Abstract
Background and aims
Repeated surface application of lime for managing subsoil acidity is slow and ineffective, resulting in an accumulation of undissolved lime (carbonate) in the topsoil. We investigated the impact of the incorporation of undissolved lime into the subsoil to improve acidity and crop performance.
Methods
The undissolved lime in 2-cm layers of topsoil (0–10 cm) from three long-term experiments in Western Australia was measured. Both limed and unlimed topsoil with the acidic subsoil of the same profile was incubated at eight incorporation rates for six weeks, followed by growing barley and wheat in the incubated soil for two weeks to assess the impact on soil acidity and crop root architecture, respectively. Furthermore, a three-year-long field experiment was conducted following strategic tillage in limed and control plots to assess the impact on soil acidity and performance of wheat, canola and barley.
Results
A significant amount of undissolved lime was concentrated in the topsoil, amounting to 1.7, 1.8 and 1.3 t/ha for the limed plots at Wongan Hills, Northam and Merredin, respectively. Incubation of 5–25% topsoil after incorporation with the acidic subsoil was enough to ameliorate subsoil acidity and to improve root length density by up to 13-fold depending on undissolved lime content in topsoils and soil type. In the field experiment, the incorporation of undissolved lime also significantly improved subsoil acidity and canola performance.
Conclusion
We concluded that the incorporation of topsoil containing sufficient undissolved lime with acidic subsoil may offer a quick amelioration of subsoil acidity.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Globally, approximately half of arable land is affected by soil acidity, which constitutes a significant challenge to agricultural productivity (Lollato et al. 2019). As soil pH decreases, the concentration of aluminium (Al) in the soil solution increases exponentially (Delhaize and Ryan 1995). Extractable aluminium concentrations (EAC) greater than 5 mg/kg become toxic to both Al-sensitive and tolerant plant species and damage plant root architecture, including root hairs, root length density and rooting depth (Delhaize and Ryan 1995; Gazey 2018; Azam and Gazey 2020). Low soil pH also decreases the availability of essential plant nutrients, such as phosphorus (Rahman et al. 2021, 2023) and ultimately reduces crop yield (Tang et al. 2003; Anderson et al. 2013).
In Australia, the impact of soil acidity is pronounced, with 50% of agricultural land constrained by low soil pH (National Land and Water Resources Audit 2001). Notably, around 23 million hectares of subsurface soil (below 10 cm from the surface up to 30–40 cm depth) (hereafter subsoil) exhibit acidic conditions. In the south-western region of Western Australia (WA), which serves as the primary grain crop growing area in WA, 72% of the topsoil (0–10 cm) samples and 45% of the subsoil samples had soil pH below the target levels for acidic soil (Gazey et al. 2013). In WA, minimum pH targets for acidic soil are 5.5 in the topsoil (0–10 cm) and 4.8 in the subsoil soil measured in 0.01 M CaCl2 extract (Gazey and Davies 2009). The consequences of soil acidity are substantial, with estimated annual penalties from grain yield loss of up to 1.6 billion Australian dollars in WA, assuming acidity is the only constraint present (Petersen 2016).
The application of agricultural lime, including limesand, crushed limestone, and crushed dolomite, is a widely adopted and economical strategy to increase the pH of acidic soil and to mitigate Al toxicity (Dodgshon 1978; Anderson et al. 2013). Lime application has also notably increased crop yield in Australia and other countries (Tang et al. 2003; Holland et al. 2019). An extensive agricultural liming program was implemented across the south-western region of WA, particularly in the late 1990s (Gazey et al. 2014). The traditional approach involves applying agricultural lime (often multiple applications over several years) on the soil surface, without incorporation into the subsoil primarily due to the adoption of the global practice of no-till cropping (Fischer and Hobbs 2019; Kassam et al. 2019; Llewellyn and Ouzman 2019). A survey of Australian grain growers showed that 80–90% of the soil in Australia does not undergo pre-sowing cultivation (Umbers 2017). Since 2006, lime application rates in WA have increased (Gazey et al. 2019), with farmers advised to apply sufficient lime to achieve and maintain topsoil and subsoil pH targets (Gazey and Davies 2009). In WA, 8.5 t/ha of lime over 24 years is the highest cumulative rate applied in a field experiment (Azam and Gazey 2020). However, the movement of agricultural lime (e.g. limesand) from the topsoil to the subsoil is slow in the semi-arid environment of southern Australia (Azam and Gazey 2020; Hume et al. 2023), which is likely to accumulate as undissolved lime in the topsoil, with the subsoil pH remaining below the target level.
Due to the limited movement of lime into the subsoil, especially in an environment with less than 400 mm annual rainfall, lime incorporation with tillage operations has been recommended for the physical distribution of lime throughout the depth of acidic soil layers and a quick increase in the subsoil pH (Scanlan et al. 2014). Recently, strategic tillage (one-off need-based tillage) has gained popularity as a method to address various soil constraints, including soil acidity, water repellence and compaction (Davies et al. 2020; Azam et al. 2023). Strategic tillage involves mixing the topsoil with the subsoil, leading to a quick improvement in subsoil pH (Azam and Gazey 2020; Hume et al. 2023). However, the amount of undissolved lime in the topsoil and the potential benefit of incorporating it into the acidic subsoil remained unknown.
In this research, we performed a comprehensive investigation involving (i) three long-term lime experiments, (ii) a laboratory incubation study, (iii) a plant growth bioassay experiment, and (iv) a three-year-long field trial. The primary objectives were to: (i) assess the undissolved lime status in three long-term lime experiments, (ii) examine the effects of incubating topsoil containing undissolved lime with acidic subsoil on pH and EAC, (iii) evaluate the impact of mixing undissolved lime into subsoil on plant root growth, and (iv) analyse the effect of incorporating topsoil containing undissolved lime, with acidic subsoil on soil pH, EAC, and crop yield under field conditions.
Methods and materials
Measurement of soil pH, EAC and undissolved lime in three long-term field experiments - case studies
In April 2018, soil samples were collected from three long-term soil acidity management experiments monitored by the Department of Primary Industries and Regional Development located in Wongan Hills (WH) (24 years), Merredin (ME) (10 years) and Northam (NO) (6 years) in WA (Table 1). Soil samples were collected from three replicated plots of the limed and control (unlimed) treatments. The selected limed plots at WH received a total of 8.5 t/ha (4 t/ha in 1994, 1.5 t/ha in 1998, and 3 t/ha in 2014) limesand. The limed plots at ME received a total of 6 t/ha (3 t/ha in 2008 and 3 t/ha in 2014) limesand, while the NO limed plots received a single high application rate of 5 t/ha limesand in 2012. More details on these experimental sites can be found in Table 1 and Azam and Gazey (2020).
