Abstract
Background and aims
Unravelling how fundamental axes of leaf and fine-root trait variation correlate and relate to nutrient availability is crucial for understanding plant distribution across edaphic gradients. While leaf traits vary consistently along soil nutrient availability gradients, the response of fine-root traits to the same gradients has yielded inconsistent results.
Methods
We studied leaf and root trait variation among 23 co-occurring plant species along a 2-million-year soil chronosequence to assess how leaf and root traits coordinate and whether this axis of trait variation relates to soil fertility.
Results
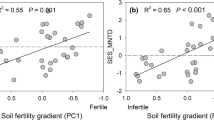
Leaf and root trait variation was primarily structured by mycorrhizal association types. However, when considering community-level traits weighted by plant species abundance, soil nutrient availability was an important driver of trait distribution. Leaves that support rapid growth on younger more fertile soils were associated with roots of larger diameter and arbuscular mycorrhizal colonization. In contrast, leaves that favor nutrient conservation on nutrient-impoverished soil were associated with greater root-hair length and phosphorus-mobilizing root exudates proxied by leaf manganese concentration. At the species level, leaf and root trait variation patterns deviated from the community-wide results, as leaves that support either rapid growth or survival were associated with a wide range of root trait syndromes.
Conclusions
Our results highlight the challenge of generalizing a specific set of root-trait values that consistently meet the requirements of leaves supporting rapid growth or survival. Hence, the leaf economic spectrum's ability to capture variation in carbon gain may not be reflected by the root economic space.




Similar content being viewed by others
Data availability
The data that support the findings of this study are openly available in ‘figshare’ at https://doi.org/10.6084/m9.figshare.6025748, reference no. https://doi.org/10.6084/m9.figshare.25838536.
Abbreviations
- AM:
-
Arbuscular mycorrhizal
- BrInt:
-
Branching intensity
- C:
-
Carbon
- D:
-
Diameter
- ECM:
-
Ectomycorrhizal
- ERM:
-
Ericoid mycorrhizal
- LTD:
-
Leaf tissue density
- Mn:
-
Manganese
- N:
-
Nitrogen
- NM:
-
Non-mycorrhizal
- P:
-
Phosphorus
- RHD:
-
Root hair density
- RHL:
-
Root hair length
- RTD:
-
Root tissue density
- SLA:
-
Specific leaf area
- SRL:
-
Specific root length
- Thick:
-
Thickness
References
Abrahão A, Ryan MH, Laliberté E, Oliveira RS, Lambers H (2018) Phosphorus- and nitrogen-acquisition strategies in two Bossiaea species (Fabaceae) along retrogressive soil chronosequences in south-western Australia. Physiol Plant 163:323–343. https://doi.org/10.1111/ppl.12704
Bergmann J, Weigelt A, van der Plas F, Laughlin DC, Kuyper TW, Guerrero-Ramirez N, Valverde-Barrantes OJ, Bruelheide H, Freschet GT, Iversen CM et al (2020) The fungal collaboration gradient dominates the root economics space in plants. Sci Adv 6(27):eaba3756. https://doi.org/10.1126/sciadv.aba3756
Bernard-Verdier M, Navas M-L, Vellend M, Violle C, Fayolle A, Garnier E (2012) Community assembly along a soil depth gradient: Contrasting patterns of plant trait convergence and divergence in a Mediterranean rangeland. J Ecol 100:1422–1433. https://doi.org/10.1111/1365-2745.12003
