Abstract
Aims
Sugarcane plants infected with Leifsonia xyli subsp. xyli (Lxx) have their primary metabolism affected with decreased levels of sugars and amino acids. Cysteine and methionine are sulfur-containing essential amino acids used for bacterial growth and the Lxx titer in sugar cane leaves could affect the animo acid concentrations. The goal of this study was to evaluate how the increase in Lxx titer affects the nutritional status and sulfur metabolism in sugar cane leaves.
Methods
Susceptible sugar cane (Saccharum officinarum) genotype: CB49260 was used in this study with low (256 cells) and high (2090 cells) Lxx titers and macronutrients and primary metabolites assessed from leaves and culms.
Results
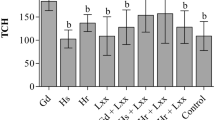
Plants with high Lxx titers accumulated more biomass in the main culm, leaves, and shoots than plants with low Lxx titers. Additionally, plants with high Lxx titers had 26% more sulfur content in leaves than plants with low Lxx titers. Higher levels of sulfate, sucrose, maltose, raffinose, shikimic acid, malate, putrescine, glycerol, and, erythritol were also present in plants with high Lxx titers; but decreased levels of methionine and glutathione in leaves. In the culm, plants with high Lxx titers also had increased levels of maltose; but decreased levels of threonine, ornithine, phenylalanine and myo-inositol when compared with plants with low Lxx titers.
Conclusions
This study demonstrated that high bacterial titers increase sulfur demand in sugar cane; however, the increased S content in the leaf did not result in higher sulfur assimilation, verified by increased sulfate but decreased methionine and glutathione levels. Therefore, our study showed that lower methionine availability, and methionine catabolism to putrescine in the leaves may fail to meet the increased sulfur organic compound demand of Lxx. The decrease in glutathione biosynthesis may reflect impaired biosynthesis or a drain on this antioxidant resulting from oxidative stresss by pathogenesis of Lxx.







Similar content being viewed by others
Data availability
The data that support the findings of this study are available from the corresponding author with a reasonable request.
Abbreviations
- APS:
-
Adenosine phosphosulfate
- DAP:
-
Days after planting
- DTNB:
-
5,5′-Dithiobis(2-nitrobenzoic acid)
- EDTA:
-
Ethylenediaminetetraacetic acid
- GR:
-
Glutathione Reductase
- GSH:
-
Reduced glutathione
- GSSH:
-
Oxidized glutathione
- GST:
-
Total glutathione
- Lxx :
-
Leifsonia xyli subsp. xyli
- MDA:
-
Malonoaldeyde
- NADPH:
-
Nicotinamide adenine dinucleotide phosphate
- PVPP:
-
Polyvinylpolypyrrolidone
- RSD:
-
Ratoon stunting disease
- SAM:
-
S-adenosylmethionine
- TCA:
-
Tricarboxylic acid
References
Álvarez C, Ángeles Bermúdez M, Romero LC, Álvarez C, Ángeles Bermúdez M, Romero LC, Gotor C, García I (2012) Cysteine homeostasis plays an essential role in plant immunity. New Phytol 193:165–177. https://doi.org/10.1111/j.1469-8137.2011.03889.x
Andreato C, Gazaffi R, Oliveira MMA, Andreato C, Gazaffi R, Oliveira MMA, Camargo LEA, Urashima AS (2022) Effect of thermotherapy, Leifsonia xyli subsp. xyli titres, sugarcane genotype and diagnostic techniques on ratoon stunt control in Brazil. J Appl Microbiol 133:1676–1687. https://doi.org/10.1111/jam.15671
Bailey RA, Bechet GR (1997) Further evidence of the effects of ratoon stunting disease on production under irrigated and rainfed conditions. In Proc S Afr Sug Technol Ass 71:97–101
Bloem E, Haneklaus S, Schnug E (2005) Significance of sulfur compounds in the protection of plants against pests and diseases. J Plant Nutr 28:763–784. https://doi.org/10.1081/PLN-200055532
Bloem E, Haneklaus S, Salac I, Bloem E, Haneklaus S, Salac I, Wickenhäuser P, Schnug E (2007) Facts and fiction about sulfur metabolism in relation to plant-pathogen interactions. Plant Biol 9:596–607. https://doi.org/10.1055/s-2007-965420
