Abstract
Background and aims
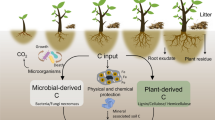
The amount and type of root exudates can influence P availability in the rhizosphere directly by desorption or dissolution of soil minerals, or indirectly by decomposition of soil organic matter (SOM). This study aimed to determine the mechanisms by which specific root exudates influence the distribution and availability of P in soils with low P availability.
Methods
Water, glucose, alanine, and oxalate were delivered through a simulated root into soils for 15 days. Zymography and planar optodes were used to image potential phosphatase activity, and O2 and pH distribution, respectively. Soils were analyzed for resin extractable inorganic P (Pi), dissolved organic C (DOC), water soluble Fe, and Al, and microbial community structure. Characterization of SOM and P were conducted using ultra-high resolution mass spectrometry and 31P solution nuclear magnetic resonance (NMR), respectively.
Results
The addition of oxalate resulted in the greatest resin extractable Pi, DOC, and water-soluble Fe, and Al compared to the other exudates suggesting destabilization of mineral associated organic matter (MAOM) and release of organic P (Po). Both 31P solution NMR and ultra-high resolution mass spectrometry analysis provided evidence of mineralization of Po released from the destabilization of MAOM.
Conclusion
The study demonstrates the important role microbial and plant-derived metal chelating ligands play in destabilizing MAOM, releasing SOM and importantly Po, that when mineralized may contribute to increasing Pi availability in soils with low P availability.








Similar content being viewed by others
Data availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
Abbreviations
- SOM:
-
Soil organic matter
- Pi :
-
Inorganic P
- DOC:
-
Dissolved organic C
- NMR:
-
Nuclear magnetic resonance
- Po :
-
Organic P
- MAOM:
-
Mineral associated organic matter
- LMW:
-
Low molecular weight
- CUE:
-
Carbon use efficiency
- IHP:
-
Myo-inositolhexakisphosphate
- RNA:
-
Ribonucleic acid
- DNA:
-
Deoxyribonucleic acid
- WEOM:
-
Water extractable organic matter
- PLFA:
-
Phospholipid fatty acid
- AM:
-
Arbuscular mycorrhizal
- G + :
-
Gram positive
- G-:
-
Gram negative
- MRPP:
-
Multi-response permutation procedure
- OM:
-
Organic matter
- FTICR MS:
-
Fourier-transform ion cyclotron resonance mass spectrometry
- PCA:
-
Principle component analysis
References
Achat DL, Bakker MR, Zeller B et al (2010) Long-term organic phosphorus mineralization in Spodosols under forests and its relation to carbon and nitrogen mineralization. Soil Biol Biochem 42:1479–1490. https://doi.org/10.1016/j.soilbio.2010.05.020
Allison VJ, Condron LM, Peltzer DA et al (2007) Changes in enzyme activities and soil microbial community composition along carbon and nutrient gradients at the Franz Josef chronosequence, New Zealand. Soil Biol Biochem 39:1770–1781. https://doi.org/10.1016/j.soilbio.2007.02.006
Apostel C, Dippold MA, Bore E, Kuzyakov Y (2017) Sorption of alanine changes microbial metabolism in addition to availability. Geoderma 292:128–134. https://doi.org/10.1016/j.geoderma.2017.01.016
Barekzai A, Mengel K (1993) Effect of microbial decomposition of mature leaves on soil pH. Zeitschrift Für Pflanzenernährung Und Bodenkunde 156:93–94. https://doi.org/10.1002/jpln.19931560115
Brant JB, Sulzman EW, Myrold DD (2006) Microbial community utilization of added carbon substrates in response to long-term carbon input manipulation. Soil Biol Biochem 38:2219–2232. https://doi.org/10.1016/j.soilbio.2006.01.022
Bulgarelli D, Schlaeppi K, Spaepen S et al (2013) Structure and functions of the bacterial microbiota of plants. Annu Rev Plant Biol 64:807–838
Burns RG (1982) Enzyme activity in soil: Location and a possible role in microbial ecology. Soil Biol Biochem 14:423–427. https://doi.org/10.1016/0038-0717(82)90099-2