Brundrett MC (2009) Mycorrhizal associations and other means of nutrition of vascular plants: Understanding the global diversity of host plants by resolving conflicting information and developing reliable means of diagnosis. Plant Soil 320:37–77. https://doi.org/10.1007/s11104-008-9877-9
Canarini A, Kaiser C, Merchant A, Richter A, Wanek W (2019) Root exudation of primary metabolites: Mechanisms and their roles in plant responses to environmental stimuli. Front Plant Sci 10:422679. https://doi.org/10.3389/fpls.2019.00157
Chapin FS (1980) The mineral nutrition of wild plants. Annu Rev Ecol Syst 11:233–260. https://doi.org/10.1146/annurev.es.11.110180.001313
Claassen VP, Zasoski RJ (1992) A containerized staining system for mycorrhizal roots. New Phytol 121:49–51. https://doi.org/10.1111/j.1469-8137.1992.tb01091.x
Comas LH, Callahan HS, Midford PE (2014) Patterns in root traits of woody species hosting arbuscular and ectomycorrhizas: Implications for the evolution of belowground strategies. Ecol Evol 4:2979–2990. https://doi.org/10.1002/ece3.1147
Craine JM (2009) Resource strategies of wild plants. Princeton University Press, Princeton
Dallstream C, Weemstra M, Soper FM (2022) A framework for fine-root trait syndromes: Syndrome coexistence may support phosphorus partitioning in tropical forests. Oikos 1:e08908. https://doi.org/10.1111/oik.08908
Dray S, Dufour A-B (2007) The ade4 package: Implementing the duality diagram for ecologists. J Stat Softw 22:1–20. https://doi.org/10.18637/jss.v022.i04
Dray S, Chessel D, Thioulouse J (2003) Co-inertia analysis and the linking of ecological data tables. Ecology 84:3078–3089. https://doi.org/10.1890/03-0178
Eissenstat DM, Graham JH, Syvertsen JP, Drouillard DL (1993) Carbon economy of sour orange in relation to mycorrhizal colonization and phosphorus status. Ann Bot 71:1–10. https://doi.org/10.1006/anbo.1993.1001
Eissenstat DM, Kucharski JM, Zadworny M, Adams TS, Koide RT (2015) Linking root traits to nutrient foraging in arbuscular mycorrhizal trees in a temperate forest. New Phytol 208:114–124. https://doi.org/10.1111/nph.13451
Ericsson T, Rytter L, Vapaavuori E (1996) Physiology of carbon allocation in trees. Biomass Bioenergy 11:115–127. https://doi.org/10.1016/0961-9534(96)00032-3
Fitter AH (1991) Costs and benefits of mycorrhizas: implications for functioning under natural conditions. Experientia 47:350–355. https://doi.org/10.1007/BF01972076
Freschet GT, Valverde-Barrantes OJ, Tucker CM, Craine JM, McCormack ML, Violle C, Fort F, Blackwood CB, Urban-Mead KR, Iversen CM et al (2017) Climate, soil and plant functional types as drivers of global fine-root trait variation. J Ecol 105:1182–1196. https://doi.org/10.1111/1365-2745.12769
Freschet GT, Roumet C, Comas LH, Weemstra M, Bengough AG, Rewald B, Bardgett RD, De Deyn GB, Johnson D, Klimešová J et al (2021) Root traits as drivers of plant and ecosystem functioning: Current understanding, pitfalls and future research needs. New Phytol 232:1123–1158. https://doi.org/10.1111/nph.17072
Funayama-Noguchi S, Shibata M, Noguchi K, Terashima I (2021) Effects of root morphology, respiration and carboxylate exudation on carbon economy in two non-mycorrhizal lupines under phosphorus deficiency. Plant Cell Environ 44:598–612. https://doi.org/10.1111/pce.13925
Guilbeault-Mayers X, Turner BL, Laliberté E (2020) Greater root phosphatase activity of tropical trees at low phosphorus despite strong variation among species. Ecology 101:e03090. https://doi.org/10.1002/ecy.3090
