Abstract
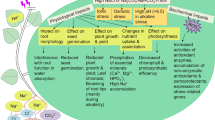
Chromium (Cr) can be easily taken up by plants and can pose significant threats to other organisms. Although it has not yet been determined that selenium (Se) is an essential element for plants, adding Se to the nutrient media reduces the toxicity of Cr in plants. However, to date, our knowledge of Se-mediated resistance to Cr stress is very limited. To better understand the molecular mechanisms of Se-mediated Cr tolerance, the gel-based proteomic approach to profile the proteins with abundance in the rapeseed (Brassica napus L.) seedlings was used. Chromium (10 μM)–treated seedlings exhibited a remarkable decrease in seedling growth and visual symptoms of toxicity characterized as leaf chlorosis. Exogenous Se (5 μM) reduced the toxicity of Cr by increasing the growth characteristics and decreasing Cr accumulation and lipid peroxidation in the leaves. The oxidative damage due to Cr was reduced with Se which could be related to elevated levels of antioxidant enzymes including superoxide dismutase (SOD), catalase (CAT), ascorbate peroxidase (APX), and guaiacol peroxidase (POD). In proteomic analysis, a total of 60 proteins were significantly influenced by Se in leaves under Cr stress, and 54 proteins were identified by mass spectrometry. Functional classification revealed that several pathways were regulated by Se, including photosynthesis and carbohydrate metabolism, stress defense, protein metabolism, and energy production. Selenium could increase the Cr tolerance by promoting photosynthesis efficiency, ROS scavenging ability, protein biosynthesis and processing, and other adaptive responses.



Similar content being viewed by others
Availability of Data and Materials
Not applicable.
References
Aebi H (1984) Catalase in vitro. Methods Enzymol 105:121–126. https://doi.org/10.1016/s0076-6879(84)05016-3
Agnihotri A, Seth CS (2016) Exogenously applied nitrate improves the photosynthetic performance and nitrogen metabolism in tomato (Solanum lycopersicum L. cv Pusa Rohini) under arsenic (V) toxicity. Physiol Mol Biol Plants 22(3):341–349. https://doi.org/10.1007/s12298-016-0370-2
Ali S, Chaudhary A, Rizwan M, Anwar HT, Adrees M, Farid M, Irshad MK (2015) Alleviation of chromium toxicity by glycinebetaine is related to elevated antioxidant enzymes and suppressed chromium uptake and oxidative stress in wheat (Triticum aestivum L.). Environ Sci Pollut R 22:10669–10678. https://doi.org/10.1007/s11356-015-4193-4
Badger MR, Price GD (1994) The role of carbonic anhydrase in photosynthesis. Annu Rev Plant Physiol Plant Mol Biol 45:369–392. https://doi.org/10.1146/annurev.pp.45.060194.002101
Baier M, Dietz KJ (1997) The plant 2-Cys peroxiredoxin BAS1 is a nuclear-encoded chloroplast protein: its expressional regulation, phylogenetic origin, and implications for its specific physiological function in plants. Plant J 12(1):179–190. https://doi.org/10.1046/j.1365-313x.1997.12010179.x
Barranco-Medina S, Lazaro JJ, Dietz KJ (2009) The oligomeric conformation of peroxiredoxins links redox state to function. FEBS Lett 583:1809–1816. https://doi.org/10.1016/j.febslet.2009.05.029
Beauchamp C, Fridovich I (1971) Superoxide dismutase: improved assays and an assay applicable to acrylamide gels. Anal Biochem 44:276–287. https://doi.org/10.1016/0003-2697(71)90370-8
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254. https://doi.org/10.1016/0003-2697(76)90527-3
Chance B, Maehly AC (1955) Assay of catalase and peroxidase. Methods Enzymol 2:746–778
Chauhan R, Awasthi S, Indoliya Y, Chauhan AS, Mishra S, Agrawal L, Srivastava S, Dwivedi S, Singh PC, Mallick S, Chauhan PS, Pande V, Chakrabarty D, Tripathi RD (2020) Transcriptome and proteome analyses reveal selenium mediated amelioration of arsenic toxicity in rice (Oryza sativa L.). J Hazard Mater 390:122122. https://doi.org/10.1016/j.jhazmat.2020.122122
