Abstract
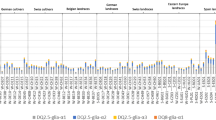
Celiac disease (CD) is an inflammatory disorder that damages the lining of the small intestine. The allergic reaction to the alpha (α)-gliadin class of gluten proteins present in wheat leads to this autoimmune response. α-Gliadin proteins of the wheat gluten form a multigene family with different number and type of CD-eliciting epitopes. The present experiment was planned to study the relative variations in the α-gliadin proteins and their CD-eliciting epitopes in the eight Triticum genotypes including three tetraploid (PDW 274, PBW34, Bansi162) and five hexaploid cultivars (C591, C273, K78, PBW 54, and 9D). Reference-based annotation followed by detailed analysis of unassigned gliadins for domain organization, epitope frequency, and phylogeny relatedness; out of total 43 gliadin genes, 8 were assigned to chromosome 6A, 26 to chromosome 6B, and 9 to 6D. Relative expression analysis revealed that durum wheat genotypes have lower expression of immunogenic epitopes than hexaploid. Chromosome 6A-specific T-cell stimulatory epitope combination was highyly expressed in all the cultivars followed by those from chromosome 6D and least from 6B. The lower proportion of T-cell stimulatory epitopes in PBW54 was due to the lower expression of the 6A encoded epitopic combination that was compensated by the relatively higher expression of non-immunogenic variants forms of 6B. This study is the initial step towards developing wheat that is less immunogenic for celiac disease patients.




Similar content being viewed by others
Abbreviations
- α:
-
Alpha
- BAM:
-
Binary alignment map
- CD:
-
Celiac disease
- NCBI:
-
National Center for Biotechnology Information
- RPKM:
-
Reads per kilobase million
- SAM:
-
Sequence alignment map
- TPM:
-
Transcript per million
References
Alaedini A, Green PH (2005) Narrative review: celiac disease: understanding a complex autoimmune disorder. Ann Intern Med 142(4):289–298. https://doi.org/10.7326/0003-4819-142-4-200502150-00011
Altenbach SB, Chang HC, Rowe MH, Yu XB, Simon-Buss A, Seabourn BW, Alaedini A (2020) Reducing the immunogenic potential of wheat flour: silencing of alpha gliadin genes in a US wheat cultivar. Front Plant Sci 11:20. https://doi.org/10.3389/fpls.2020.00020
Altenbach SB, Chang HC, Yu XB, Seabourn BW, Green PH, Alaedini A (2019) Elimination of omega-1, 2 gliadins from bread wheat (Triticum aestivum) flour: effects on immunogenic potential and end-use quality. Front Plant Sci 10:580. https://doi.org/10.3389/fpls.2019.00580
Anders S, Pyl PT, Huber W (2015) HTSeq—a Python framework to work with high-throughput sequencing data. Bioinformatics 31(2):166–169. https://doi.org/10.1093/bioinformatics/btu638
Barro F, Iehisa JC, Giménez MJ, García-Molina MD, Ozuna CV, Comino I, Gil-Humanes J (2016) Targeting of prolamins by RNA i in bread wheat: effectiveness of seven silencing-fragment combinations for obtaining lines devoid of coeliac disease epitopes from highly immunogenic gliadins. Plant Biotechnol J 14(3):986–996. https://doi.org/10.1111/pbi.12455
Borrill P, Ramirez-Gonzalez R, Uauy C (2016) expVIP: a customizable RNA-seq data analysis and visualization platform. Plant Physiol 170(4):2172–2186
Chen F, Xu C, Chen M, Wang Y, Xia G (2008) A new α-gliadin gene family for wheat breeding: somatic introgression line II-12 derived from Triticum aestivum and Agropyron elongatum. Mol Breed 22(4):675–685. https://doi.org/10.1007/s11032-008-9208-0
Colomba MS, Gregorini A (2012) Are ancient durum wheats less toxic to celiac patients? A study of α-gliadin from Graziella Ra and Kamut. Scientific World J. https://doi.org/10.1100/2012/837416