Topsoil samples were systematically collected from a soil pit face at 2-cm depth intervals, spanning 0–2, 2–4, 4–6, 6–8, and 8–10 cm depths in both limed and control plots. Four samples were collected from each plot and bulked. The analysis encompassed measurements of soil pH (Method 4B1, Rayment and Lyons 2011), EAC (Bromfield 1987) and soil undissolved lime (carbonate content) utilising the pressure calcimeter principle (Horváth et al. 2005). Horváth et al. (2005) method was used as the cost of the equipment was low and we found it was reasonably accurate to differentiate between a limed soil from an untreated acidic control soil. Carbonate concentration was then converted into the amount per hectare by multiplying it with the volume and bulk density of the soil in the 0–10 cm soil. Additionally, a separate set of soil core samples (4 cores per plot, bulked at the same depth interval) was obtained from 0 to 10, 10–20, 20–30, 30–40, 40–50, and 50–60 cm depths for the determination of soil pH (Method 4B1, Rayment and Lyons 2011) and EAC (Bromfield 1987). All soil samples were subjected to drying at 40 °C in a forced-draught oven and subsequently passed through a 2 mm sieve before undergoing chemical analysis.
Laboratory incubation study
Based on the undissolved lime contents in the case studies, approximately 25 kg of bulk topsoil samples from the 0–4 cm depth and approximately 50 kg of bulk subsoil samples from the 20–30 cm depth (representing the most acidic layer in the soil profile) were collected of three replicated limed and control plots at each of the three field experimental sites (WH, NO and ME). The topsoils (0–4 cm depth) of both limed and control plots were incubated in petri dishes (90 mm diameter, 14 mm height) with acidic subsoils (20–30 cm depth) of the same plot after incorporating at the rate of 0 (subsoil only), 5, 10, 15, 20, 25, 50 and 100% (topsoil only) (total 200 g of soil), giving the experimental design of 3 soils x 2 lime treatments x 8 incorporation rates x 3 replications (a total of 144 Petri dishes). The incorporated soils were incubated at 25 °C and wetted to field capacity (10 g/g gravimetric water content) and maintained throughout the experiment. After six weeks of incubation, a subsample of around 20 g soil was oven-dried at 40 °C and ground before undergoing analysis for pH (Method 4B1, Rayment and Lyons 2011) and EAC (Bromfield 1987).
Root growth in incubated soil in a growth chamber
After six weeks of incubation, 2 × 90 g subsamples were taken from each Petri dish and filled in two cylindrical plastic vials (10 cm deep x 3 cm diameter). One wheat (Triticum aestivum L. var Scepter) seed was planted in one vial, and one barley (Hordeum vulgare L. var La Trobe) seed was planted in the other vial, giving the experimental design of 2 plant species x 3 soils x 2 lime rates x 8 incorporation rates x 3 replications (a total of 288 vials). Before planting, seeds were hydrated in a petri dish for three days until the first roots began to grow and the coleoptile started to appear (kept in a growth chamber at 25 °C and 80% relative humidity) to enable plants to be grown with uniform germination. One germinated seed was transplanted in each vial at 1 cm below the soil surface and allowed to grow for two weeks in the growth chamber before they were harvested. Soils were wetted to field capacity and maintained throughout the experiment. At harvesting, plant roots were separated from the soil using a gentle jet of water over a 0.5 mm sieve. All roots from each vial were scanned using a high-resolution scanner (600 dpi, Epson Perfection V800 Scanner, Epson Australia Pty Ltd.). Total and fine root length density (RLD), root surface area and diameter were measured using WinRhizo software (v. 2005c; Régent Instruments Inc., Quebec, Canada). In this study the maximum diameter of the barley and wheat roots remained below 2.0 mm, therefore, a maximum threshold of 2.0 mm was applied to separate roots from any other plant materials. While a threshold diameter of 0.5 mm was applied to separate fine roots from the rest of the roots.
Field trial: incorporation of limed topsoil into the acidic subsoil using a rotary hoe
The field experiment was conducted at the Wongan Hills site which received three applications of limesand between 1994 and 2014 (Table 2). The trial was established in 1994 using a randomised design with four replications (paired plots of 1.8 m x 30 m) with three lime rates of 0, 2 and 4 t/ha (Table 2). In 1998, one plot from each pair was re-limed with 1.5 t/ha, altering the design to a split-plot design (single plots of 1.8 m x 30 m) (Table 2). In 2014, an additional 3 t/ha of lime was surface applied to half of each plot (single plots of 1.8 m x 15 m) (Table 2).
In April 2018, a strategic deep tillage treatment was applied on both limed and control plots at the WH site to incorporate topsoil with undissolved lime with the acidic subsoil. The plots were subdivided into three incorporation treatments at depths of 0, 15 and 25 cm (plots of 1.8 m x 5 m) using a rotary hoe (Howard, HR20). However, 15 cm incorporation depth was not precisely achieved, hence data were not collected for this depth treatment. The selected lime treatments for the study are listed in Table 2. Soil samples (four cores per depth interval per plot and bulked) were collected in 2020 from the depths of 0–10, 10–20, and 20–30 cm using a stainless-steel pipe with a 40 mm inner diameter, to measure pH (Method 4B1, Rayment and Lyons 2011) and EAC (Bromfield 1987). The trial was consistently cropped from 2018 to 2020 with a sequence of wheat, canola, and barley. Crops were harvested using a combine plot harvester (ZÜRN 150, Wintersteiger, Ried im Innkreis, Austria), and yields were measured. Water use efficiency (WUE) was estimated using methods described by French and Schultz (1984).
Statistical analysis
GenStat (Version 22, VSN International, Oxford, UK) software was used to conduct statistical analyses. A linear model was employed for the ANOVA for each measured parameter to compare treatment effects with polynomial contrasts. At each soil sampling depth, lime rates were considered as the factor and soil pH, EAC and the amount of undissolved carbonate as the variate. For comparing the yield response in each year, lime rates and incorporation depths were considered as factors while yield was considered as variate. A linear mixed model was employed in GenStat to account for the random effects from the repeated liming applications in WH and ME experiments. The model utilised the restricted maximum likelihood (REML) algorithm directive for its application. Fisher’s protected least significant difference (LSD) was applied at the 95% significance level to assess differences among the treatments.
