Abstract
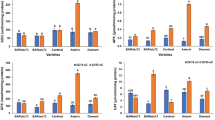
Hydrogen sulfide (H2S) is a gaseous signaling molecule that has been shown to improve drought resistance in plants. In this study, comparative physiological and proteomic analyses were performed to investigate the underlying mechanism by which H2S alleviates drought stress in wheat seedlings. Pretreatment with sodium hydrosulfide (NaHS), an H2S donor, significantly increased height and decreased lipid peroxidation levels in wheat seedlings under drought stress. The antioxidant levels (ascorbate and reduced glutathione), and the contents of starch and soluble sugars were all increased in plants pretreated with NaHS. Using the iTRAQ (isobaric tags for relative and absolute quantitation) technique, 120 proteins were found to be significantly regulated by NaHS under drought stress (57 increased and 63 decreased) compared with drought treatment without NaHS. Functional annotation showed that these proteins are mainly related to carbohydrate metabolism, photosynthesis, signal transduction, protein synthesis, stress, and secondary metabolism. Nine proteins were selected to examine the transcriptional levels of their corresponding genes, and most of them displayed expression patterns similar to the proteomic profiles. These results show that H2S can alleviate drought stress in wheat seedlings by regulating multiple biochemical pathways and that energy and carbon metabolism, signal transduction, antioxidant capacity, and protein synthesis may play important roles in the regulation of drought stress in plants.








Similar content being viewed by others
References
Alexieva V, Sergiev I, Mapelli S, Karanov E (2001) The effect of drought and ultraviolet radiation on growth and stress markers in pea and wheat. Plant Cell Environ 24:1334–1337
Baroja-Fernández E, Muñoz FJ, Akazawa T, Pozueta-Romero J (2001) Reappraisal of the currently prevailing model of starch biosynthesis in photosynthetic tissues: a proposal involving the cytosolic production of ADP-glucose by sucrose synthase and occurrence of cyclic turnover of starch in the chloroplast. Plant Cell Physiol 42(12):1311–1320. https://doi.org/10.1093/pcp/pce175
Bird CR, Koller CR, Auffret AD, Huttly AK, Howel CJ, Dyer TA, Gray JC (1985) The wheat chloroplast gene for CFO subunit I of ATP synthase contains a large intron. EMBO J 6:1381–1388
Blank A, McKeon TA (1989) Single-strand-preferring nuclease activity in wheat leaves is increased in senescence and is negatively photoregulated. Proc Natl Acad Sci U S A 86(9):3169–3173. https://doi.org/10.1073/pnas.86.9.3169
Chaves MM, Flexas J, Pinheiro C (2009) Photosynthesis under drought and salt stress: regulation mechanisms from whole plant to cell. Ann Bot 103(4):551–560. https://doi.org/10.1093/aob/mcn125
Chen J, Wu F, Wang W, Zheng C, Lin G, Dong X, He J, Pei Z, Zheng H (2011a) Hydrogen sulphide enhances photosynthesis through promoting chloroplast biogenesis, photosynthetic enzyme expression, and thiolredox modification in Spinacia oleracea seedlings. J Exp Bot 62(13):4481–4493. https://doi.org/10.1093/jxb/err145
Chen Y, Guo Q, Liu L, Liao L, Zhu Z (2011b) Influence of fertilization and drought stress on the growth and production of secondary metabolites in Prunella vulgaris L. J Med Plant Res 5:1749–1755
Chinkers M (2001) Protein phosphatase 5 in signal transduction. Trends Endocrinol Metab 12(1):28–32. https://doi.org/10.1016/S1043-2760(00)00335-0
