Abstract
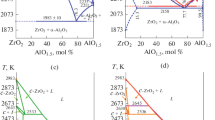
Isothermal sections at 1250 and 1650 °C have been constructed for the Al2O3-ZrO2-Er2O3 phase diagram. Phase equilibria at these temperatures have been identified. It is not found that there are any ternary compounds or noticeable regions of solid solutions based on the components or binary compounds. Conditionally quasibinary sections have been identified: Er3A5-F, ErA-F, and Er2A-F, which triangulate the Al2O3-ZrO2-Er2O3 ternary system.
Similar content being viewed by others
References
S. N. Lakiza, L. M. Lopato, and A. V. Shevchenko, “Interactions in the Al2O3-ZrO2-Y2O3 system,” Poroshk. Metall., Nos. 9–10, 46–51 (1994).
F. Bertaut and F. Forrat, “Etude des combinaisons des oxydes des terres rares avec l’alumine et la galline,” C. R. Acad. Sci., 243, No. 17, 1219–1222 (1956).
S. J. Schneider, R. S. Roth, and J. L. Waring, “Solid state reactions involving oxides of trivalent cations,” J. Res. Nat. Bur. Stand., 65A, No. 4, 345–374 (1961).
M. Mizuno, T. Yamada, and T. Nogushi, “Phase diagrams of the systems Al2O3-Ho2O3 and Al2O3-Er2O3 at high temperatures,” J. Ceram. Soc. Jap., 87, No. 8, 404–412 (1979).
P. Wu and A. D. Pelton, “Coupled thermodynamic phase diagram assessment of the rare earth oxide-aluminum oxide binary systems,” J. Alloys Comp., No. 179, 259–287 (1992).
H. Yamane, K. Ogawara, and T. Hirai, “Phase transition of rare-earth aluminates (RE4Al2O9) and rare earth gallates,” J. Am. Ceram. Soc., 78, No. 9, 2385–2390 (1995).
L. M. Lopato, A. V. Shevchenko, A. E. Kushchevskii, and S. G. Tresvyatskii, “Polymorphic transitions in rare-earth oxides at high temperatures,” Izv. AN SSSR, Neorgan. Materialy, 10, No. 8, 1481–1487 (1974).
M. R. Thornber, D. J. M. Bevan, and E. Summerville, “Mixed oxide of the type MO2 (fluorite)-M2O3. V. Phase studies in the system ZrO2-M2O3 (M = Sc, Yb, Er, Dy),” J. Solid State Chem., 1, No. 3–4, 545–553 (1970).
A. Rouanet, “Contribution a l’etude des systems zircone-oxydes des lanthanides au voisinage de la fusion,” Rev. Int. Hautes Temp. Refract., 8, No. 2, 161–180 (1977).
P. Duran, “The system erbia-zirconia,” J. Am. Ceram. Soc., 60, No. 11–12, 510–513 (1977).
C. Pascual and P. Duran, “Phase equilibria and ordering in the erbia-zirconia system,” J. Mater. Sci., 16, No. 11, 3067–3076 (1981).
L. M. Lopato, V. P. Red’ko, G. I. Gerasimyuk, and A. V. Shevchenko, “Synthesis of some REE zirconates (hafnates),” Poroshk. Metall., No. 4, 73–75 (1990).
M. Yoshima, T. Noma, N. Ishizava, and M. Yoshimura, “Stable and metastable phase relationships in the system ZrO2-ErO1.5,” J. Am. Ceram. Soc., 74, No. 3, 510–513 (1991).
E. R. Andrievskaya, L. M. Lopato, and A. V. Shevchenko, “The liquidus surface in the ZrO2-Y2O3-Er2O3 system,” Neorgan. Materialy, 32, No. 6, 721–726 (1996).
S. M. Lakiza, V. P. Red’ko, and L. M. Lopato, “Phase diagram for the Al2O3-ZrO2-Nd2O3 system. I. Isothermal sections of the phase diagram at 1250 and 1650°C,” Poroshk.Metall., Nos. 7–8, 77–86 (2003).
S. M. Lakiza and L. M. Lopato, “Stable and metastable phase relations in the system Alumina-Zirconia-Yttria,” J. Am. Ceram. Soc., 80, No. 4, 893–902 (1997).
R. D. Shannon and C. T. Prewitt, “Effective ionic radii in oxides and fluorides,” Acta Cryst., B25, No. 5, 924–946 (1969).
L. M. Lopato, A. V. Shevchenko, and A. E. Kushchevskii, “A study of highly refractory oxide systems,” Poroshk. Metall., No. 1, 88–92 (1972).
Author information
Authors and Affiliations
Additional information
__________
Translated from Poroshkovaya Metallurgiya, Nos. 11–12(452), pp. 55–64, November–December, 2006.
Rights and permissions
About this article
Cite this article
Lakiza, S.M., Red’ko, V.P. & Lopato, L.M. Phase diagram of the Al2O3-ZrO2-Er2O3 system. I. Isothermal sections at 1250 and 1650°C. Powder Metall Met Ceram 45, 553–562 (2006). https://doi.org/10.1007/s11106-006-0119-5
Received:
Issue Date:
DOI: https://doi.org/10.1007/s11106-006-0119-5