Results
Status of soil pH, EAC and undissolved lime content in three long-term experiments
The initial top and subsoil pH (measured at the beginning of the study in 2018) was below the target levels in most of the soil samples collected from the control plots of all sites except for the 40–60 cm soil depth at WH and NO (Fig. 1a, b and c). The control topsoil pH ranged from 4.3 to 5.1 for all the soils collected from all the locations (Fig. 1a, b and c). The subsoil pH of the control plots ranged from 3.8 to 4.5. The lime application significantly increased top- and subsoil pH compared to the control plots at all the locations (Fig. 1a, b and c). Lime application elevated topsoil pH above the target, ranging from pH 5.9 to 6.5 for all the locations. In the subsoil, especially in 10–30 cm, none of the lime-treated plots achieved the target pH level of 4.8 at all locations excepth for the 10–20 cm depth at WH.
Stratification of soil pH (a, b and c) and exchangeable aluminium concentration (EAC) (d, e and f) at 10 cm depth intervals to 60 cm soil depth in 2018 at Wongan Hills (a and d), Northam (b and e) and Merredin (c and f) sites in Western Australia. The horizontal bars indicate the standard error of the mean values of the respective parameters. The LSD at 5% for a = 0.56, b = 0.46, c = 0.43, d = 4.17, e = 5.27 and f = 8.62
The EAC varied among different sampling depths and sites. The highest EAC was found at the control plot of ME site (18.6 mg/kg at 50–60 cm depth, Fig. 1f), followed by the NO site (15.3 mg/kg at 10–20 cm depth, Fig. 1e), and the WH site (10.2 mg/kg, at the 20–30 cm depth, Fig. 1d). The addition of lime decreased the EAC concentration below the toxic level in both top- and subsoils of all the sites except for 20–30 cm and below 40 cm at ME (Fig. 1d, e and f). The lime application significantly decreased topsoil EAC to 0.2 mg/kg at NO and ME and 0.6 mg/kg at WH, compared to EAC of 4.3, 3.7 and 2.7 mg/kg in the control topsoils at NO, ME and WH, respectively (Fig. 1d, e and f). The addition of lime significantly decreased the subsoil EAC as low as 0.1 mg/kg at 30–40 cm depth at NO, followed by 0.7 mg/kg at WH at 30–40 cm depth at WH and 1.9 mg/kg at ME at 10–20 cm depth.
When the soil samples were collected from the pit face at 2 cm intervals to 10 cm depth, soil pH and EAC were significantly stratified within the topsoil. Soil pH decreased (Fig. 2a, b and c) and EAC increased (Fig. 2d, e and f) as the soil depth increased for both the limed and control plots. The highest pH (ranging from 6.6 to 7.0 was recorded in the top 2 cm of soil for all the limed plots and the lowest pH (ranged from 5.1 to 5.7) at 8–10 cm. The average topsoil (0–10 cm) pH in the control plots was 5.2, 4.3 and 4.6 compared to pH of 6.4, 6.2 and 6.3 in the limed plots at WH, NO and ME sites, respectively. The average topsoil EAC was 1.6, 6.3 and 6.0 mg/kg for the control and < 0.2, 0.3 and 0.3 mg/kg for the limed plots at WH, NO and ME sites, respectively.
Stratification of soil pH (a, b and c), exchangeable aluminium concentration (EAC) (d, e and f) and undissolved lime content (g, h and i) (CO3, g/kg) at 2 cm depth intervals to10 cm in 2018 at Wongan Hills (a, d and g), Northam (b, e and h) and Merredin (c, f and i) sites in Western Australia. The horizontal bars indicate the standard error of the mean values of the respective parameters. The LSD at 5% for a = 0.72, b = 0.55, c = 0.65, d = 1.14, e = 4.40, f = 3.50, g = 0.64, h = 1.02 and i = 0.74
There was a significant difference in undissolved lime content between sampling depths and lime treatments (Fig. 2g, h and i). The highest carbonate content was recorded in the 0–2 cm depth (3.3 g/kg at WH, 4.5 g/kg at NO and 1.6 g/kg at ME sites) and then decreased as the depth increased at all the locations. The average topsoil carbonate content was 1.20, 1.30 and 0.90 g/kg for the limed and 0.60, 0.30 and 0.23 g/kg for the control plots at WH, NO and ME sites, respectively. The total undissolved lime was equivalent to 1.7, 1.8 and 1.3 t/ha of lime in the topsoil for the limed plots at WH, NO and ME, respectively.
Effect of incorporation of limed topsoil on soil pH and EAC in incubation study
The incorporation of topsoil from both limed and control plots with the acidic subsoil increased soil pH and decreased EAC (Fig. 3). For all sites, the incorporation of limed topsoil with acidic subsoil had a significantly greater increase in the soil pH compared to the incorporation of control topsoil (Fig. 3a, b and c). In particular, the incorporation of less than 10% limed topsoil (from WH and NO) increased subsoil pH above the target pH (4.8) and decreased EAC below the toxic level (Fig. 3d and e). In contrast, subsoil pH did not increase to the target with the incorporation of the highest rate of the control topsoils from NO and ME sites (Fig. 3b and c). The effect of the incorporation of control topsoils was more prominent on subsoil EAC than on pH, as EAC decreased below the toxic level but at higher topsoil incorporation rates (more than 50% in NO and 100% in ME) compared to the limed soil (Fig. 3e and f).
The effect of incubation of the limed and control topsoil with the acidic subsoil on pH (a, b, and c) and EAC (d, e and f) at Wongan Hills (a and d), Northam (b and e) and Merredin (c and f) soils. The vertical lines indicate the standard error of the mean values. The LSD at 5% for a = 0.11, b = 0.05, c = 0.04, d = 0.41, e = 0.68 and f = 1.11
There was a strong exponential relation between soil pH and EAC for both the limed and control plots (r2 value ranged from 0.88 to 0.99) (Appendix Fig. 8). The relationship between pH and EAC was similar (fitted lines were closer to each other) for the limed soil at all sites (r2 value ranged from 0.88 to 0.94, Appendix Fig. 8b). However, there was a difference for the control soils from different sites (r2 value ranged from 0.92 to 0.99, Appendix Fig. 8a). In the control soil, at the same soil pH (particularly at pH 4–4.2), the NO soil had a higher EAC, followed by WH and ME. At the low pH range (pH < 4), the ME soil had a higher EAC than the WH soil (Appendix Fig. 8a).