Choi Y, Lee Y, Jeon B, Staiger C, Lee Y (2008) Phosphatidylinositol 3- and 4-phosphate modulate actin filament reorganization in guard cells of day flower. Plant Cell Environ 31(3):366–377. https://doi.org/10.1111/j.1365-3040.2007.01769.x
Christou A, Filippou P, Manganaris G, Fotopoulos V (2014) Sodium hydrosulfide induces systemic thermotolerance to strawberry plants through transcriptional regulation of heat shock proteins and aquaporin. BMC Plant Biol 14(1):42. https://doi.org/10.1186/1471-2229-14-42
Christou A, Manganaris G, Papadopoulos I, Fotopoulos V (2013) Hydrogen sulfide induces systemic tolerance to salinity and non-ionic osmotic stress in strawberry plants through modification of reactive species biosynthesis and transcriptional regulation of multiple defence pathways. J Exp Bot 64(7):1953–1966. https://doi.org/10.1093/jxb/ert055
Chu P, Yan G, Yang Q, Zhai L, Zhang C, Zhang F, Guan R (2014) iTRAQ-based quantitative proteomics analysis of Brassica napus leaves reveals pathways associated with chlorophyll deficiency. J Proteome 113:244–259
Conesa A, Götz S, García-Gómez JM, Terol J, Talón M, Robles M (2005) Blast2GO: a universal tool for annotation, visualization and analysis in functional genomics research. Bioinformatics 21(18):3674–3676. https://doi.org/10.1093/bioinformatics/bti610
Despres B, Bouissonnié WH, Gomord V, Guilleminot J, Grellet F, Berger F, Delseny M, Devic M (2003) Three SAC1-like genes show overlapping patterns of expression in Arabidopsis but are remarkably silent during embryo development. Plant J 34(3):293–306. https://doi.org/10.1046/j.1365-313X.2003.01720.x
DeWald D, Torabinejad J, Jones C, Shope J, Cangelosi A, Thompson J, Prestwich G, Hama H (2001) Rapid accumulation of phosphatidylinositol 4, 5-bisphosphate and inositol 1, 4, 5-trisphosphate correlates with calcium mobilization in salt-stressed Arabidopsis. Plant Physiol 126(2):759–769. https://doi.org/10.1104/pp.126.2.759
Foyer C, Rowell J, Walker D (1983) Measurement of the ascorbate content of spinach leaf protoplasts and chloroplasts during illumination. Planta 157(3):239–244. https://doi.org/10.1007/BF00405188
Gao F, Zhou Y, Zhu W, Li X, Fan L, Zhang G (2009) Proteomic analysis of cold stress-responsive proteins in Thellungiella rosette leaves. Planta 230(5):1033–1046. https://doi.org/10.1007/s00425-009-1003-6
Griffith OW (1980) Determination of glutathione and glutathione disulfide using glutathione reductase and 2-vinylpyridine. Anal Biochem 106(1):207–212. https://doi.org/10.1016/0003-2697(80)90139-6
Hernández I, Alegrel L, Munné-Bosch S (2004) Drought-induced changes in flavonoids and other low molecular weight antioxidants in Cistus clusii grown under Mediterranean field conditions. Tree Physiol 24(11):1303–1311. https://doi.org/10.1093/treephys/24.11.1303
Hervé T, Michel Z, Catherine D, Valerie M (2007) Plant proteomics: methods and protocols. Methods Mol Biol 355
Hrazdina G, Jensen RA (2003) Spaial organization of enzymes plant metabolic pathways. Annu Rev Plant Biol 43:241–267
Hu K, Wang Q, Hu L, Gao S, Wu J, Li Y, Zheng J, Han Y, Liu Y, Zhang H (2014) Hydrogen sulfide prolongs postharvest storage of fresh-cut pears (Pyrus pyrifolia) by alleviation of oxidative damage and inhibition of fungal growth. PLoS One 9(1):e85524. https://doi.org/10.1371/journal.pone.0085524