Edgar RC (2004) MUSCLE: multiple sequence alignment with high accuracy and high throughput. Nucleic Acid Res 32(5):1792–1797. https://doi.org/10.1093/nar/gkh340
FAO (2021) World Food and Agriculture - Statistical Yearbook 2021. Rome.https://doi.org/10.4060/cb4477en
Green PH, Jabri B (2006) Celiac disease. Annu Rev Med 57:207–221. https://doi.org/10.1146/annurev.med.57.051804.122404
Grover J, Chhuneja P, Midha V, Ghia JE, Deka D, Mukhopadhyay CS, Sood A (2019) Variable immunogenic potential of wheat: prospective for selection of innocuous varieties for celiac disease patients via in vitro approach. Front Immunol 10:84. https://doi.org/10.3389/fimmu.2019.00084
Hailegiorgis D, Seid E, Lee C, Yun SJ (2022) Variations in immunodominant epitope and molecular conformation of alpha-gliadins in elite Ethiopian durum wheat cultivars. J Crop Sci Biotechnol 25(3):325–336. https://doi.org/10.1007/s12892-021-00134-0
Hausch F, Shan L, Santiago NA, Gray GM, Khosla C (2002) Intestinal digestive resistance of immunodominant gliadin peptides. Am J Physiol Gastrointest Liver Physiol 283:996–1003. https://doi.org/10.1152/ajpgi.00136.2002
Inomata N (2009) Wheat allergy. Curr Opin Allergy Clin Immunol 9(3):238–243. https://doi.org/10.1097/ACI.0b013e32832aa5bc
International Maize and Wheat Improvement Center (CIMMYT) (2016) Wheat Research, http://www.cimmyt.org/global-wheat-research/(2016). Accessed 23 Dec 2020
International Wheat Genome Sequencing Consortium (IWGSC), Appels R, Eversole K, Stein N, Feuillet C, Keller B, Singh NK (2018) Shifting the limits in wheat research and breeding using a fully annotated reference genome. Sci 361(6403):eaar7191
Juhász A, Békés F, Wrigley CW (2015) Applied food protein chemistry, Ustunol Z, Chapter 11 Wheat Proteins. Michigan
Kaur A, Bains NS, Sood A, Yadav B, Sharma P, Kaur S, Chhuneja P (2017) Molecular characterization of α-gliadin gene sequences in Indian wheat cultivars vis-à-vis celiac disease eliciting epitopes. J Plant Biochem Biotechno 26(1):106–112. https://doi.org/10.1007/s13562-016-0367-5
Kumar S, Stecher G, Li M, Knyaz C, Tamura K (2018) MEGA X: molecular evolutionary genetics analysis across computing platforms. Mole Biol Evol 35(6):1547. https://doi.org/10.1093/molbev/msy096
Malalgoda M, Meinhardt SW, Simsek S (2018) Detection and quantitation of immunogenic epitopes related to celiac disease in historical and modern hard red spring wheat cultivars. Food Chem 264:101–107. https://doi.org/10.1016/j.foodchem.2018.04.131
Molberg Ø, Uhlen AK, Jensen T, Flæte NS, Fleckenstein B, Arentz-Hansen H, Sollid LM (2005) Mapping of gluten T-cell epitopes in the bread wheat ancestors: implications for celiac disease. Gastroenterol 128(2):393–401. https://doi.org/10.1053/j.gastro.2004.11.003
Pearce S, Vazquez-Gross H, Herin SY, Hane D, Wang Y, Gu YQ, Dubcovsky J (2015) WheatExp: an RNA-seq expression database for polyploid wheat. BMC Plant Biol 15(1):1–8. https://doi.org/10.1186/s12870-015-0692-1
Pilolli R, Gadaleta A, Mamone G, Nigro D, De Angelis E, Montemurro N, Monaci L (2019) Scouting for naturally low-toxicity wheat genotypes by a multidisciplinary approach. Scientific Rep 9(1):1–15. https://doi.org/10.1038/s41598-018-36845-8
Pizzuti D, Buda A, D’Odorico A, D’Incà R, Chiarelli S, Curioni A, Martines D (2006) Lack of intestinal mucosal toxicity of Triticum monococcum in celiac disease patients. Scand J Gastroenterol 41(11):1305–1311. https://doi.org/10.1080/00365520600699983
Ramirez-Gonzalez RH, Leggett RM, Waite D, Thanki A, Drou N, Caccamo M, Davey R (2013) StatsDB: platform-agnostic storage and understanding of next generation sequencing run metrics. F1000Research 2. https://doi.org/10.12688/f1000research.2-248.v2