Burns RG, DeForest JL, Marxsen J et al (2013) Soil enzymes in a changing environment: current knowledge and future directions. Soil Biol Biochem 58:216–234. https://doi.org/10.1016/j.soilbio.2012.11.009
Buyer JS, Sasser M (2012) High throughput phospholipid fatty acid analysis of soils. Appl Soil Ecol 61:127–130. https://doi.org/10.1016/j.apsoil.2012.06.005
Cade-Menun BJ (2015) Improved peak identification in 31P-NMR spectra of environmental samples with a standardized method and peak library. Geoderma 257–258:102–114. https://doi.org/10.1016/j.geoderma.2014.12.016
Cade-Menun BJ, Liu CW (2013) Solution phosphorus-31 nuclear magnetic resonance spectroscopy of soils from 2005 to 2013: a review of sample preparation and experimental parameters. Soil Sci Soc Am J 78:19–37. https://doi.org/10.2136/sssaj2013.05.0187dgs
Cade-Menun BJ, Preston CM (1996) A comparison of soil extraction procedures for 31P NMR spectroscopy. Soil Sci 161(11):770–785
Clarholm M, Skyllberg U, Rosling A (2015) Organic acid induced release of nutrients from metal-stabilized soil organic matter – the unbutton model. Soil Biol Biochem 84:168–176. https://doi.org/10.1016/j.soilbio.2015.02.019
Daly AB, Jilling A, Bowles TM, Buchkowski RW, Frey SD, Kallenbach DM, Keiluweit M, Mooshammer M, Schimel JP, Grandy AS (2021) A holistic framework integrating plant-microbe-mineral regulation of soil bioavailable nitrogen. Biogeochemistry 154(2):211–229. https://doi.org/10.1007/s10533-021-00793-9
Dijkstra FA, Bader NE, Johnson DW, Cheng W (2009) Does accelerated soil organic matter decomposition in the presence of plants increase plant N availability? Soil Biol Biochem 41:1080–1087. https://doi.org/10.1016/j.soilbio.2009.02.013
Dittmar T, Koch B, Hertkorn N, Kattner G (2008) A simple and efficient method for the solid-phase extraction of dissolved organic matter (SPE-DOM) from seawater. Limnol Oceanogr Methods 6:230–235. https://doi.org/10.4319/lom.2008.6.230
Fontaine S, Mariotti A, Abbadie L (2003) The priming effect of organic matter: a question of microbial competition? Soil Biol Biochem 35:837–843. https://doi.org/10.1016/S0038-0717(03)00123-8
Fox TR, Comerford NB (1992) Influence of oxalate loading on phosphorus and aluminum solubility in spodosols. Soil Sci Soc Am J 56:290–294. https://doi.org/10.2136/sssaj1992.03615995005600010046x
Fox TR, Comerford NB, McFee WW (1990) Kinetics of phosphorus release from spodosols: effects of oxalate and formate. Soil Sci Soc Am J 54:1441–1447. https://doi.org/10.2136/sssaj1990.03615995005400050038x
Frey SD, Lee J, Melillo JM, Six J (2013) The temperature response of soil microbial efficiency and its feedback to climate. Nat Clim Chang 3:395–398. https://doi.org/10.1038/nclimate1796
Geurts R, Franssen H (1996) Signal transduction in rhizobium-induced nodule formation. Plant Physiol 112:447 LP – 453. https://doi.org/10.1104/pp.112.2.447
Golubev SV, Bauer A, Pokrovsky OS (2006) Effect of pH and organic ligands on the kinetics of smetite dissolution at 25oC. Geochim Cosmochim Acta 70(17):4436–4451. https://doi.org/10.1016/j.gca.2006.06.1557
Graham JH, Leonard RT, Menge JA (1981) Membrane-mediated decrease in root exudation responsible for phorphorus inhibition of vesicular-arbuscular mycorrhiza formation. Plant Physiol 68:548–552
Grayston SJ, Vaughan D, Jones D (1997) Rhizosphere carbon flow in trees, in comparison with annual plants: the importance of root exudation and its impact on microbial activity and nutrient availability. Appl Soil Ecol 5:29–56. https://doi.org/10.1016/S0929-1393(96)00126-6
Guo J, McCulley RL, McNear DH Jr (2015) Tall fescue cultivar and fungal endophyte combinations influence plant growth and root exudate composition. Front Plant Sci 6:183. https://doi.org/10.3389/fpls.2015.00183
Hamer U, Marschner B (2005) Priming effects in different soil types induced by fructose, alanine, oxalic acid and catechol additions. Soil Biol Biochem 37:445–454. https://doi.org/10.1016/j.soilbio.2004.07.037
