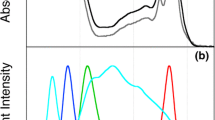
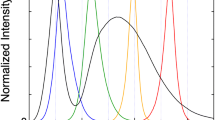
Abstract
Photosynthetic organisms change the quantity and/or quality of their pigment–protein complexes and the interactions among these complexes in response to light conditions. In the present study, we analyzed light adaptation of the unicellular red alga Cyanidioschyzon merolae, whose pigment composition is similar to that of cyanobacteria because its phycobilisomes (PBS) lack phycoerythrin. C. merolae were grown under different light qualities, and their responses were measured by steady-state absorption, steady-state fluorescence, and picosecond time-resolved fluorescence spectroscopies. Cells were cultivated under four monochromatic light-emitting diodes (blue, green, yellow, and red), and changes in pigment composition and energy transfer were observed. Cells grown under blue and green light increased their relative phycocyanin levels compared with cells cultured under white light. Energy-transfer processes to photosystem I (PSI) were sensitive to yellow and red light. The contribution of direct energy transfer from PBS to PSI increased only under yellow light, while red light induced a reduction in energy transfer from photosystem II to PSI and an increase in energy transfer from light-harvesting chlorophyll protein complex I to PSI. Differences in pigment composition, growth, and energy transfer under different light qualities are discussed.





Similar content being viewed by others

Abbreviations
- APC:
-
Allophycocyanin
- Car:
-
Carotenoid
- Chl:
-
Chlorophyll
- FDAS:
-
Fluorescence decay-associated spectrum (spectra)
- LED:
-
Light-emitting diodes
- LHC:
-
Light-harvesting chlorophyll protein complex
- PBS:
-
Phycobilisome
- PC:
-
Phycocyanin
- PE:
-
Phycoerythrin
- PS:
-
Photosystem
- TRFS:
-
Time-resolved fluorescence spectrum (spectra)
References
Abe S, Murakami A, Ohki K, Aruga Y, Fujita Y (1994) Changes in stoichiometry among PSI, PSII and Cyt b 6 -f complexes in response to chromatic light for cell growth observed with the red alga Porphyra yezoensis. Plant Cell Physiol 35:901–906
Akimoto S, Yokono M, Hamada F, Teshigahara A, Aikawa S, Kondo A (2012) Adaptation of light-harvesting systems of Arthrospira platensis to light conditions, probed by time-resolved fluorescence spectroscopy. Biochim Biophys Acta 1817:1483–1489
Akimoto S, Yokono M, Aikawa S, Kondo A (2013) Modification of energy-transfer processes in the cyanobacterium, Arthrospira platensis, to adapt to light conditions, probed by time-resolved fluorescence spectroscopy. Photosynth Res 117:235–243
Arteni AA, Liu LN, Aartsma TJ, Zhang YZ, Zhou BC, Boekema EJ (2008) Structure and organization of phycobilisomes on membranes of the red alga Porphyridium cruentum. Photosynth Res 95:169–174
Bruce D, Biggins J, Steiner T, Thewalt M (1985) Mechanism of the light state transition in photosynthesis. IV. Picosecond fluorescence spectroscopy of Anacystis nidulans and Porphyridium cruentum in state 1 and state 2 at 77 K. Biochim Biophys Acta 806:237–246
Butler WL, Kitajima M (1975) Energy transfer between photosystem II and photosystem I in chloroplasts. Biochim Biophys Acta 396:72–85
Ciniglia C, Yoon HS, Pollio A, Pinto G, Bhattacharya D (2004) Hidden biodiversity of the extremophilic cyanidiales red algae. Mol Ecol 13:1827–1838
Cunningham FX Jr, Dennenberg RJ, Jursinic PA, Gantt E (1990) Growth under red light enhances photosystem II relative to photosystem I and phycobilisomes in the red alga Porphyridium cruentum. Plant Physiol 93:888–895
Enami I, Fukuda I (1975) Mechanisms of the acido- and thermo-phily of Cyanidium caldarium Geitler I. Effects of temperature, pH and light intensity on the photosynthetic oxygen evolution of intact and treated cells. Plant Cell Physiol 16:211–220