Davidson RM, Reeves PA, Manosalva PM, Leach JE (2009) Germins: a diverse protein family important for crop improvement. Plant Sci 177:499–510. https://doi.org/10.1016/j.plantsci.2009.08.012
de Souza Cardoso AA, de Lima Gomes TF, Rodrigues Antonio JR, Guimaraes Guilherme LR, Liu J, Li L, de Souza Silva ML (2022) Sulfate availability and soil selenate adsorption alleviate selenium toxicity in rice plants. Environ Exp Bot 201:104971. https://doi.org/10.1016/j.envexpbot.2022.104971
del Rio LA, Pastori GM, Palma JM, Sandalio LM, Sevilla F, Corpas FJ, Jimenez A, Lopez-Huertas E, Hemandez JA (1998) The activated oxygen role of peroxisomes in senescence. Plant Physiol 116:1195–1200. https://doi.org/10.1104/pp.116.4.1195
Dixit V, Pandey V, Shyam R (2001) Differential antioxidative responses to cadmium in roots and leaves of pea (Pisum sativum L. cv. Azad). J Exp Bot 52:1101–1109. https://doi.org/10.1093/jexbot/52.358.1101
Feng RW, Wang LZ, Yang JG, Zhao PP, Zhu YM, Li YP, Yu Y, Liu H, Rensing C, Wu Z, Ni R, Zheng SA (2021) Underlying mechanisms responsible for restriction of uptake and translocation of heavy metals (metalloids) by selenium via root application in plants. J Hazard Mater 402:123570. https://doi.org/10.1016/j.jhazmat.2020.123570
Feng X, Ma Q (2021) Transcriptome and proteome profiling revealed molecular mechanism of selenium responses in bread wheat (Triticum aestivum L.). BMC Plant Biol 21:584. https://doi.org/10.1186/s12870-021-03368-w
Finka A, Mattoo RU, Goloubinoff P (2016) Experimental milestones in the discovery of molecular chaperones as polypeptide unfolding enzymes. Annu Rev Biochem 85:715–742. https://doi.org/10.1146/annurev-biochem-060815-014124
Gupta S, Seth CS (2021) Salicylic acid alleviates chromium (VI) toxicity by restricting its uptake, improving photosynthesis and augmenting antioxidant defense in Solanum lycopersicum L. Physiol Mol Biol Plants 27(11):2651–2664. https://doi.org/10.1007/s12298-021-01088-x
Handa N, Kohli SK, Sharma A, Thukral AK, Bhardwaj R, Alyemeni MN, Wijaya L, Ahmad P (2018) Selenium ameliorates chromium toxicity through modifications in pigment system, antioxidative capacity, osmotic system, and metal chelators in Brassica juncea seedlings. S Afr J Bot 119:1–10. https://doi.org/10.1016/j.sajb.2018.08.003
Handa N, Kohli SK, Sharma A, Thukral AK, Bhardwaj R, Abd_Allah EF, Alqarawi AA, Ahmad P (2019) Selenium modulates dynamics of antioxidative defence expression, photosynthetic attributes and secondary metabolites to mitigate chromium toxicity in Brassica juncea L. plants. Environ Exp Bot 161:180–192 https://doi.org/10.1016/j.envexpbot.2018.11.009
Heath RL, Packer L (1968) Photoperoxidation in isolated chloroplasts: I. Kinetics and stoichiometry of fatty acid peroxidation. Arch Biochem Biophys 125:189–198. https://doi.org/10.1016/0003-9861(68)90654-1
Hurkman WJ, Tanaka CK (1986) Solubilization of plant membrane proteins for analysis by two-dimensional gel electrophoresis. Plant Physiol 81:802–806. https://doi.org/10.1104/pp.81.3.802
James D, Borphukan B, Fartyal D, Ram B, Yadav R, Singh J, Manna M, Panditi V, Sheri V, Achary M, Reddy MK (2018) Concurrent overexpression of OsGS1;1 and OsGS2 genes in transgenic rice (Oryza sativa L.): impact on tolerance to abiotic stresses. Front Plant Sci 9:786. https://doi.org/10.3389/fpls.2018.00786
Jiang B, Gong Y, Gao J, Sun T, Liu Y, Oturan N, Oturan MA (2019) The reduction of Cr(VI) to Cr(III) mediated by environmentally relevant carboxylic acids: State-of-the-art and perspectives. J Hazard Mater 365:205–226. https://doi.org/10.1016/j.jhazmat.2018.10.070
Jiang H, Lin W, Jiao H, Liu J, Chan L, Liu X, Wang R, Chen T (2021) Uptake, transport, and metabolism of selenium and its protective effects against toxic metals in plants: a review. Metallomics 13(7):mfab040. https://doi.org/10.1093/mtomcs/mfab040
Kapoor RR, Mfarrej MFB, Alam P, Rinklebe J, Ahmad P (2022) Accumulation of chromium in plants and its repercussion in animals and humans. Environ Pollut 301:119044. https://doi.org/10.1016/j.envpol.2022.119044