Effect of incorporation of limed topsoil with the acidic subsoil on crop root architecture
Barley roots
The subsoils from the limed plots without the incorporation (% topsoil) had 2.4–4.2 times greater total RLD (Fig. 4a, b and c) and significantly smaller root diameters (Appendix Fig. 9) compared to the subsoils (0% topsoil) from the control plots for all the locations. The impact of the incorporation of limed topsoil was stronger on the fine RLD (roots with a diameter ≤ 0.5 mm, Appendix Fig. 10) than on the total RLD (Fig. 4a, b and c). For example, the incorporation of 50% limed topsoil with subsoil increased total RLD by 6.1–17.1 times whereas the fine RLD increased by 13.3–43.6 times compared to the subsoils (0% topsoil) from control plots for all the locations. Similarly, subsoils from limed plots without the incorporation of topsoil (0%) had 1.6–2.5 times greater root surface area of barley compared to the subsoils (0% topsoil) from the control plots for all the locations (Fig. 5a, b and c). The incorporation of 50% limed topsoil with subsoil increased surface area by 3.1–6.2 times compared to the subsoils (0% topsoil) from control plots for all the locations (Fig. 5a, b and c).
Wheat roots
The subsoils from the limed plots without the incorporation of topsoil (0%) had 1.4–2.9 times greater RLD (Fig. 4d, e and f) of wheat compared to the subsoils (0% topsoil) from control plots for all the locations. The impact of the incorporation of limed topsoil was similar on both fine RLD (roots with a diameter ≤ 0.5 mm, Appendix Fig. 10) and total RLD (Fig. 4d, e and f) of wheat. The incorporation of 50% limed topsoil with the subsoil increased total RLD by 1.4–1.8 times whereas fine RLD increased by 1.6–1.7 times compared to the subsoils (0% topsoil) from the limed plots for all the locations. Subsoils from the limed plots without the incorporation of topsoil (0%) had only 1.1–1.5 times greater root surface area of the wheat compared to the subsoils (0% topsoil) from the control plots (Fig. 5a, b and c) for all the locations. The incorporation of 50% limed topsoil with subsoil increased the root surface area of wheat by 1.2–2.5 times compared to subsoils (0% topsoil) from control plots (Fig. 5d, e and f).
The effect of incorporation of the limed and control topsoil with the acidic subsoil on the barley (a, b, and c) and wheat (d, e and f) root length density in soils from Wongan Hills (a and d), Northam (b and e) and Merredin (c and f). A threshold of 2.0 mm was applied to separate roots from any other plant materials. The vertical lines indicate the standard error of the mean values. The LSD at 5% for a = = 0.35, b = 0.25, c = 0.23, d = 0.42, e = 0.29 and f = 0.34
The effect of incorporation of the limed and control topsoil with the acidic subsoil on the root surface area of barley (a, b, and c) and wheat (d, e and f) in soils from Wongan Hills (a and d), Northam (b and e) and Merredin (c and f). A threshold of 2.0 mm was applied to separate roots from any other plant materials. The vertical lines indicate the standard error of the mean values. The LSD at 5% for a = 2.42, b = 2.14, c = 1.93, d = 2.68, e = 2.29 and f = 3.02
Effect of incorporation of limed topsoil with the acidic subsoil on soil pH and EAC- field experiment
Soil pH
The addition of lime to the topsoil (0–10 cm), irrespective of rates and incorporation, significantly increased pH except for L2 compared to the control (L0) (Fig. 6a). Among all the lime rates, L2.2 and L4.2 recorded significantly higher topsoil pH compared to all other lime rates (Fig. 6a). At 10–20 cm depth, without incorporation, soil pH significantly increased but only with the higher rates of lime (≥ 4 t/ha) compared to L0, whereas the incorporation of limed topsoil significantly increased pH at this depth with all the lime rates except for L2.1 (Fig. 6b). The incorporation of limed topsoil with the acidic subsoil had up to 0.7 units higher pH compared to without incorporation treatments with all the lime rates at 10–20 cm depth (Fig. 6b). At 20–30 cm depth, without incorporation, none of the lime rates increased the soil pH above the target (Fig. 6c). With incorporation, only the highest lime rate (L4.2) reached the target soil pH (Fig. 6c). The incorporation of limed topsoil with the acidic subsoil increased soil pH by up to 0.4 units compared to without incorporation treatment at 20–30 cm depth (Fig. 6c).
The effect of incorporation of limed topsoil with acidic subsoil on soil pH (a, b and c) and EAC (d, e and f) at the field experiment at Wongan Hills. Lime rates L0, L2, L2.1, L4, L4.1, L2.2 and L4.2 indicate the total lime addition at the rate of 0, 2, 3.5, 4, 5.5, 6.5 and 8.5 t/ha from 1994 to 2014 at Wongan Hills. The vertical bars indicate the standard error of the mean values of the respective variables. The LSD at 5% for a = 0.38, b = 0.50, c = 0.34, d = 0.10, e = 1.10 and f = 2.29
Soil EAC
In the topsoil (0–10 cm), there was no difference in EAC between lime rates and incorporation treatments. The EAC were below the toxic level (5 mg/kg) in the topsoil (Fig. 6d). All the plots also had a lower EAC than the toxic level at this 10–20 cm depth (Fig. 6e). At 10–20 cm depth, with or without incorporation, higher rates of lime (≥ 4 t/ha) had significantly lower EAC compared to the L0 (Fig. 6e). At 10–20 cm depth, with incorporation, lower rates of lime (L2 and L2.1) significantly decreased EAC compared to the without incorporation treatment. At 20–30 cm depth, L0 recorded the EAC above the toxic level with or without incorporation (Fig. 6f). Without incorporation, all lime rates greater than 2 t/ha had significantly lower EAC than the L0 and L2 at 20–30 cm (Fig. 6f). With the incorporation all lime rates had lower EAC than the toxic level at 20–30 cm (Fig. 6f). With incorporation, L2 had significantly lower EAC than the same rate of lime without incorporation at 20–30 cm depth (Fig. 6f).
Effect of incorporation of limed topsoil with acidic subsoil on crop yield and water use efficiency (WUE)
Crop yield
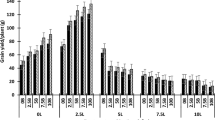
The addition of lime resulted in a significant increase in grain yield compared to the control (L0) depending on the lime rate, incorporation of undissolved lime and the crop (Fig. 7a). The addition of lime without incorporation significantly increased wheat yield with L4 in 2018, canola yield with L4.1 and L4.2 in 2019 and barley yield with all the lime rates in 2020 compared to the control (L0). The incorporation of topsoil containing undissolved lime from the plots with higher lime rates (L4.2) significantly increased canola (2019) yield compared to the control (Fig. 7a). There was no significant difference in the yield between incorporation and without incorporation treatments, irrespective of lime rates, for wheat (2018) and barley (2020). The highest yield advantage was recorded by barley (up to 1.6 t/ha), followed by wheat (up to 1.2 t/ha) and canola (up to 0.3 t/ha) compared to the control.