Ribeiro M, Rodriguez-Quijano M, Nunes FM, Carrillo JM, Branlard G, Igrejas G (2016) New insights into wheat toxicity: breeding did not seem to contribute to a prevalence of potential celiac disease’s immunostimulatory epitopes. Food Chem 213:8–18. https://doi.org/10.1016/j.foodchem.2016.06.043
Salentijn EM, Esselink DG, Goryunova SV, van der Meer IM, Gilissen LJ, Smulders MJ (2013) Quantitative and qualitative differences in celiac disease epitopes among durum wheat varieties identified through deep RNA-amplicon sequencing. BMC Genom 14(1):1–16. https://doi.org/10.1186/1471-2164-14-905
Sánchez-León S, Gil-Humanes J, Ozuna CV, Giménez MJ, Sousa C, Voytas DF, Barro F (2018) Low-gluten, nontransgenic wheat engineered with CRISPR/Cas9. Plant Biotechnol J 16(4):902–910
Sapone A, Bai JC, Ciacci C, Dolinsek J, Green PH, Hadjivassiliou M, Fasano A (2012) Spectrum of gluten-related disorders: consensus on new nomenclature and classification. BMC Med 10(1):1–12. https://doi.org/10.1186/1741-7015-10-13
Shan L, Molberg Ø, Parrot I, Hausch F, Filiz F, Gray GM, Khosla C (2002) Structural basis for gluten intolerance in celiac sprue. Science 297(5590):2275–2279
Sharma N, Bhatia S, Chunduri V, Kaur S, Sharma S, Kapoor P, Garg M (2020) Pathogenesis of celiac disease and other gluten related disorders in wheat and strategies for mitigating them. Front Nutr. https://doi.org/10.3389/fnut.2020.00006
Shewry PR, Halford NG, Lafiandra D (2003) Genetics of wheat gluten proteins. Adv Genet 49:111–184. https://doi.org/10.1016/S0065-2660(03)01003-4
Shewry PR, Tatham AS (2016) Improving wheat to remove coeliac epitopes but retain functionality. J Cereal Sci 67:12–21. https://doi.org/10.1016/j.jcs.2015.06.005
Vader LW, Stepniak DT, Bunnik EM, Kooy YM, De Haan W, Drijfhout JW, Koning F (2003) Characterization of cereal toxicity for celiac disease patients based on protein homology in grains. Gastroenterol 125(4):1105–1113. https://doi.org/10.1016/S0016-5085(03)01204-6
Van Herpen TW, Goryunova SV, van der Schoot J, Mitreva M, Salentijn E, Vorst O, Smulders MJ (2006) Alpha-gliadin genes from the A, B, and D genomes of wheat contain different sets of celiac disease epitopes. BMC Genom 7(1):1–13. https://doi.org/10.1186/1471-2164-7-1
Voorrips R (2002) MapChart: software for the graphical presentation of linkage maps and QTLs. J Hered 93(1):77–78. https://doi.org/10.1093/jhered/93.1.77
Acknowledgements
The authors would like to thank the Director of Punjab agricultural University, Ludhiana Panjab, and Executive Director of the National Agri-Food Biotechnology Institute (NABI), Mohali, India for support.
Author information
Authors and Affiliations
Contributions
P. C. and M. G. drafted the manuscript layout and helped in overall supervision during manuscript preparation. Conceptualization—P. C., J. R., I. Y.; methodology—J. R, S.K., I.Y.; software—J. R, S.K., I.Y. validation—P. C., M. G., and S. K; formal analysis—S. K., I. Y.; writing-original draft preparation—M. G. and S. K.; writing-review and editing—S. K., P. C., M. G. supervision—M. G, P. C.; project administration—P. C.; funding acquisition—P. C. All authors have read and agreed to the published version of the manuscript.
Corresponding authors
Ethics declarations
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Key Message
Diversity in alpha-gliadin epitopes open up doors for detection of wheat lines with lower immunogenicity.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Arora, J.K., Kaur, S., Yadav, I. et al. Investigation of Differential Expression of Alpha-Gliadin Genes with Variable Immunogenic Epitopes in Hexa- and Tetraploid Wheat Cultivars Suggests Some Have Lower Immunogenicity. Plant Mol Biol Rep 41, 134–144 (2023). https://doi.org/10.1007/s11105-022-01356-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11105-022-01356-4