Hue NV, Amien I (1989) Aluminum detoxification with green manures. Commun Soil Sci Plant Anal 20:1499–1511. https://doi.org/10.1080/00103628909368164
Hütsch BW, Augustin J, Merbach W (2002) Plant rhizodeposition — an important source for carbon turnover in soils. J Plant Nutr Soil Sci 165:397–407. https://doi.org/10.1002/1522-2624(200208)165:4%3c397::AID-JPLN397%3e3.0.CO;2-C
Jagadamma S, Mayes MA, Phillips JR (2012) Selective sorption of dissolved organic carbon compounds by Temperate Soils. PLoS ONE 7:e50434
Jarosch KA, Kandeler E, Frossard E, Bünemann EK (2019) Is the enzymatic hydrolysis of soil organic phosphorus compounds limited by enzyme or substrate availability? Soil Biol Biochem 139:107628. https://doi.org/10.1016/j.soilbio.2019.107628
Jilling A, Keiluweit M, Contosta AR et al (2018) Minerals in the rhizosphere: overlooked mediators of soil nitrogen availability to plants and microbes. Biogeochemistry 139:103–122. https://doi.org/10.1007/s10533-018-0459-5
Jilling A, Keiluweit M, Gutknecht JLM, Grandy AS (2021) Priming mechanisms providing plants and microbes access to mineral-associated organic matter. Soil Biol Biochem 158:108265. https://doi.org/10.1016/j.soilbio.2021.108265
Jones DL (1998) Organic acids in the rhizosphere – a critical review. Plant Soil 205:25–44. https://doi.org/10.1023/A:1004356007312
Jones DL, Prabowo AM, Kochian LV (1996) Kinetics of malate transport and decomposition in acid soils and isolated bacterial populations: the effect of microorganisms on root exudation of malate under Al stress. Plant Soil 182:239–247. https://doi.org/10.1007/BF00029055
Jones DL, Hodge A, Kuzyakov Y (2004) Plant and mycorrhizal regulation of rhizodeposition. New Phytol 163:459–480. https://doi.org/10.1111/j.1469-8137.2004.01130.x
Jones DL, Nguyen C, Finlay RD (2009) Carbon flow in the rhizosphere: carbon trading at the soil–root interface. Plant Soil 321:5–33. https://doi.org/10.1007/s11104-009-9925-0
Joshi SR, Morris JW, Tfaily MM et al (2021) Low soil phosphorus availability triggers maize growth stage specific rhizosphere processes leading to mineralization of organic P. Plant Soil 459:423–440. https://doi.org/10.1007/s11104-020-04774-z
Keiluweit M, Bougoure JJ, Nico PS et al (2015) Mineral protection of soil carbon counteracted by root exudates. Nat Clim Chang 5:588–595. https://doi.org/10.1038/nclimate2580
Kuzyakov Y, Blagodatskaya E (2015) Microbial hotspots and hot moments in soil: concept & review. Soil Biol Biochem 83:184–199. https://doi.org/10.1016/j.soilbio.2015.01.025
Kuzyakov Y, Friedel JK, Stahr K (2000) Review of mechanisms and quantification of priming effects. Soil Biol Biochem 32:1485–1498. https://doi.org/10.1016/S0038-0717(00)00084-5
Lehmann J, Kleber M (2015) The contentious nature of soil organic matter. Nature 528:60–68. https://doi.org/10.1038/nature16069
Li H, Bolscher T, Winnick M, Tfaily MM, Cardon ZG et al (2021) Simple plant and microbial exudates destabilize mineral-associated organic matter via multiple pathways. Environ Sci Technol 55(5):33–3398. https://doi.org/10.1021/acs.est.0c04592
Malý S, Královec J, Hampel D (2009) Effects of long-term mineral fertilization on microbial biomass, microbial activity, and the presence of r- and K-strategists in soil. Biol Fertil Soils 45:753–760. https://doi.org/10.1007/s00374-009-0388-5
Marschner P (2008) The role of rhizosphere microorganisms in relation to P uptake by plants BT - the ecophysiology of plant-phosphorus interactions. In: White PJ, Hammond JP (eds). Springer Netherlands, Dordrecht, pp 165–176
McAllister CF, Lepo JE (1983) Succinate transport by free-living forms of Rhizobium japonicum. J Bacteriol 153:1155–1162
McCune B, Grace JB, Urban D (2002) Analysis of ecological communities. MjM Software, Gleneden Beach, Oregon
McGill WB, Cole CV (1981) Comparative aspects of cycling of organic C, N, S and P through soil organic matter. Geoderma 26:267–286. https://doi.org/10.1016/0016-7061(81)90024-0
