.php?page=Hoagland's+nutrient+solution
Munns R, James RA (2003) Screening methods for salinity tolerance: a case study with tetraploid wheat. Plant Soil 253:201–218
Najafpour MM, Allakhverdiev SI (2015) Recent progress in the studies of structure and function of photosystems I and II. J Photochem Photobiol B 152:173
Nouri M-Z, Moumeni A, Komatsu S (2015) Abiotic stresses: insight into gene regulation and protein expression in photosynthetic pathways of plants. Int J Mol Sci 16:20392–20416
Ohnishi N, Kashino Y, Satoh K, Ozawa S-i, Takahashi Y (2007) Chloroplast-encoded polypeptide PsbT is involved in the repair of primary electron acceptor QA of photosystem II during photoinhibition in Chlamydomonas reinhardtii. J Biol Chem 282:7107–7115
Ostersetzer O, Cooke AM, Watkins KP, Barkan A (2005) CRS1, a chloroplast group II intron splicing factor, promotes intron folding through specific interactions with two intron domains. Plant Cell 17:241–255
Parida A, Das AB, Das P (2002) NaCl stress causes changes in photosynthetic pigments, proteins, and other metabolic components in the leaves of a true mangrove, Bruguiera parviflora, in hydroponic cultures. J Plant Biol 45:28–36
Peng L, Yamamoto H, Shikanai T (2011) Structure and biogenesis of the chloroplast NAD (P) H dehydrogenase complex. Biochim Biophys Acta 1807:945–953
Peng L, Fukao Y, Fujiwara M, Shikanai T (2012) Multistep assembly of chloroplast NADH dehydrogenase-like subcomplex A requires several nucleus-encoded proteins, including CRR41 and CRR42, in Arabidopsis. Plant Cell 24:202–214
Pintó-Marijuan M, Munné-Bosch S (2014) Photo-oxidative stress markers as a measure of abiotic stress-induced leaf senescence: advantages and limitations. J Exp Bot 65:3845–3857
Ray DK, Mueller ND, West PC, Foley JA (2013) Yield trends are insufficient to double global crop production by 2050. PLoS ONE 8:e66428
Reddy AS, Marquez Y, Kalyna M, Barta A (2013) Complexity of the alternative splicing landscape in plants. Plant Cell 25:3657–3683
Rodrigues NF, Fonseca GC, Kulcheski FR, Margis R (2017) Salt stress affects mRNA editing in soybean chloroplasts. Genet Mol Biol 40:200–208
Sablok G, Mudunuri SB, Edwards D, Ralph PJ (2016) Chloroplast genomics: expanding resources for an evolutionary conserved miniature molecule with enigmatic applications. Curr Plant Biol 7:34–38
Sanchez DH et al (2008) Integrative functional genomics of salt acclimatization in the model legume Lotus japonicus. Plant J 53:973–987
Saradhi A, Pardha P, Mohanty P (1992) Enhancement of photosystem II photoreactions and high pH stability in thylakoids from cotyledonary leaves of Brassica juncea raised under sodium chloride stress. Physiol Plant 86:189–196
Schöttler MA, Flügel C, Thiele W, Bock R (2007) Knock-out of the plastid-encoded PetL subunit results in reduced stability and accelerated leaf age-dependent loss of the cytochrome b6f complex. J Biol Chem 282:976–985
Schwenkert S, Legen J, Takami T, Shikanai T, Herrmann RG, Meurer J (2007) Role of the low-molecular-weight subunits PetL, PetG, and PetN in assembly, stability, and dimerization of the cytochrome b6f complex in tobacco. Plant Physiol 144:1924–1935
Sickler CM, Edwards GE, Kiirats O, Gao Z, Loescher W (2007) Response of mannitol-producing Arabidopsis thaliana to abiotic stress. Funct Plant Biol 34:382–391
Singh RP, Runthala A, Khan S, Jha PN (2017) Quantitative proteomics analysis reveals the tolerance of wheat to salt stress in response to Enterobacter cloacae SBP-8. PLoS ONE 12:e0183513
Spetea C, Rintamäki E, Schoefs B (2014) Changing the light environment: chloroplast signalling and response mechanisms. Philos Trans R Soc Lond B 369:20130220
Stepien P, Johnson GN (2009) Contrasting responses of photosynthesis to salt stress in the glycophyte Arabidopsis and the halophyte Thellungiella: role of the plastid terminal oxidase as an alternative electron sink. Plant Physiol 149:1154–1165
Stoppel R, Meurer J (2013) Complex RNA metabolism in the chloroplast: an update on the psbB operon. Planta 237:441–449
Sun A-Z, Guo F-Q (2016) Chloroplast retrograde regulation of heat stress responses in plants. Front Plant Sci 7:398
Suo J, Zhao Q, David L, Chen S, Dai S (2017) Salinity response in chloroplasts: insights from gene characterization. Int J Mol Sci 18:1011
Swiatecka-Hagenbruch M, Liere K, Börner T (2007) High diversity of plastidial promoters in Arabidopsis thaliana. Mol Genet Genomics 277:725–734
Tamburino R et al (2017) Chloroplast proteome response to drought stress and recovery in tomato (Solanum lycopersicum L.). BMC Plant Biol 17:40
Tang X, Mu X, Shao H, Wang H, Brestic M (2015) Global plant-responding mechanisms to salt stress: physiological and molecular levels and implications in biotechnology. Crit Rev Biotechnol 35:425–437
Tseng C-C, Sung T-Y, Li Y-C, Hsu S-J, Lin C-L, Hsieh M-H (2010) Editing of accD and ndhF chloroplast transcripts is partially affected in the Arabidopsisvanilla cream1 mutant. Plant Mol Biol 73:309–323
Umate P et al (2007) Deletion of PsbM in tobacco alters the QB site properties and the electron flow within photosystem II. J Biol Chem 282:9758–9767
Van Den Bekerom RJ, Dix PJ, Diekmann K, Barth S (2013) Variations in efficiency of plastidial RNA editing within ndh transcripts of perennial ryegrass (Lolium perenne) are not linked to differences in drought tolerance. AoB Plants 5:plt035
Acknowledgements
We thank Institute of Science, High technology and Environmental Sciences, Graduate University of Advanced Technology for financial support (No. 94/5149). We also thank Iran National Foundation Science for financial support (No. 90003984).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Mirzaei, S., Mansouri, M., Mohammadi-Nejad, G. et al. Comparative assessment of chloroplast transcriptional responses highlights conserved and unique patterns across Triticeae members under salt stress. Photosynth Res 136, 357–369 (2018). https://doi.org/10.1007/s11120-017-0469-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11120-017-0469-5