Guilbeault-Mayers X, Laliberté E (2024) Root phosphatase activity is coordinated with the root conservation gradient across a phosphorus gradient in a lowland tropical forest. New Phytol NA:NA-NA. https://doi.org/10.1111/nph.19567
Guyonnet JP, Cantarel AAM, Simon L, Haichar FEZ (2018) Root exudation rate as functional trait involved in plant nutrient-use strategy classification. Ecol Evol 8:8573–8581. https://doi.org/10.1002/ece3.4383
Hothorn T, Bretz F, Westfall P (2008) Simultaneous inference in general parametric models. Biom J 50:346–363. https://doi.org/10.1002/bimj.200810425
Huang G, Hayes PE, Ryan MH, Pang J, Lambers H (2017) Peppermint trees shift their phosphorus-acquisition strategy along a strong gradient of plant-available phosphorus by increasing their transpiration at very low phosphorus availability. Oecologia 185:387–400. https://doi.org/10.1007/s00442-017-3961-x
Jager MM, Richardson SJ, Bellingham PJ, Clearwater MJ, Laughlin DC (2015) Soil fertility induces coordinated responses of multiple independent functional traits. J Ecol 103:374–385. https://doi.org/10.1111/1365-2745.12366
Jakobsen I, Rosendahl L (1990) Carbon flow into soil and external hyphae from roots of mycorrhizal cucumber plants. New Phytol 115:77–83. https://doi.org/10.1111/j.1469-8137.1990.tb00924.x
Jakobsen I, Abbott LK, Robson AD (1992) External hyphae of vesicular-arbuscular mycorrhizal fungi associated with Trifolium subterraneum L. New Phytol 120:371–380. https://doi.org/10.1111/j.1469-8137.1992.tb01077.x
Kattge J, Bönisch G, Diaz S, Lavorel S, Prentice IC, Leadley P, Tautenhahn S, Werner G, Günther A (2020) TRY plant trait database-enhanced coverage and open access. Glob Chang Biol 26:119–188. https://doi.org/10.1111/gcb.14904
Kendrick GW, Wyrwoll KH, Szabo BJ (1991) Pliocene-Pleistocene coastal events and history along the western margin of Australia. Quat Sci Rev 10:419–439. https://doi.org/10.1016/0277-3791(91)90005-F
Kramer-Walter KR, Bellingham PJ, Millar TR, Smissen RD, Richardson SJ, Laughlin DC (2016) Root traits are multidimensional: specific root length is independent from root tissue density and the plant economic spectrum. J Ecol 104:1299–1310. https://doi.org/10.1111/1365-2745.12562
Laliberté E (2017) Below-ground frontiers in trait-based plant ecology. New Phytol 213:1597–1603. https://doi.org/10.1111/nph.14247
Laliberté E, Lambers H, Burgess TI, Wright SJ (2015) Phosphorus limitation, soil-borne pathogens and the coexistence of plant species in hyperdiverse forests and shrublands. New Phytol 206:507–521. https://doi.org/10.1111/nph.13203
Lambers H (2022) Phosphorus acquisition and utilization in plants. Annu Rev Plant Biol 73:17–42. https://doi.org/10.1146/annurev-arplant-102720-125738
Lambers H, Shane MW, Cramer MD, Pearse SJ, Veneklaas EJ (2006) Root structure and functioning for efficient acquisition of phosphorus: Matching morphological and physiological traits. Ann Bot 98:693–713. https://doi.org/10.1093/aob/mcl114
Lambers H, Hayes PE, Laliberté E, Oliveira RS, Turner BL (2015a) Leaf manganese accumulation and phosphorus-acquisition efficiency. Trends Plant Sci 20:83–90. https://doi.org/10.1016/j.tplants.2014.10.007
Lambers H, Martinoia E, Renton M (2015b) Plant adaptations to severely phosphorus-impoverished soils. Curr Opin Plant Biol 25:23–31. https://doi.org/10.1016/j.pbi.2015.04.002