McNear DH Jr (2013) The rhizosphere-roots, soil and everything in between. Nat Educ Knowl 4:1
Menezes-Blackburn D, Paredes C, Zhang H et al (2016) Organic acids regulation of chemical–microbial phosphorus transformations in soils. Environ Sci Technol 50:11521–11531. https://doi.org/10.1021/acs.est.6b03017
Menezes-Blackburn D, Bol R, Klumpp E et al (2021) Citric acid effect on the abundance, size and composition of water-dispersible soil colloids and its relationship to soil phosphorus desorption: a case study. J Soil Sci Plant Nutr 21:2436–2446. https://doi.org/10.1007/s42729-021-00534-9
Monther MT, Kamaruzaman S (2012) Arbuscular mycorrhizal fungi and plant root exudates bio-communications in the rhizosphere. Afr J Microbiol Res 6:7295–7301
Murphy J, Riley JP (1962) A modified single solution method for the determination of phosphate in natural waters. Anal Chim Acta 27:31–36. https://doi.org/10.1016/S0003-2670(00)88444-5
Nannipieri P (1994) The potential use of soil enzymes as indicators of productivity, sustainability and pollution, soil biota management in sustainable farming systems. In: Pankhurst CE, Doube BM, Gupta VVSR, Grace PR (eds). CSIRO, East Melbourne, pp 238–244
Oliverio AM, Geisen S, Delgado-Baquerizo M et al (2020) The global-scale distributions of soil protists and their contributions to belowground systems. Sci Adv 6:eaax8787. https://doi.org/10.1126/sciadv.aax8787
Olsen AA, Rimstidt JD (2008) Oxalate-promoted forsterite dissolution at low pH. Geochim Cosmochim Acta 72(7):1758–1766. https://doi.org/10.1016/j.gca.2007.12.026
Paterson E (2003) Importance of rhizodeposition in the coupling of plant and microbial productivity. Eur J Soil Sci 54:741–750. https://doi.org/10.1046/j.1351-0754.2003.0557.x
Ramette A (2007) Multivariate analyses in microbial ecology. FEMS Microbiol Ecol 62:142–160. https://doi.org/10.1111/j.1574-6941.2007.00375.x
Ramos ME, Garcia-Palma S, Rozalen M, Johnston CT, Huertas FJ (2014) Kinetics of montmorilloite dissolution an experimental study of the effect of oxalate. Chem Geology 363:283–292. https://doi.org/10.1016/j.chemgeo.2013.11.014
Razavi BS, Zarebanadkouki M, Blagodatskaya E, Kuzyakov Y (2016) Rhizosphere shape of lentil and maize: spatial distribution of enzyme activities. Soil Biol Biochem 96:229–237. https://doi.org/10.1016/j.soilbio.2016.02.020
Reed JL, Vo TD, Schilling CH, Palsson BO (2003) An expanded genome-scale model of Escherichia coli K-12 (iJR904 GSM/GPR). Genome Biol 4:R54. https://doi.org/10.1186/gb-2003-4-9-r54
Renella G, Landi L, Valori F, Nannipieri P (2007) Microbial and hydrolase activity after release of low molecular weight organic compounds by a model root surface in a clayey and a sandy soil. Appl Soil Ecol 36:124–129. https://doi.org/10.1016/j.apsoil.2007.01.001
Rodríguez H, Fraga R, Gonzalez T, Bashan Y (2006) Genetics of phosphate solubilization and its potential applications for improving plant growth-promoting bacteria. Plant Soil 287:15–21
Rovira AD (1969) Plant root exudates. Bot Rev 35:35–57. https://doi.org/10.1007/BF02859887
Schindelin J, Arganda-Carreras I, Frise E et al (2012) Fiji: an open-source platform for biological-image analysis. Nat Methods 9:676–682. https://doi.org/10.1038/nmeth.2019
Schneider KD, Cade-Menun BJ, Lynch DH, Voroney RP (2016) Soil phosphorus forms from organic and conventional forage fields. Soil Sci Soc Am J 80(2):328–340. https://doi.org/10.2136/sssaj2015.09.0340
Schwertmann U (1991) Solubility and dissolution of iron oxides. Plant and Soil 130:1–25. https://www.jstor.org/stable/42937281. Accessed 5 Nov 2023
Sibbesen E (1983) Phosphate soil tests and their suitability to assess the phosphate status of soil. J Sci Food Agric 34:1368–1374. https://doi.org/10.1002/jsfa.2740341209
Sokol NW, Sanderman J, Bradford MA (2019) Pathways of mineral-associated soil organic matter formation: Integrating the role of plant carbon source, chemistry, and point of entry. Glob Chang Biol 25:12–24. https://doi.org/10.1111/gcb.14482