The effect of incorporation of limed topsoil with acidic subsoil on (a) yield and (b) water use efficiency (WUE). Lime rates L0, L2, L4, L2.1, L4.1, L2.2 and L4.2 indicate the total lime addition at the rate of 0, 2, 4, 3.5, 5.5, 6.5 and 8.5 t/ha from 1994 to 2014 at Wongan Hills. The vertical bars indicate the standard error of the mean values of the respective variables. The LSD was at 5% for yield 0.76, 0.16 and 0.86 and for WUE 2.99, 0.91 and 6.09 in 2018 (wheat), 2019 (canola) and 2020 (barley), respectively
Water Use Efficiency (WUE)
As grain yield increased with the lime application, the WUE of wheat, canola and barley also increased (Fig. 7b). Without the incorporation, all lime rates significantly increased the WUE of wheat, canola and barley crops compared to the control. WUE of wheat increased from 11.4 kg/mm/ha in L0 to 14.6 kg/mm/ha in L4, of canola from 7.0 kg/mm/ha in L0 to 8.0 kg/mm/ha in L4.2 and of barley from 23.4 kg/mm/ha in L0 to 34.7 kg/mm/ha in L4.2. With the incorporation of undissolved lime in the plots with higher rates (L4.2) significantly increased canola WUE compared to the control and showed no significant difference for wheat and barley (Fig. 7b).
Discussion
The pH of the control soils from all locations were below the target levels in both top and subsoil as of 2018 (Fig. 1), 6 to 24 years after the first application of lime, in this study. Surface application of lime without incorporation raised topsoil pH above the target level across all locations. However, subsoil pH remained below the target, specifically at 10–60 cm at ME, 10–30 cm at NO, and 20–30 cm at WH sites. Previous studies also reported an insignificant increase in subsoil pH following surface application of lime (Anderson et al. 2013; Kisinyo et al. 2014; Rheinheimer et al. 2018; Azam and Gazey 2020; Kuma et al. 2022; Hume et al. 2023). Slow improvement in subsoil pH from liming was attributed to the limited solubility of lime when there is no contact with acidic soil (Anderson et al. 2013) and slow movement of alkali from lime down the soil profile (Sumner et al. 1986; Conyers and Scott 1989; Azam and Gazey 2020) in an environment where annual rainfall is less than 400 mm. The experimental sites are situated in a semi-arid zone (ten-year average rainfall range from 319 to 395 mm, Table 1) where soils remain dry for nearly six months in a year (Azam et al. 2024), might describe the reason for the accumulation of undissolved lime on the top 4 cm of the soil. Long-term monitoring at the WH site suggests it may take many years for subsoil pH to increase following the surface application of lime without incorporation. In this study, we demonstrated that surface application of up to 8.5 t/ha limesand could not achieve/maintain target subsoil pH (20–30 cm) even after 26 years (Appendix Fig. 11).
This study shows that repeated surface applications of lime over the years resulted in an accumulation of undissolved lime in topsoils, mostly in 0–4 cm, however, the amount varied based on the rate of lime application, level of soil acidity, the time elapse between time application and sampling of soil. A similar finding was reported in South Australia (Hume et al. 2023) and WA (Azam and Gazey 2020). This study, for the first time, demonstrates that incorporation and incubation of topsoil containing undissolved lime with acidic subsoil significantly increased soil pH, as carbonates from undissolved lime neutralised subsoil acidity. The incorporation of control topsoil with acidic subsoil also raised soil pH and decreased soil EAC, likely due to the dilution of topsoil with higher pH and the addition of organic matter from topsoil to the acidic subsoil (Zhang et al. 2023; Azam and Gazey 2023). Soil EAC inversely and exponentially correlates with soil pH (Bojórquez-Quintal et al. 2017; Gillespie et al. 2021), as also seen in this study (r2 = 0.88–0.99) (Appendix Fig. 8). However, this study suggests that once lime is applied, the variability in soil pH and EAC might be minimised across the soil types as we saw a closer relationship between soil pH and EAC for all soils. However, the relationship between soil pH and EAC of the control soils varied across the study sites.
The NO soil contained the highest amount of carbonate content in the topsoil (Fig. 2g, h and i) due to the recent one-off surface addition of lime at a high rate (5 t/ha in 2012) compared to WH and ME (3t/ha in 2014) and hence a larger fraction of lime remained undissolved (Anderson et al. 2013). Therefore, the incorporation of only 5% of topsoil from NO with the acidic subsoil was sufficient to increase soil pH above the target (Fig. 3b). This is equivalent to incorporating 4 cm topsoil to 80 cm depth. Therefore, we concluded that the incorporation of previously limed topsoil with the acidic subsoil could be sufficient to increase the subsoil pH without any further addition of lime, depending on the concentration of undissolved lime in the topsoil. This is further evident from the field experiment at WH, where the incorporation of the topsoil from 8.5 t/ha lime rate (containing undissolved lime) with the acidic subsoil, the target pH was reached rapidly and maintained over the duration of the experiment (see Azam and Gazey 2020 and Fig. 6c). However, where a similar soil type is never been limed, it could be more effective to apply fresh lime and incorporate immediately as demonstrated in Azam and Gazey (2020).
Amelioration of subsoil acidity is important, particularly in facilitating plant root exploration (Fageria and Nascente 2014; Azam et al. 2023). The increase in subsoil pH due to incorporation and incubation of topsoil containing undissolved lime has significantly and exponentially decreased soil EAC below the toxic level and allowed plant roots to grow higher RLD and surface area, with more fine roots, compared to the control soil in this study. Overall root diameter also decreased when limed topsoil was incorporated with the acidic subsoil (Appendix 2). Similar observations were reported in previous studies (Delhaize and Ryan 1995; Gazey et al. 2019; Azam and Gazey 2020). Plant roots with similar lengths but smaller diameters provide more surface area for the uptake of water and nutrients compared to root systems with larger diameters (El Amrani 2023). Amelioration of subsoil acidity through the incorporation of topsoil containing undissolved lime increased barley and wheat RLD and surface area compared to the incorporation of topsoil without undissolved lime in all soils in this study (Figs. 4 and 5).