Lambers H, Albornoz F, Kotula L, Laliberté E, Ranathunge K, Teste FP, Zemunik G (2018) How belowground interactions contribute to the coexistence of mycorrhizal and non-mycorrhizal species in severely phosphorus-impoverished hyperdiverse ecosystems. Plant Soil 424:11–33. https://doi.org/10.1007/s11104-017-3427-2
Lambers H, de Britto CP, Cawthray GR, Denton MD, Finnegan PM, Hayes PE, Oliveira RS, Power SC, Ranathunge K, Shen Q, Wang X, Zhong H (2022) Strategies to acquire and use phosphorus in phosphorus-impoverished and fire-prone environments. Plant Soil 476:133–161. https://doi.org/10.1007/s11104-022-05464-8
Lambers H, Poorter H (1992) Inherent variation in growth rate between higher plants: A search for physiological causes and ecological consequences. In Begon M, Fitter AH (Eds.) Advances in Ecological Research. Elsevier Academic Press, Amsterdam, 23:187–261. https://doi.org/10.1016/S0065-2504(08)60148-8
Lenth R (2020) Emmeans: Estimated Marginal Means, aka Least-Squares Means. R package version 1.5.1. https://CRAN.R-project.org/package=emmeans
Liu G, Freschet GT, Pan X, Cornelissen JHC, Li Y, Dong M (2010) Coordinated variation in leaf and root traits across multiple spatial scales in Chinese semi-arid and arid ecosystems. New Phytol 188:543–553. https://doi.org/10.1111/j.1469-8137.2010.03388.x
Lv C, Wang C, Li Y, Zhou Z (2023) Coordination among root exudation C, mycorrhizal colonization, and functional traits and their responses to drought in five temperate tree species. For Ecol Manag 546:121316. https://doi.org/10.1016/j.foreco.2023.121316
Lynch JP, Ho MD (2005) Rhizoeconomics: Carbon costs of phosphorus acquisition. Plant Soil 269:45–56. https://doi.org/10.1007/s11104-004-1096-4
McKane RB, Johnson LC, Shaver GR, Nadelhoffer KJ, Rastetter EB, Fry B, Giblin AE, Kielland K, Kwiatkowski BL, Laundre JA et al (2002) Resource-based niches provide a basis for plant species diversity and dominance in arctic tundra. Nature 415:68–71. https://doi.org/10.1038/415068a
Meier IC, Tuckmantel T, Heitkotter J, Muller K, Preusser S, Wrobel TJ, Kandeler E, Marschner B, Leuschner C (2020) Root exudation of mature beech forests across a nutrient availability gradient: the role of root morphology and fungal activity. New Phytol 226:583–594. https://doi.org/10.1111/nph.16389
Oksanen J, Blanchet FG, Friendly M, Kindt R, Legendre P, McGlinn D, Minchin PR, O’Hara RB, Simpson GL, Solymos P et al (2020) vegan: Community Ecology Package (Version 2.5–7). https://CRAN.R-project.org/package=vegan
Ordoñez JC, Van Bodegom PM, Witte J-PM, Wright IJ, Reich PB, Aerts R (2009) A global study of relationships between leaf traits, climate and soil measures of nutrient fertility. Glob Ecol Biogeogr 18:137–149. https://doi.org/10.1111/j.1466-8238.2008.00441.x
Peres-Neto PR, Jackson DA, Somers KM (2003) Giving meaningful interpretation to ordination axes: Assessing loading significance in principal component analysis. Ecology 84:2347–2363. https://doi.org/10.1890/00-0634
Pinheiro J, Bates D, Sarkar D, R Core Team (2020) Nlme: Linear and nonlinear mixed effects models. R package version 3.1–149. https://CRAN.R-project.org/package=nlme
Playford PE, Cockbain AE, Lowe GH (1976) Geology of the Perth Basin, Western Australia; Bulletin 124 of the Geological Survey of Western Australia. Geological Survey of Western Australia, Perth, Australia
R Core Team (2020) A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. https://www.R-project.org/