Gantt E (1981) Phycobilisomes. Ann Rev Plant Physiol 32:327–347
Ghosh AK, Govindjee (1966) Transfer of the excitation energy in Anacystis nidulans grown to obtain different pigment ratios. Biophys J 6:611–619
Gutu A, Kehoe DM (2012) Emerging perspectives on the mechanisms, regulation, and distribution of light color acclimation in cyanobacteria. Mol Plant 5:1–13
Kaňa R, Kotabová E, Lukeš M, Papáček Š, Matonoha C, Liu L-N, Prášil O, Mullineaux CW (2014) Phycobilisome mobility and its role in the regulation of light harvesting in red algae. Plant Physiol 165:1618–1631
Kehoe DM, Grossman AR (1996) Similarity of a chromatic adaptation sensor to phytochrome and ethylene receptors. Science 273:1409–1412
Liu H, Zhang H, Niedzwiedzki DM, Prado M, He G, Gross ML, Blankenship RE (2013) Phycobilisomes supply excitations to both photosystems in a megacomplex in cyanobacteria. Science 342:1104–1107
Marquardt J, Rhiel E (1997) The membrane-intrinsic light-harvesting complex of the red alga Galdieria sulphuraria (formerly Cyanidium caldarium): biochemical and immunochemical characterization. Biochim Biophys Acta 1320:153–164
Mimuro M, Kikuchi H (2003) Antenna systems and energy transfer in Cyanophyta and Rhodophyta. In: Green BR, Parson WW (eds) Light-harvesting antennas in photosynthesis. Kluwer Academic Publishers, Dordrecht, pp 281–306
Mimuro M, Akimoto S, Tomo T, Yokono M, Miyashita H, Tsuchiya T (2007) Delayed fluorescence observed in the nanosecond time region at 77 K originates directly from the photosystem II reaction center. Biochim Biophys Acta 1767:327–334
Mimuro M, Yokono M, Akimoto S (2010) Variations in photosystem I properties in the primordial cyanobacterium Gloeobacter violaceus PCC 7421. Photochem Photobiol 86:62–69
Misumi O, Matsuzaki M, Nozaki H, Miyagishima S, Mori T, Nishida K, Yagisawa F, Yoshida Y, Kuroiwa H, Kuroiwa T (2005) Cyanidioschyzon merolae genome. A tool for facilitating comparable studies on organelle biogenesis in photosynthetic eukaryotes. Plant Physiol 137:567–585
Mullineaux CW (1992) Excitation energy transfer from phycobilisomes to photosystem I in a cyanobacterium. Biochim Biophys Acta 1100:285–292
Tsekos I, Niell FX, Aguilera J, Figueroa FL, Delivopoulos SG (2002) Ultrastructure of the vegetative gametophytic cells of Porphyra leucosticta (Rhodophyta) grown in red, blue and green light. Phycol Res 50:251–264
Wolfe GR, Cunningham FX, Durnford D, Green BR, Gantt E (1994) Evidence for a common origin of chloroplasts with light-harvesting complexes of different pigmentation. Nature 367:566–568
Yokono M, Akimoto S, Koyama K, Tsuchiya T, Mimuro M (2008) Energy transfer processes in Gloeobacter violaceus PCC 7421 that possesses phycobilisomes with a unique morphology. Biochim Biophys Acta 1777:55–65
Yokono M, Murakami A, Akimoto S (2011) Excitation energy transfer between photosystem II and photosystem I in red algae: larger amounts of phycobilisome enhance spillover. Biochim Biophys Acta 1807:847–853
Acknowledgments
The authors thank Prof. Y. Kashino and Dr. N. Kashino for providing technical information about the culturing procedure. This work was supported in part by a grant from the Kurita Water and Environment Foundation to S. Aikawa (No. 13A021).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Ueno, Y., Aikawa, S., Kondo, A. et al. Light adaptation of the unicellular red alga, Cyanidioschyzon merolae, probed by time-resolved fluorescence spectroscopy. Photosynth Res 125, 211–218 (2015). https://doi.org/10.1007/s11120-015-0078-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11120-015-0078-0