Kaur C, Ghosh A, Pareek A, Sopory SK, Singla-Pareek SL (2014) Glyoxalases and stress tolerance in plants. Biochem Soc Trans 42(2):485–490. https://doi.org/10.1042/BST20130242
Kim SY, Jang HH, Lee JR, Sung NR, Lee HB, Lee DH, Park DJ, Kang CH, Chung WS, Lim CO, Yun DJ, Kim WY, Lee KO, Lee SY (2009) Oligomerization and chaperone activity of a plant 2-Cys peroxiredoxin in response to oxidative stress. Plant Sci 177(3):227–232. https://doi.org/10.1016/j.plantsci.2009.05.010
Kozuleva M, Goss T, Twachtmann M, Rudi K, Trapka J, Selinski J, Ivanov B, Garapati P, Steinhoff HJ, Hase T, Scheibe R, Klare JP, Hanke GT (2016) Ferredoxin:NADP(H) oxidoreductase abundance and location influences redox poise and stress tolerance. Plant Physiol 172:1480–1493. https://doi.org/10.1104/pp.16.01084
Kumar D, Seth CS (2022) Photosynthesis, lipid peroxidation, and antioxidative responses of Helianthus annuus L. against chromium (VI) accumulation. Int J Phytoremediat 24(6):590–599. https://doi.org/10.1080/15226514.2021.1958747
Marieschi M, Gorbi G, Zanni C, Sardella A, Torelli A (2015) Increase of chromium tolerance in Scenedesmus acutus after sulfur starvation: chromium uptake and compartmentalization in two strains with different sensitivities to Cr(VI). Aquat Toxicol 167:124–133. https://doi.org/10.1016/j.aquatox.2015.08.001
Membre N, Bernier F, Staiger D, Berna A (2000) Arabidopsis thaliana germin-like proteins: common and specific features point to a variety of functions. Planta 211:345–354. https://doi.org/10.1007/s004250000277
Meng HB, Hua SJ, Shamsi IH, Jilani G, Li YL, Jiang LX (2009) Cadmium induced stress on the seed germination and seedling growth of Brassica napus L., and its alleviation through exogenous plant growth regulators. Plant Growth Regul 58:47–59. https://doi.org/10.1007/s10725-008-9351-y
Nakano Y, Asada K (1981) Hydrogen peroxide is scavenged by ascorbate specific peroxidase in spinach chloroplasts. Plant Cell Physiol 22:867–880. https://doi.org/10.1093/oxfordjournals.pcp.a076232
Pilon-Smits EA (2019) On the ecology of selenium accumulation in plants. Plants 8(7):197. https://doi.org/10.3390/plants8070197
Pilon-Smits EA, Quinn CF (2010) Selenium metabolism in plants. In: Cell biology of metals and nutrients, Heidelberg: Springer, Berlin, pp 225–241. https://doi.org/10.1007/978-3-642-10613-2_10
Prinsi B, Espen L (2015) Mineral nitrogen sources differently affect root glutamine synthetase isoforms and amino acid balance among organs in maize. BMC Plant Biol 15:96. https://doi.org/10.1186/s12870-015-0482-9
Qing X, Zhao X, Hu C, Wang P, Zhang Y (2015) Selenium alleviates chromium toxicity by preventing oxidative stress in cabbage (Brassica campestris L. ssp. Pekinensis) leaves. Ecotox Environ Safe 114:179–189. https://doi.org/10.1016/j.ecoenv.2015.01.026
Seth CS, Kumar Chaturvedi P, Misra V (2008) The role of phytochelatins and antioxidants in tolerance to Cd accumulation in Brassica juncea L. Ecotoxicol Environ Saf 71(1):76–85. https://doi.org/10.1016/j.ecoenv.2007.10.030
Seth CS, Remans T, Keunen E, Jozefczak M, Gielen H, Opdenakker K, Weyens N, Vangronsveld J, Cuypers A (2012) Phytoextraction of toxic metals: a central role for glutathione. Plant Cell and Environ 35:334–346. https://doi.org/10.1111/j.1365-3040.2011.02338.x
Shahid M, Shamshad S, Rafiq M, Khalid S, Bibi I, Niazi NK, Dumat C, Rashid MI (2017) Chromium speciation, bioavailability, uptake, toxicity and detoxification in soil-plant system: a review. Chemosphere 178:513–533. https://doi.org/10.1016/j.chemosphere.2017.03.074
Singh D, Agnihotri A, Seth CS (2017) Interactive effects of EDTA and oxalic acid on chromium uptake, translocation and photosynthetic attributes in Indian mustard (Brassica juncea L. var. Varuna). Curr Sci 112(10):2034–2042. https://doi.org/10.18520/CS/V112/I10/2034-2042