Raven JA, Lambers H, Smith SE, Westoby M (2018) Costs of acquiring phosphorus by vascular land plants: Patterns and implications for plant coexistence. New Phytol 217(4):1420–1427. https://doi.org/10.1111/nph.14967
Read DJ, Leake JR, Perez-Moreno J (2004) Mycorrhizal fungi as drivers of ecosystem processes in heathland and boreal forest biomes. Canad J Bot 82:1243–1263. https://doi.org/10.1139/b04-123
Reich PB (2014) The world-wide ‘fast–slow’ plant economics spectrum: A traits manifesto. J Ecol 102:275–301. https://doi.org/10.1111/1365-2745.12211
Reich PB, Walters MB, Ellsworth DS (1992) Leaf life-span in relation to leaf, plant, and stand characteristics among diverse ecosystems. Ecol Monogr 62:365–392. https://doi.org/10.2307/2937116
Reich PB, Walters MB, Ellsworth DS (1997) From tropics to tundra: Global convergence in plant functioning. Proc Natl Acad Sci 94:13730–13734. https://doi.org/10.1073/pnas.94.25.13730
Sell M, Ostonen I, Rohula-Okunev G, Rusalepp L, Rezapour A, Kupper P (2022) Responses of fine root exudation, respiration and morphology in three early successional tree species to increased air humidity and different soil nitrogen sources. Tree Physiol 42:557–569. https://doi.org/10.1093/treephys/tpab118
Shane MW, Cramer MD, Funayama-Noguchi S, Cawthray GR, Millar AH, Day DA, Lambers H (2004) Developmental physiology of cluster-root carboxylate synthesis and exudation in Harsh Hakea. Expression of phosphoenolpyruvate carboxylase and the alternative oxidase. Plant Physiol 135:549–560. https://doi.org/10.1104/pp.103.035659
Smith JE, Read DJ (2008) Mycorrhizal symbiosis, 3rd edn. Elsevier Academic Press, Amsterdam
Sun L, Ataka M, Han M, Han Y, Gan D, Xu T, Guo Y, Zhu B (2021) Root exudation as a major competitive fine-root functional trait of 18 coexisting species in a subtropical forest. New Phytol 229:259–271. https://doi.org/10.1111/nph.16865
Tjoelker MG, Craine JM, Wedin D, Reich PB, Tilman D (2005) Linking leaf and root trait syndromes among 39 grassland and savannah species. New Phytol 167:493–508. https://doi.org/10.1111/j.1469-8137.2005.01428.x
Treseder KK, Allen MF (2002) Direct nitrogen and phosphorus limitation of arbuscular mycorrhizal fungi: a model and field test. New Phytol 155:507–515. https://doi.org/10.1046/j.1469-8137.2002.00470.x
Turner BL (2008) Resource partitioning for soil phosphorus: A hypothesis. J Ecol 96:698–702. https://doi.org/10.1111/j.1365-2745.2008.01384.x
Turner BL, Romero TE (2009) Short-term changes in extractable inorganic nutrients during storage of tropical rain forest soils. Soil Sci Soc Am J 73:1972–1979. https://doi.org/10.2136/sssaj2008.0407
Turner BL, Hayes PE, Laliberté E (2018) A climosequence of chronosequences in southwestern Australia. Eur J Soil Sci 69:69–85. https://doi.org/10.1101/113308
Valverde-Barrantes OJ, Freschet GT, Roumet C, Blackwood CB (2017) A worldview of root traits: The influence of ancestry, growth form, climate and mycorrhizal association on the functional trait variation of fine-root tissues in seed plants. New Phytol 215:1562–1573. https://doi.org/10.1111/nph.14571
Vierheilig H, Coughlan AP, Wyss U, Piché Y (1998) Ink and vinegar, a simple staining technique for arbuscular-mycorrhizal fungi. Appl Environ Microbiol 64:5004–5007. https://doi.org/10.1128/AEM.64.12.5004-5007.1998
Walker TW, Syers JK (1976) The fate of phosphorus during pedogenesis. Geoderma 15:1–19. https://doi.org/10.1016/0016-7061(76)90066-5