Jin H, Sun Y, Woe Y, Young J, Ho B, Hyun S, Chang H, Mi R, Sun Y, Mukhamad S, Dae J, Kyun O, Min G, Sang Y (2011) Heat-induced chaperone activity of serine⁄threonine protein phosphatase 5 enhances thermotolerance in Arabidopsis thaliana. New Phytol 191:692–705
Li W, Li M, Zhang W, Welti R, Wang X (2004) The plasma membrane-bound phospholipase ddelta enhances freezing tolerance in Arabidopsis thaliana. Nat Biotechnol 22(4):427–433. https://doi.org/10.1038/nbt949
Lisjak M, Teklic T, Wilson ID, Wood ME, Whiteman M, Hancock JT (2011) Hydrogen sulfide effects on stomatal apertures. Plant Signal Behav 6(10):1444–1446. https://doi.org/10.4161/psb.6.10.17104
Liu ZY, Zhang YH, Tong W, Wang H, Zhao WG, Long LI (2012) Molecular cloning, sequence feature and induced expression analysis of galactokinase gene in mulberry. Sci Seric 38:959–967
Livak KJ, Schmitthen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCT method. Methods 25(4):402–408. https://doi.org/10.1006/meth.2001.1262
Takabayashi A, Ishikawa N, Obayashi T, Ishida S, Obokata J, Endo T, Sato F (2009) Three novel subunits of Arabidopsis chloroplastic NAD(P)H dehydrogenase identified by bioinformatic and reverse genetic approaches. Plant J 57(2):207–219. https://doi.org/10.1111/j.1365-313X.2008.03680.x
Ma DY, Sun DX, Wang CY, Li YG, Guo TC (2014) Expression of flavonoid biosynthesis genes and accumulation of flavonoid in wheat leaves in response to drought stress. Plant Physiol Biochem 80:60–66. https://doi.org/10.1016/j.plaphy.2014.03.024
Ma D, Ding H, Wang C, Qin H, Han Q, Hou J, Lu H, Xie Y, Guo T (2016) Alleviation of drought stress by hydrogen sulfide is partially related to the abscisic acid signaling pathway in wheat. PLoS One 11(9):e0163082. https://doi.org/10.1371/journal.pone.0163082
Mogensen VO, Jensen HE, Rab MA (1985) (1985) Grain yield, yield components, drought sensitivity and water use efficiency of spring what subjected to water stress at various growth stages. Irrigation Sci 6:131–140
Moriya Y, Itoh M, Okuda S, Yoshizawa AC, Kanehisa M (2007) KAAS: an automatic genome annotation and pathway reconstruction server. Nucleic Acids Res 35:182–185
Ngara R, Ndimba R, Borchjensen J, Jensen ON, Ndimba B (2012) Identificaftion and profiling of salinity stress-responsive proteins in sorghum bicolor seedlings. J Proteome 75(13):4139–4150. https://doi.org/10.1016/j.jprot.2012.05.038
Ndimba BK, Chivasa S, Simon WJ, Slabas AR (2005) Identification of Arabidopsis salt and osmotic stress responsive proteins using two-dimensional difference gel electrophoresis and mass spectrometry. Proteomics 5(16):4185–4196. https://doi.org/10.1002/pmic.200401282
Pružinská A, Tanner G, Anders I, Roca M, Hörtensteiner S (2004) ChlorophyII breakdown: pheophorbide a oxygenase is a Rieske-type iron-sulfur protein, encoded by the accelerated cell death 1 gene. Proc Natl Acad Sci U S A 100:15259–15264
Rhee S, Bae Y (1997) Regulation of phosphoinositide-specific phospholipase C Isozymes. J Biol Chem 272(24):15045–15048. https://doi.org/10.1074/jbc.272.24.15045
Schmidt A (2005) Metabolic background of H2S release from plants. Landbauforschung Völkenrode 283:121–129
Shen Y, Wang W, Zhang W, Zhu L, Li B (2015) Hydrogen sulfide facilitating enhancement of antioxidant ability and maintainance of fruit quality of kiwifruits during low-temperatures storage. Trans CSAE 31:367–372
Shi H, Wang B, Yang P, Li Y, Miao F (2016) Differences in sugar accumulation and mobilization between sequential and non-sequential senescence wheat cultivars under natural and drought conditions. PLoS One 11(11):e0166155. https://doi.org/10.1371/journal.pone.0166155