Spohn M, Kuzyakov Y (2013) Phosphorus mineralization can be driven by microbial need for carbon. Soil Biol Biochem 61:69–75. https://doi.org/10.1016/j.soilbio.2013.02.013
Spohn M, Kuzyakov Y (2014) Spatial and temporal dynamics of hotspots of enzyme activity in soil as affected by living and dead roots—a soil zymography analysis. Plant Soil 379:67–77. https://doi.org/10.1007/s11104-014-2041-9
Spohn M, Ermak A, Kuzyakov Y (2013) Microbial gross organic phosphorus mineralization can be stimulated by root exudates – a 33P isotopic dilution study. Soil Biol Biochem 65:254–263. https://doi.org/10.1016/j.soilbio.2013.05.028
Srinivasan K, Mahadevan R (2010) Characterization of proton production and consumption associated with microbial metabolism. BMC Biotechnol 10:2. https://doi.org/10.1186/1472-6750-10-2
Stumm W, Furrer G (1987) The dissolution of oxides and aluminum silicates: Examples of surface-coordination- controlled kinetics. In: Stumm W (ed) Aquatic surface chemistry. Wiley, Hoboken, pp 197–219
Tfaily MM, Chu RK, Toyoda J et al (2017) Sequential extraction protocol for organic matter from soils and sediments using high resolution mass spectrometry. Anal Chim Acta 972:54–61. https://doi.org/10.1016/j.aca.2017.03.031
Turner BL, Richardson AE (2004) Identification of scyllo-inositol phosphates in soil by solution phosphorus-31 nuclear magnetic resonance spectroscopy. Soil Sci Soc Am J 68(3):802–808. https://doi.org/10.2136/sssaj2004.8020
Wasaki J, Yamamura T, Shinano T, Osaki M (2003) Secreted acid phosphatase is expressed in cluster roots of lupin in response to phosphorus deficiency. Plant Soil 248:129–136. https://doi.org/10.1023/A:1022332320384
Wei L, Chen C, Xu Z (2010) Citric acid enhances the mobilization of organic phosphorus in subtropical and tropical forest soils. Biol Fertil Soils 46:765–769. https://doi.org/10.1007/s00374-010-0464-x
Wilson WA, Roach PJ, Montero M et al (2010) Regulation of glycogen metabolism in yeast and bacteria. FEMS Microbiol Rev 34:952–985. https://doi.org/10.1111/j.1574-6976.2010.00220.x
Yan F, Schubert S, Mengel K (1996) Soil pH increase due to biological decarboxylation of organic anions. Soil Biol Biochem 28:617–624. https://doi.org/10.1016/0038-0717(95)00180-8
Yuan Y, Zhao W, Zhang Z et al (2018) Impacts of oxalic acid and glucose additions on N transformation in microcosms via artificial roots. Soil Biol Biochem 121:16–23
Acknowledgements
We thank Joe Kupper, James W. Morris, and Martin Vandiviere for their help with soil sampling and analysis.
Funding
This work was supported in part by NIFA-AFRI award # 2016-67019-25281. A portion of this research was performed on a project award (https://doi.org/10.46936/genr.proj.2017.50047/60006261) from the Environmental Molecular Sciences Laboratory, a DOE Office of Science User Facility sponsored by the Biological and Environmental Research program under Contract No. DE-AC05-76RL01830.
Author information
Authors and Affiliations
Contributions
The study was conceptualized and designed by Sunendra R. Joshi and David H. McNear Jr. Material preparation, data collection and analysis were performed by Sunendra R. Joshi, David H. McNear Jr., Robert P. Young, and Malak M. Tfaily. The first draft of the manuscript was written by Sunendra R. Joshi with all authors providing review and comments. All authors read and approved of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors have no relevant financial or non-financial interests to disclose.
Additional information
Responsible Editor: N. Jim Barrow.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
ESM 1
(DOCX 1229 kb)
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Joshi, S.R., Tfaily, M.M., Young, R.P. et al. Root exudates induced coupled carbon and phosphorus cycling in a soil with low phosphorus availability. Plant Soil 498, 371–390 (2024). https://doi.org/10.1007/s11104-023-06442-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-023-06442-4