Abstract
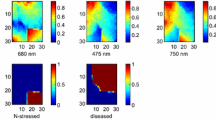
The objective of this research was to detect plant stress caused by disease infestation and to discriminate this type of stress from nutrient deficiency stress in field conditions using spectral reflectance information. Yellow Rust infected winter wheat plants were compared to nutrient stressed and healthy plants. In-field hyperspectral reflectance images were taken with an imaging spectrograph. A normalisation method based on reflectance and light intensity adjustments was applied. For achieving high performance stress identification, Self-Organising Maps (SOMs) and Quadratic Discriminant Analysis (QDA) were introduced. Winter wheat infected with Yellow Rust was successfully recognised from nutrient stressed and healthy plants. Overall performance using five wavebands was more than 99%.



Similar content being viewed by others
References
Blakeman, R.H., Bryson, R.J., & Dampney, P. (2000). Assessing crop condition in real time using high resolution satellite imagery. Aspects of Applied Biology, Remote Sensing in Agriculture, 60, 163–171.
Brown, J.K.M., & Hovmøller, M.S. (2002). Epidemiology—aerial dispersal of pathogens on the global and continental scales and its impact on plant disease. Science, 297, 537–541.
Carter, G.A., & Knapp, A.K. (2001). Leaf optical properties in higher plants: Linking spectral characteristics to stress and chlorophyll concentration. American Journal of Botany, 88(4), 677–684.
Chou, H.M., Bundock, N., Rolfe, S.A., & Scholes, J.D. (2000). Infection of Arabidopsis thaliana leaves with Albugo candida (white blister rust) causes a reprogramming of host metabolism. Molecular Plant Pathology, 1(2), 99–113.
Cibula, W.G., & Carter, G.A. (1992). Identification of a far-red reflectance response to ectomycorrhizae in slash pine. International Journal of Remote Sensing, 13, 925–932.
Dumont, K., & De Baerdemaeker, J. (2001). In field wheat nitrogen assessment using hyperspectral imaging techniques. In G. Grenier, & S. Blackmore (Eds.), Proceedings of the third European conference on precision agriculture (pp. 905–910). Montpellier, France: Agro Montpellier ENSAM.
EU project Framework Programme V, QLK5-1999-01280, acronym “OPTIDIS”, title “Development of an OPTIcal detection system for DISeases in field crops with a view to reduce pesticides by targeted application”.
Hansen, P.M., & Schjoerring, J.K. (2003). Reflectance measurement of canopy biomass and nitrogen status in wheat crops using normalized difference vegetation indices and partial least squares regression. Remote Sensing of Environment, 86(4), 542–553.
Klecka, W.R. (1980). Discriminant analysis. Newbury Park, CA, USA: Sage Publications.
Kohonen, T. (2001). Self-organizing maps. Berlin, Germany: Springer-Verlag, p. 501.
Lorenzen, B., & Jensen, A. (1989). Changes in spectral properties induced in Barley by cereal Powdery Mildew. Remote Sensing Environment, 27, 201–209.
Masoni, A., Laura, E., & Mariotti, M. (1996). Spectral properties of leaves deficient in iron, sulphur, magnesium and manganese. Agronomy Journal, 88(6), 937–943.
McCartney, H.A., & Fitt, B.D.L. (1998). Dispersal of foliar fungal plant pathogens: Mechanisms, gradients and spatial patterns. In D.G. Jones (Ed.), The epidemiology of plant pathogens (pp. 138–160). The Netherlands Dordrecht: Kluwer.
Polischuk, V.P., Shadchina, T.M., Kompanetz, T.I., Budzanivskaya, I.G., Sozinov, A.A. (1997). Changes in reflectance spectrum characteristic of Nicotiana debneyi plant under the influence of viral infection. Archives of Phytopathology and Plant Protection, 31(1), 115–119.
Rapilly, F., (1979). Yellow rust epidemiology. Annual Review of Phytopathology, 17, 59–73.
Rouse, J.W. Jr., Haas, R.H., Deering, D.W., Schell, J.A., & Harlan, J.C. (1974). Monitoring the vernal advancement and retrogradation (green wave effect) of natural vegetation. Greenbelt, MD, USA: NASA/GSFC Type III Final Report, p. 371.
Sasaki, Y., Okamoto, T., Imou, K., & Torii, T. (1998). Automatic diagnosis of plant disease-spectral reflectance of healthy and diseased leaves. In T. Kozai, H. Murase, & T. Hoshi (Eds.), Proceedings of 3rd IFAC/CIGR workshop on artificial intelligence in agriculture, Makuhari, Chiba, Japan (pp. 158–163). New York, USA: Pergammon Press.
Waggoner, P.E., & Aylor, D.E. (2000). Epidemiology: A science of patterns. Annual Review of Phytopathology, 38, 71–94.
West, J.S., Bravo, C., Oberti, R., Lemaire, D., Moshou, D., & McCartney, A. (2003). The potential of optical canopy measurement for targeted control of field crop diseases. Annual Review of Phytopathology, 41, 593–614.
Wright, D.L., Rasmussen, V.P., Neale, C.M.U., Harman, K., Searle, G., Grant, D., & Holle, C. (2001). A comparison of nitrogen stress detection methods in spring wheat. In Proceedings of ERIM third international conference on geospatial information in agriculture and forestry. Denver, Colorado, USA.
Zadoks, J.C., & Vandenbosch, F. (1994). On the spread of plant-disease—a theory on foci. Annual Review of Phytopathology, 32, 503–521.
Acknowledgements
This work was performed as part of the OPTIDIS project (QLK5-CT1999-01280), which was funded by the EU under the Quality of Life Programme—Framework V.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Moshou, D., Bravo, C., Wahlen, S. et al. Simultaneous identification of plant stresses and diseases in arable crops using proximal optical sensing and self-organising maps. Precision Agric 7, 149–164 (2006). https://doi.org/10.1007/s11119-006-9002-0
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11119-006-9002-0