Bryson GM, Mills HA, Sasseville DN, Jones JB Jr, Barker AB (2014) Plant analysis handbook III: a guide to sampling, preparation, analysis, interpretation and use of results of agronomic and horticultural crop plant tissue. Macro-Micro Publishing, Inc, Athens, Georgia
Caldana C, Li Y, Leisse A, Caldana C, Li Y, Leisse A, Zhang Yi, Bartholomaeus L, Fernie AR, Willmitzer L, Giavalisco P (2013) Systemic analysis of inducible target of rapamycin mutants reveal a general metabolic switch controlling growth in Arabidopsis thaliana. Plant J 73:897–909. https://doi.org/10.1111/tpj.12080
Carvalho G, Silva TGER, Munhoz AT, Carvalho G, da Silva TGER, Munhoz AT, Monteiro-Vitorello CB, Azevedo RA, Melotto M, Camargo LEA (2016) Development of a qPCR for Leifsonia xyli subsp. xyli and quantification of the effects of heat treatment of sugarcane cuttings on Lxx. Crop Prot 80:51–55. https://doi.org/10.1016/j.cropro.2015.10.029
Castro-Moretti FR, Cocuron J-C, Cia MC, Castro-Moretti FR, Cocuron J-C, Cia MC, Cataldi TR, Labate CA, Alonso AP, Camargo LEA (2021) Targeted metabolic profiles of the Leaves and Xylem Sap of two sugarcane genotypes infected with the Vascular Bacterial Pathogen Leifsonia xyli subsp. xyli. Metabolites 11:234. https://doi.org/10.3390/metabo11040234
Chakraborty M, Ford R, Soda N et al (2023) Ratoon Stunting Disease (RSD) of Sugarcane : A Review Emphasizing Detection Strategies and Challenges. Phytopathology 1–46
Chen YP, Xing LP, Wu GJ, Chen Y-P, Xing L-P, Wu G-J, Wang H-Z, Wang X-E, Cao A-Z, Chen P-D (2007) Plastidial glutathione reductase from Haynaldia villosa is an enhancer of powdery mildew resistance in wheat (Triticum aestivum). Plant Cell Physiol 48:1702–1712. https://doi.org/10.1093/pcp/pcm142
Cia MC, de Carvalho G, Azevedo RA, Cia MC, de Carvalho G, Azevedo RA, Monteiro-Vitorello CB, Souza GM, Nishiyama-Junior MY, Lembke CG, Antunes de Faria RSdC, Marques JPR, Melotto M, Camargo LEA (2018) Novel insights into the early stages of Ratoon Stunting Disease of Sugarcane inferred from transcript and protein analysis. Phytopathology 108:1455–1466. https://doi.org/10.1094/PHYTO-04-18-0120-R
Davis MJ, Vidaver AM, Harris RW (1984) Clavibacter: a New Genus Containing Some Phytopathogenic Coryneform Bacteria, Including Clavibacter xyli subsp. xyli sp. nov., subsp. nov. and Pathogens That Cause Ratoon Stunting Disease of Sugarcane and Bermudagrass Stunting Disease Clavibacter. https://doi.org/10.1099/00207713-34-2-107
Davis MJ, Dean JL, Harrison NA (1988) Quantitative varability of Clavibacter xyli subsp. xyli population in sugarcane cultivars differing in resistance to ratoon stunting disease. Phytopathology 78:462–468
De Souza AP, Grandis A, Arenque-Musa BC, Buckeridge MS (2018) Diurnal variation in gas exchange and nonstructural carbohydrates throughout sugarcane development. Funct Plant Biol 45:865–876. https://doi.org/10.1071/FP17268
Dias VD, Filho RC, da Cunha MG (2019) Comparison of Leifsonia xyli subsp. xyli molecular detection in heat-treated sugarcane setts. Pesqui Agropecu Trop 49:1–7. https://doi.org/10.1590/1983-40632019v4955132
dos Santos Panta AM, Souza S, Gagliardi JL et al (2022) Investigation of Leifsonia xyli subsp. xyli in insects. Res Soc Dev 2022:1–9
Evtushenko LI, Dorofeeva LV, Subbotin SA et al (2000) Leifsonia poae gen. nov., sp. nov., isolated from nematode galls on Poa annua, and reclassification of ‘ Corynebacterium aquaticum ’ Leifson 1962 as Leifsonia aquatica (ex Leifson 1962) gen. nov., nom. rev., comb. nov. and Clavibacter xyli Da. Int J Syst Evol Microbiol 50:371–380
Faria RSCA, Cia MC, Monteiro-Vitorello CB et al (2020) Characterization of genes responsive to osmotic and oxidative stresses of the sugarcane bacterial pathogen Leifsonia xyli subsp. xyli. Brazilian J Microbiol 51:77–86. https://doi.org/10.1007/s42770-019-00163-6
Fatima U, Senthil-Kumar M (2015) Plant and pathogen nutrient acquisition strategies. Front Plant Sci 6:1–12. https://doi.org/10.3389/fpls.2015.00750
Ferreira DF (2011) SISVAR: a computer analysis system. Cienc e Agrotecnologia 35:1039–1042