Sinha P, Poland J, Schnölzer M, Rabilloud T (2001) A new silver staining apparatus and procedure for matrix-assisted laser desorption/ionization-time of flight analysis of proteins after two-dimensional electrophoresis. Proteomics 1:835–840. https://doi.org/10.1002/1615-9861(200107)1:7%3c835::AID-PROT835%3e3.0.CO;2-2
Srivastava D, Tiwari M, Dutta P, Singh P, Chawda K, Kumari M, Chakrabarty D (2021) Chromium stress in plants: toxicity, tolerance and phytoremediation. Sustainability 13(9):4629. https://doi.org/10.3390/su13094629
Sun H, Dai H, Wang X, Wang G (2016) Physiological and proteomic analysis of selenium-mediated tolerance to Cd stress in cucumber (Cucumis sativus L.). Ecotox Environ Safe 133:114–126. https://doi.org/10.1016/j.ecoenv.2016.07.003
Szklarczyk D, Franceschini A, Kuhn M, Simonovic M, Roth A, Minguez P, Doerks T, Stark M, Muller J, Bork P, Jensen LJ, von Mering C (2011) The STRING database in 2011: functional interaction networks of proteins, globally integrated and scored. Nucleic Acids Res 39:D561–D568. https://doi.org/10.1093/nar/gkq973
Vecchia FD, Nardi S, Santoro V, Pilon-Smits E, Schiavon M (2023) Brassica juncea and the Se-hyperaccumulator Stanleya pinnata exhibit a different pattern of chromium and selenium accumulation and distribution while activating distinct oxidative stress-response signatures. Environ Pollut 320:121048. https://doi.org/10.1016/j.envpol.2023.121048
von Ballmoos C, Dimroth P (2007) Two distinct proton binding sites in the ATP synthase family. Biochemistry 46:11800–11809. https://doi.org/10.1021/bi701083v
Wakeel A, Xu M (2020) Chromium Morpho-Phytotoxicity Plants 9(5):564. https://doi.org/10.3390/plants9050564
Wan Y, Wang K, Liu Z, Yu Y, Wang Q, Li H (2019) Effect of selenium on the subcellular distribution of cadmium and oxidative stress induced by cadmium in rice (Oryza sativa L.). Environ Sci Pollut R 26(16):16220–16228. https://doi.org/10.1007/s11356-019-04975-9
Wolucka BA, Montagu MV (2003) GDP-mannose 3′,5′-epimerase forms GDP-L-gulose, a putative intermediate for the de Novo biosynthesis of vitamin C in plants. J Biol Chem 278:47483–47490. https://doi.org/10.1074/jbc.M309135200
Yadav M, Praveen Gupta P, Seth CS (2022) Foliar application of α-lipoic acid attenuates cadmium toxicity on photosynthetic pigments and nitrogen metabolism in Solanum lycopersicum L. Acta Physiol Plant 44:112. https://doi.org/10.1007/s11738-022-03445-z
Yildiz M, Terzi H (2021) Exogenous cysteine alleviates chromium stress via reducing its uptake and regulating proteome in roots of Brassica napus L. seedlings. S Afr J Bot 139:114–121. https://doi.org/10.1016/j.sajb.2021.02.021
Yildiz M, Terzi H, Bingül N (2013) Protective role of hydrogen peroxide pretreatment on defense systems and BnMP1 gene expression in Cr(VI)-stressed canola seedlings. Ecotoxicology 22(8):1303–1312. https://doi.org/10.1007/s10646-013-1117-2
Zeng F, Wu X, Qiu B, Wu F (2014) Physiological and proteomic alterations in rice (Oryza sativa L.) seedlings under hexavalent chromium stress. Planta 240:291–308. https://doi.org/10.1007/s00425-014-2077-3
Zhao Y, Hu C, Wang X, Qing X, Wang P, Zhang Y, Zhang X, Zhao X (2019) Selenium alleviated chromium stress in Chinese cabbage (Brassica campestris L. ssp. Pekinensis) by regulating root morphology and metal element uptake. Ecotox Environ Safe 173:314–321. https://doi.org/10.1016/j.ecoenv.2019.01.090
Funding
This study has been funded by the Scientific Research Projects Coordination Unit (Project No: 20.FEN.BİL.36) of Afyon Kocatepe University.
Author information
Authors and Affiliations
Contributions
All authors have contributed equally to this work.
Corresponding author
Ethics declarations
Ethical Approval
Not applicable.
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Doğuş, H., Yıldız, M., Terzi, H. et al. Evaluation of Selenium Influence on the Alleviation of Chromium Stress in Rapeseed by Physiological and Proteomic Approaches. Plant Mol Biol Rep 41, 559–572 (2023). https://doi.org/10.1007/s11105-023-01384-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11105-023-01384-8