Shi H, Wang X, Tan D, Reiter R, Chan Z (2015) Comparative physiological and proteomic analyses reveal the actions of melatonin in the reduction of oxidative stress in Bermuda grass (Cynodon dactylon L. Pers). J Pineal Res 59(1):120–131. https://doi.org/10.1111/jpi.12246
SPSS (2006) SPSS version 15.0 for Windows. SPSS Inc., Chicago
Stephenson TJ, Mcintyre CL, Collet C, Xue GP (2007) Genome-wide identification and expression analysis of the NF-Y family of transcription factors in Triticum aestivum. Plant Mol Biol 65(1-2):77–92. https://doi.org/10.1007/s11103-007-9200-9
Stobrawa K, Lorenc-Plucinska G (2007) Changes in carbohydrate metabolism in fine roots of the native European black poplar (Popuplus nigra L.) in a heavy-metal-polluted environment. Sci Tot Environ 373(1):157–165. https://doi.org/10.1016/j.scitotenv.2006.11.019
Ross P, Huang Y, Marchese J, Williamson B, Parker K, Hattan S, Khainovski N, Pillai S, Dey S, Daniels S, Purkayastha S, Juhasz P, Martin S, Bartlet-Jones M, He F, Jacobson A, Pappin D (2004) Multiplexed protein quantitation in Saccharomyces cerevisiae using amine-reactive isobaric tagging reagents. Mol Cell Proteomics 3(12):1154–1169. https://doi.org/10.1074/mcp.M400129-MCP200
Uraji M, Katagiri T, Okuma E, Ye W, Hossain MA, Masuda C, Miura A, Nakamura Y, Mori IC, Shinozaki K, Murata Y (2012) Cooperative function of PLD and PLD1 in abscisic acid-induced stomatal closure in Arabidopsis. Plant Physiol 159(1):450–460. https://doi.org/10.1104/pp.112.195578
Wang Y, Qu Z, Zhang Y, Ma J, Guo J, Han Q, Huang L, Kang Z (2008) Construction of a cDNA library and analysis of expressed sequence tags in association with the incompatible interaction between wheat and Puccinia striiformis. Sci Agric Sin 41:3376–3381
Wang Y, Shen W, Chan Z, Wu Y (2015) Endogenous cytokinin overproduction modulates ROS homeostasis and decreases salt stress resistance in Arabidopsis thaliana. Front Plant Sci 6:8
Wisniewski JR, Zougman A, Nagaraj N, Mann M (2009) Universal sample preparation method for proteome analysis. Nature Method 6(5):359–362. https://doi.org/10.1038/nmeth.1322
Zhang H, Jiao H, Jiang CX, Wang SH, Wei ZJ, Luo JP, Jones R (2010a) Hydrogen sulfide protects soybean seedlings against drought-induced oxidative stress. Acta Physiol Plant 32(5):849–857. https://doi.org/10.1007/s11738-010-0469-y
Zhang H, Tan Z, Hu L, Wang S, Luo J, Jones R (2010b) Hydrogen sulfide alleviates aluminum toxicity in germinating wheat seedlings. J Integr Plant Biol 52(6):556–567. https://doi.org/10.1111/j.1744-7909.2010.00946.x
Zheng Y, Jia A, Ning T, Xu J, Li Z, Jiang G (2008) Potassium nitrate application alleviates sodium chloride stress in winter wheat cultivars differing in salt tolerance. J Plant Physiol 165(14):1455–1465. https://doi.org/10.1016/j.jplph.2008.01.001
Acknowledgements
This work was supported by the Science and Technology Support Program [grant number 2015BAD26B00] and the Key Scientific Research Project of Higher Education Institution [grant number 15A210004].
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Ding, H., Han, Q., Ma, D. et al. Characterizing Physiological and Proteomic Analysis of the Action of H2S to Mitigate Drought Stress in Young Seedling of Wheat. Plant Mol Biol Rep 36, 45–57 (2018). https://doi.org/10.1007/s11105-017-1055-x
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11105-017-1055-x