The improvement in root architecture may be crucial in determining how crops adapt to environmental changes and optimise growth, yield and WUE (El Amrani 2023). An increase in the root length and exploration of soil is directly related to the plant biomass and yield (Comas et al. 2013; Jung et al. 2019; Zhou et al. 2022). In general, in the constrained soils of southwestern WA, crop root growth is often restricted to the top 20–25 cm of soil, which reduces the volume of soil explored by crop roots for water and nutrients, significantly restricting yield (Azam and Gazey 2020). In both incubation and field experiments, the incorporation of topsoil containing undissolved lime with the acidic subsoil increased the pH to the target level and decreased the EAC well below the toxic range to facilitate the crop growth and yield, which is in line with previous findings (Azam and Gazey 2020; Azam et al. 2022). However, the yield difference between treatments with and without incorporation of undissolved lime was significant for canola but not for wheat and barley in this study. This could be due to a shallow depth of incorporation (only 25 cm) and sensitivity of the crop species used in this study, as reported in Azam and Gazey (2023). Nevertheless, the improvements in soil pH and EAC were significant, and this may generate longer-term yield benefits. However, if soil pH could be ameliorated to deeper than 25 cm, potentially requiring further addition of lime, we would expect the yield benefit to be greater (Azam et al. 2022).
As Australia is one of the highest adopters of minimum tillage-based cropping systems (Fischer and Hobbs 2019; Kassam et al. 2019; Llewellyn and Ouzman 2019), There is concern that re-introduction of the tillage practices may destroy the benefits achieved so far (Blanco-Canqui and Wortmann 2020) which is argued by several studies showing that strategic tillage has a low impact on the benefits of long-term conservation agriculture practices (e.g. Baan et al. 2009; Blanco-Canqui and Wortmann 2020). It is reported that the adoption of no-till-based practices is one of the main factors contributing to increased subsoil acidity (Page et al. 2020) and the surface application of lime takes a long time to ameliorate subsoil acidity (Azam and Gazey 2020). In addition, the benefit of using strategic deep tillage for lime incorporation can last for several years (Conyers et al. 2003; Davies et al. 2020; Azam et al. 2022), hence not requiring further intervention for a long period. Therefore, we recommend the incorporation of undissolved lime (paddock with good liming history) in the topsoil using strategic deep tillage for quick amelioration of subsoil acidity. It should be noted that strategic deep tillage can incur a significant cost, but the return on this investment is profitable (Davies et al. 2020). It is also recommended to conduct strategic deep tillage when soil conditions are ideal (e.g. moist but not too wet), and no strong wind is forecast as well as establish a crop as soon as possible to cover the tilled soil optimise the benefits and restore the benefits of no-till cropping (Nunes et al. 2015; Davies et al. 2020). Literature suggests that increasing the incorporation depth might result in higher crop yield than we observed in this study (Azam et al. 2022).
Conclusions
In the three field experiments included in this study, the application of lime in previous years via surface application has proven adequate to elevate topsoil pH above the target level. However, a considerable portion of lime remains unused and resides in the surface top few centimetres of the soil. Deep incorporation of this undissolved topsoil lime into the subsoil with strategic deep tillage has the potential to substantially increase subsoil pH, decrease EAC promptly, and enhance plant growth. Further lime application without implementing deep tillage practices is unlikely to bring significant benefits in terms of increasing subsoil pH and economic returns in the short-term.
The utilisation of strategic deep tillage to incorporate undissolved lime from the topsoil into the subsoil may accelerate the amelioration of acidic subsoil, contingent upon the concentration of undissolved lime in the topsoil. If undissolved lime is abundant in the topsoil, its incorporation into the subsoil could increase the efficiency of previously applied lime and improve the economic benefit. However, strategic deep tillage will incur a cost, but the return on this investment is likely to be beneficial.
Data availability
The datasets used and/or analysed during the current study available from the corresponding author on reasonable request.
References
Anderson NP, Hart JM, Sullivan DM, Horneck DA, Pirelli GJ, Christensen NW (2013) Applying lime to raise soil pH for crop production (western Oregon). EM9057. Oregon State University Extension Service. https://extension.oregonstate.edu/sites/default/files/documents/em9057.pdf. Accessed 10 Oct 2023
Azam G, Gazey C (2020) Slow movement of alkali from surface-applied lime warrants the introduction of strategic tillage for rapid amelioration of subsurface acidity in south-western Australia. Soil Res 59:97–106. https://doi.org/10.1071/SR19329
Azam G, Gazey C (2023) Shallow incorporation of lime and gypsum has limited benefit over the sole-surface application of lime for improving grain yield and water use efficiency in the low rainfall region of Western Australia. Sustain Agric Res 12(2):1–57. https://doi.org/10.5539/sar.v12n2p57
Azam G, Gazey C, Davies S, Scanlan C (2022) Mind the depth of soil amelioration to surpass the yield gap in Western Australia. In: Proceedings of the 20th Agronomy Australia Conference; 18–22 September 2022; Toowoomba, Qld, Australia. Available via: http://agronomyaustraliaproceedings.org/images/sampledata/2022/Soil/ASAazam_g_422s.pdf. Accessed 10 Oct 2023
Azam G, Rahman MS, Wickramarachchi K (2023) Strategic tillage for sustaining the productivity of broadacre cropping in the arid and semi-arid regions of southern Australia. In: De Sousa R (ed) Strategic Till and Soil Manag- New Perspectives. Intechopen. https://doi.org/10.5772/intechopen.112875
Azam G, Rahman MS, Scanlan C, Rahman MH, Gazey R, Gazey C (2024) Soil profile re-engineering doubled the yield and water use efficiency of crops over six years on acidic and compacted sand in a water-limited environment. Sci Rep (under review). https://assets-eu.researchsquare.com/files/rs-4710141/v1_covered_2622d1e2-e3ce-4d34-a2f9-4816834bd167.pdf