Walker LR, Wardle DA, Bardgett RD, Clarkson BD (2010) The use of chronosequences in studies of ecological succession and soil development. J Ecol 98:725–736. https://doi.org/10.1111/j.1365-2745.2010.01664.x
Wang Q, Xiao J, Ding J, Zou T, Zhang Z, Liu Q, Yin H (2021) Differences in root exudate inputs and rhizosphere effects on soil N transformation between deciduous and evergreen trees. Plant Soil 458:277–289. https://doi.org/10.1007/s11104-019-04156-0
Ward EB, Duguid MC, Kuebbing SE, Lendemer JC, Bradford MA (2022) The functional role of ericoid mycorrhizal plants and fungi on carbon and nitrogen dynamics in forests. New Phytol 235:1701–1718. https://doi.org/10.1111/nph.18307
Weemstra M, Mommer L, Visser EJW, van Ruijven J, Kuyper TW, Mohren GMJ, Sterck FJ (2016) Towards a multidimensional root trait framework: A tree root review. New Phytol 211:1159–1169. https://doi.org/10.1111/nph.14003
Weigelt A, Mommer L, Andraczek K, Iversen CM, Bergmann J, Bruelheide H, Fan Y, Freschet GT, Guerrero-Ramírez NR, Kattge J et al (2021) An integrated framework of plant form and function: The belowground perspective. New Phytol 232:42–59. https://doi.org/10.1111/nph.17590
Wen Z, White PJ, Shen J, Lambers H (2022) Linking root exudation to belowground economic traits for resource acquisition. New Phytol 233:1620–1635. https://doi.org/10.1111/nph.17854
Williams A, Langridge H, Straathof AL, Muhamadali H, Hollywood KA, Goodacre R, de Vries FT (2022) Root functional traits explain root exudation rate and composition across a range of grassland species. J Ecol 110:21–33. https://doi.org/10.1111/1365-2745.13630
Wright IJ, Reich PB, Westoby M, Ackerly DD, Baruch Z, Bongers F, Cavender-Bares J, Chapin T, Cornelissen JH, Diemer M et al (2004) The worldwide leaf economics spectrum. Nature 428:821–827. https://doi.org/10.1038/nature02403
Zuur AF, Ieno EN, Walker N, Saveliev AA, Smith GM (2009) Mixed effects models and extensions in ecology with R. Springer, New York
Acknowledgements
Special thanks to Sharyn and Shaun Cody; without their kindness and advice, the project would not have been successful. We extend our sincere appreciation to David Poissant and Caroline Fink-Mercier for their invaluable assistance during fieldwork. We also express our profound gratitude for the invaluable comments and suggestions provided by two anonymous reviewers and Editor Dr. M. Luke McCormack.
Funding
Funding for this research was provided by a Discovery Grant from the Natural Sciences and Engineering Research Council of Canada (NSERC; grant RGPIN-2014–06106 and RGPIN-2019–04537). XGM received additional support from the Fonds de recherche du Québec-Nature et technologies.
Author information
Authors and Affiliations
Contributions
XGM, EL and HL conceived the study; XGM and EL conceived the methodology; XGM conducted the data collection; XGM analyzed the data; XGM, HL and EL interpreted the results; XGM led the writing of the manuscript. All authors contributed to the draft versions.
Corresponding author
Ethics declarations
Competing interests
None that need to be declared.
Additional information
Responsible Editor: Michael Luke McCormack.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Guilbeault-Mayers, X., Lambers, H. & Laliberté, E. Coordination among leaf and fine-root traits along a strong natural soil fertility gradient. Plant Soil (2024). https://doi.org/10.1007/s11104-024-06740-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11104-024-06740-5