Baan CD, Grevers MCJ, Schoenau JJ (2009) Effects of a single cycle of tillage on long-term no-till prairie soils. Can J Soil Sci 89:521–530. https://doi.org/10.4141/cjss08041
Blanco-Canqui H, Wortmann CS (2020) Does occasional tillage undo the ecosystem services gained with no-till? A review. Soil Till Res 198:104534. https://doi.org/10.1016/j.still.2019.104534
Bojórquez-Quintal E, Escalante-Magaña C, Echevarría-Machado I, Martínez-Estévez M (2017) Aluminum, a friend or foe of higher plants in acid soils. Front Plant Sci 8:1767. https://doi.org/10.3389/fpls.2017.01767
Bromfield SM (1987) Simple tests for the assessment of aluminium and manganese levels in acid soils. Aust J Exp Agric 27:399–404. https://doi.org/10.1071/EA9870399
Comas LH, Becker SR, Cruz VMV, Byrne PF, Dierig DA (2013) Root traits contributing to plant productivity under drought. Front Plant Sci 4:442. https://doi.org/10.3389/fpls.2013.00442
Conyers MK, Scott BJ (1989) The influence of surface incorporated lime on subsurface soil acidity. Aust J Experi Agric 29(2):201–207. https://doi.org/10.1071/EA9890201
Conyers MK, Heenan DP, McGhie WJ, Poile GP (2003) Amelioration of acidity with time by limestone under contrasting tillage. Soil Till Res 72(1):85–94. https://doi.org/10.1016/S0167-1987(03)00064-3
Davies S, Armstrong R, Macdonald LM, Condon J, Peterson (2020) Soil constraints: a role for strategic deep tillage. In: Pratley J, Kirkegaard J (eds) Australian agriculture in 2020: from conservation to automation. Agronomy Australia and Charles Stuart University, pp 117–135
Delhaize E, Ryan PR (1995) Aluminum toxicity and tolerance in plants. Plant Phys 107:315–321
Dodgshon RA (1978) Land improvement in Scottish farming: marl and lime in Roxburghshire and Berwickshire in the eighteenth century. Agricultural History Rev 26(1):1–14
El Amrani B (2023) Exploring the importance of root architecture plasticity in plant adaptation to environmental constraints. Plant Speci Bio 38(5):234–244. https://doi.org/10.1111/1442-1984.12410
Fageria NK, Nascente AS (2014) Management of soil acidity of south American soils for sustainable crop production. Advans Agron 128:221–275. https://doi.org/10.1016/B978-0-12-802139-2.00006-8
Fischer RA, Hobbs P (2019) Tillage: global update and prospects. In: Pratley J, Kirkegaard J (eds) Australian agriculture in 2020: from conservation to automation. Graham Centre for Agricultural Innovation, vol 2678. Charles Sturt University, Wagga Wagga, pp 3–19
French RJ, Schultz JE (1984) Water use efficiency of wheat in a Mediterranean-type environment. I. The relation between yield, water use and climate. Aust J Agric Res 35(6):743–764. https://doi.org/10.1071/AR9840743
Gazey C (2018) Effects of soil acidity. Department of Agriculture and Food, Western Australia. Available via https://www.agric.wa.gov.au/soil-acidity/effects-soil-acidity?page=0%2C0. Accessed 10 Oct 2023
Gazey C, Davies S (2009) Soil acidity: a guide for WA farmers and consultants. Department of Agriculture and Food, Western Australia. Bulletin 4784
Gazey C, Andrew J, Griffin E (2013) Soil acidity. In: Report card on sustainable natural resource use in agriculture. Department of Agriculture and Food, Western Australia
Gazey C, Davies S, Master R (2014) Soil acidity: a guide for WA farmers and consultants, 2nd edn. Department of Primary Industries and Regional Development, Western Australia, Perth. Bulletin 4858
Gazey C, Azam G, Clausen J, Rengel Z (2019) Soil quality eBook 4: soil acidity. SoilsWest, Perth
Gillespie CJ, Antonangelo JA, Zhang H (2021) The response of soil pH and exchangeable Al to alum and lime amendments. Agric 11(6):547. https://doi.org/10.3390/agriculture11060547
Holland JE, White PJ, Glendining MJ, Goulding KWT, McGrath SP (2019) Yield responses of arable crops to liming-an evaluation of relationships between yields and soil pH from a long-term liming experiment. Euro J Agron 105:176–188. https://doi.org/10.1016/j.eja.2019.02.016
Horváth B, Opara-Nadi O, Beese F (2005) A simple method for measuring the carbonate content of soils. Soil Sci Soci Amer J 69(4):1066–1068. https://doi.org/10.2136/sssaj2004.0010
Hume R, Marschner P, Mason S, Schilling RK, Hughes B, Mosley LM (2023) Measurement of lime movement and dissolution in acidic soils using mid-infrared spectroscopy. Soil Till Res 233:105807. https://doi.org/10.1016/j.still.2023.105807
IUSS. World Reference Base for Soil Resources (2015) Report no. world soil resources reports no. 106, (Working Group WRB, FAO, Rome, Italy). Accessed 10 Oct 2023
Jung Y, Park K, Jensen KH, Kim W, Kim HY (2019) A design principle of root length distribution of plants. J Royal Soci Inter 16(161):20190556. https://doi.org/10.1098/rsif.2019.0556
Kassam A, Friedrich T, Derpsch R (2019) Global spread of conservation agriculture. Int J Environ Stud 76:29–51. https://doi.org/10.1080/00207233.2018.1494927
Kisinyo PO, Othieno CO, Gudu SO, Okalebo JR, Opala PA, Ng’Etich WK, Nyambati RO, Ouma EO, Agalo JJ, Kebeney SJ, Too EJ (2014) Immediate and residual effects of lime and phosphorus fertilizer on soil acidity and maize production in western Kenya. Experi Agric 50(1):128–143. https://doi.org/10.1017/S0014479713000318
Kuma M, Yilma G, Redi M (2022) Residual effect of lime rate after five years and p fertilizer rates on bread wheat (Triticum Aestivum L.) yield on acidic soil in Banja district, Northwestern Ethiopia. Global J Agric Res 10(3):13–24
Llewellyn R, Ouzman J (2019) Conservation agriculture in Australia: 30 years on. In: Eds PJ, KirkegaardJ (eds) Australian agriculture in 2020: from conservation to automation: Agronomy Australia and Charles Sturt University, Wagga Wagga, pp 21–31
Lollato RP, Ochsner TE, Arnall DB, Griffin TW, Edwards JT (2019) From field experiments to regional forecasts: upscaling wheat grain and forage yield response to acidic soils. Agron J 111(1):287–302. https://doi.org/10.2134/agronj2018.03.0206
McArthur WM (2004) Reference soils of south-western Australia. Department of Primary Industries and Regional Development, Western Australia, Perth. Book. https://library.dpird.wa.gov.au/books/4. Accessed 10 Oct 2023
National Land and Water Resources Audit (2001) National Land and Water Resources Audit, Catchment, River and Estuary Condition in Australia, Canberra. Government Report. http://audit.ea.gov.au/ANRA/agriculture/docs/national/Agriculture_Contents.html. Accessed 10 Oct 2023
Nunes MR, Denardin JE, Pauletto EA, Faganello A, Pinto LF (2015) Effect of soil chiseling on soil structure and root growth for a clayey soil under no-tillage. Geoderma 259:149–155. https://doi.org/10.1016/j.geoderma.2015.06.003
Page KL, Dang YP, Dalal RC (2020) The ability of conservation agriculture to conserve soil organic carbon and the subsequent impact on soil physical, chemical, and biological properties and yield. Fronts Sustain Food Sys 4:31. https://doi.org/10.3389/fsufs.2020.00031
Peel MC, Finlayson BL, McMahon TA (2007) Updated world map of the Köppen-Geiger climate classification. Hydrol Earth Syst Sci 11:1633–1644. https://doi.org/10.5194/hess-11-1633-2007
Petersen E (2016) Economic analysis of the impact and management of subsoil constraints. Grain Industry Association of Western Australia. 2019 Perth Crop Updates. https://grdc.com.au/resources-and-publications/grdc-update-papers/tab-content/grdc-update-papers/2016/02/economic-analysis-of-the-impacts-and-management-of-subsoil-constraints. Accessed 10 Oct 2023
Rahman MS, Schefe C, Rajput S, Keizer D, Weatherley A (2021) O-aryl and carbonyl carbon contents of food waste and biosolid predict P availability in an acidic soil. Fronts Sustain Food Sys 4:609788. https://doi.org/10.3389/fsufs.2020.609788
Rahman MS, Schefe C, Weatherley A (2023) The combined addition of citric and aromatic organic acids to an acid soil prolongs phosphorus availability. Soil Sci Soci Ameri J 87(4):797–807. https://doi.org/10.1002/saj2.20532
Rayment GE, Lyons DJ (2011) Soil chemical methods-Australasia. CSIRO Publishing, Melbourne
Rheinheimer DS, Tiecher T, Gonzatto R, Zafar M, Brunetto G (2018) Residual effect of surface-applied lime on soil acidity properties in a long-term experiment under no-till in a southern Brazilian sandy Ultisol. Geoderma 313:7–16. https://doi.org/10.1016/j.geoderma.2017.10.024
Scanlan C, Brennan R, Sarre G (2014) Shifting investment from nutrients to lime and cultivation on acid soils: is an immediate payback possible? In: The Proceedings of Agribusiness Crop Updates (ABCU’ 14); 24–25 February 2014; Perth, WA, Australia: ABCU; 2014. pp 1–6
Sumner ME, Shahandeh H, Bouton J, Hammel J (1986) Amelioration of an acid soil profile through deep liming and surface application of gypsum. Soil Sci Sociof Ameri J 50:1254–1258. https://doi.org/10.2136/sssaj1986.03615995005000050034x
Tang C, Rengel Z, Diatloff E, Gazey C (2003) Responses of wheat and barley to liming on a sandy soil with subsoil acidity. Field Crops Resear 80:235–244. https://doi.org/10.1016/S0378-4290(02)00192-2
Umbers A (2017) Farm practices survey report 2016 [Internet]. 2017. Available via https://grdc.com.au/resources-and-publications/all-publications/publications/2018/farm-practices-survey-report-2016. Accessed 10 Oct 2023
Zhang S, Zhu Q, De Vries W, Ros GH, Chen X, Muneer MA, Zhang F, Wu L (2023) Effects of soil amendments on soil acidity and crop yields in acidic soils: a world-wide meta-analysis. J Environ Manag 345:118531. https://doi.org/10.1016/j.jenvman.2023.118531
Zhou J, Zhang Z, Xin Y, Chen G, Wu Q, Liang X, Zhai Y (2022) Effects of planting density on root spatial and temporal distribution and yield of winter wheat. Agron 12(12):3014. https://doi.org/10.3390/agronomy12123014
Acknowledgements
The authors would like to thank the Department of Primary Industries and Regional Development (DPIRD) and the Grain Research and Development Corporation for supporting this research under the DAW00252 project. We would also like to thank supporting staff Kanch Wickramarachchi, Daron Malinowski, Richard Bowles and others at the Wongan Hills field research facility of DPIRD. The authors are grateful to Dr Craig Scanlan for allowing soil sample collection from the Northam trial. We are thankful to Dr Mario D’Antuono and Andrew van Burgel for their advice on trial design. The authors also express their gratitude to Tom Edwards, Dr Stephen Davies and Chris Gazey (who initially established the Wongan Hills experiment) for reviewing the manuscript internally. We are thankful to the two anonymous reviewers for their thoughtful comments towards the improvement of this manuscript.
Funding
Open Access funding enabled and organized by CAUL and its Member Institutions.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflicts of interest.
Additional information
Responsible Editor: Miroslav Nikolic.
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Appendices
Appendix 1
Fig. 8
Appendix 2
Fig. 9
The effect of incorporation of the limed and control topsoil with the acidic subsoil collected from Wongan Hills (a and d), Northam (b and e) and Merredin (c and f) on the average root diameter of barley (a, b, and c) and wheat (d, e and f). A threshold of 2.0 mm was applied to separate roots from any other plant materials. The vertical lines indicate the standard error of the mean values. The LSD at 5% for a = 0.04, b = 0.04, c = 0.06, d = 0.07, e = 0.07 and f = 0.08
Appendix 3
Fig. 10
The effect of incorporation of the limed and control topsoil with the acidic subsoil on the fine root length density of barley (a, b, and c) and wheat (d, e and f) in soils from Wongan Hills (a and d), Northam (b and e) and Merredin (c and f). A threshold of 2.0 mm was applied to separate roots from any other plant materials. All roots with a diameter of ≤ 0.5 were considered as fine roots. The vertical lines indicate the standard error of the mean values. The LSD at 5% for a = 0.35, b = 0.26, c = 0.25, d = 0.42, e = 0.33 and f = 0.37
Appendix 4
Fig. 11
Periodic measurement of topsoil (a) (0–10 cm) and subsoil (b and c) (10–30 cm) pHat the Wongan Hills. Lime rates L0, L2, L4, L2.1, L4.1, L2.2 and L4.2 indicate the total lime addition at the rate of 0, 2, 4, 3.5, 5.5, 6.5 and 8.5 t/ha from 1994 to 2014 at Wongan Hills. The vertical lines on the top of the bar indicate the standard error of the mean values. T25 indicates incorporation up to 25 cm
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Azam, G., Rahman, M.S. Incorporation of undissolved lime from previous applications can ameliorate subsoil acidity promptly and improve crop performance on sandy soils of the semi-arid regions of Western Australia. Plant Soil (2024). https://doi.org/10.1007/s11104-024-06957-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11104-024-06957-4