Fu X, Li C, Zhou X, Fu X, Li C, Zhou X, Liu S, Wu F (2016) Physiological response and sulfur metabolism of the V. dahliae-infected tomato plants in tomato/potato onion companion cropping. Sci Rep 6:1–11. https://doi.org/10.1038/srep36445
Garcia FHS, Daneluzzi GS, Mazzafera P et al. (2021a) Ratoon Stunting Disease (Leifsonia xyli subsp. xyli) affects source-sink relationship in sugarcane by decreasing sugar partitioning to tillers. Physiol Mol Plant Pathol 116. https://doi.org/10.1016/j.pmpp.2021.101723
Garcia FHS, Herculino T, Coelho RD et al (2021b) Impact of the colonization of Leifsonia xyli subsp. xyli in a susceptible sugarcane genotype on water status and physiological traits. Eur J Plant Plant Pathol 159:1–11. https://doi.org/10.1007/s10658-021-02209-8
Gu G, Yang S, Yin X et al (2021) Sulfur induces resistance against canker caused by Pseudomonas syringae pv. Actinidae via phenolic components increase and morphological structure modification in the kiwifruit stems. Int J Mol Sci 22:1–13. https://doi.org/10.3390/ijms222212185
Guo Y, Hu M, Liu LL et al. (2019) Activities of key enzymes in the C4 pathway and anatomy of sugarcane infected by Leifsonia xyli subsp. xyli. J Appl Microbiol 1–11. https://doi.org/10.1111/jam.14444
Heath R, Packer L (1968) Photoperoxidation in isolated chloroplasts of fatty acid peroxidation chlorophyll. Arch Biochem biophisics 126:189–198
Heidari P, Mazloomi F, Nussbaumer T, Barcaccia G (2020) Insights into the SAM synthetase Gene Family and its roles in Tomato Seedlings under Abiotic stresses and hormone treatments. Plants 9:586
Jobe TO, Zenzen I, Karvansara PR, Kopriva S (2019) Integration of sulfate assimilation with carbon and nitrogen metabolism in transition from C 3 to C 4 photosynthesis. 70:4211–4221. https://doi.org/10.1093/jxb/erz250
Kashyap A, Planas-Marquès M, Capellades M, Kashyap A, Planas-Marquès M, Capellades M, Valls M, Coll NS (2021) Blocking intruders: inducible physico-chemical barriers against plant vascular wilt pathogens. J Exp Bot 72:184–198. https://doi.org/10.1093/jxb/eraa444
Kazeem SA, Ikotun B (2019) Effect of Leifsonia xyli subsp. xyli on growth and yield of sugarcane in Nigeria. J Plant Pathol 101:587–595. https://doi.org/10.1007/s42161-019-00246-1
Kopriva S, Koprivova A (2005) Sulfate assimilation and glutathione synthesis in C4 plants. Photosynth Res 86:363–372. https://doi.org/10.1007/s11120-005-3482-z
Koprivova A, Kopriva S (2016) Hormonal control of sulfate uptake and assimilation. Plant Mol Biol 91:617–627. https://doi.org/10.1007/s11103-016-0438-y
Lima K, Borges R, Salvato F et al (2018) Temporal dynamic responses of roots in contrasting tomato genotypes to cadmium tolerance. 245–258. https://doi.org/10.1007/s10646-017-1889-x
Lisec J, Schauer N, Kopka J, Lisec J, Schauer N, Kopka J, Willmitzer L, Fernie AR (2006) Gas chromatography mass spectrometry-based metabolite profiling in plants. Nat Protoc 1:387–396. https://doi.org/10.1038/nprot.2006.59
Lowe-Power TM, Hendrich CG, von Roepenack-Lahaye E et al (2018) Metabolomics of tomato xylem sap during bacterial wilt reveals Ralstonia solanacearum produces abundant putrescine, a metabolite that accelerates wilt disease. Environ Microbiol 20:1330–1349. https://doi.org/10.1111/1462-2920.14020
Lu C, Jiang Y, Yue Y et al (2023) Glutathione and neodiosmin feedback sustain plant immunity. J Exp Bot 74:976–990. https://doi.org/10.1093/jxb/erac442
Madhu SA, Kaur A et al (2022) Glutathione peroxidases in plants: innumerable role in abiotic stress tolerance and plant development. J Plant Growth Regul. https://doi.org/10.1007/s00344-022-10601-9
Malavolta E, Vitti GC, Oliveira SA (1997) Avaliação do estado nutricional das plantas: Princípios e aplicações. Potafos, Piracicaba
Marques JPR, Cia MC, Batista de Andrade Granato A, Muniz LF, Appezzato-da-Glória B, Camargo LEA (2022) Histopathology of the Shoot Apex of Sugarcane Colonized by Leifsonia xyli subsp. xyli. Phytopathology® 112(10):2062–2071
Martins SJ, de Medeiros FHV, de Souza RM et al (2015) Common bean growth and health promoted by rhizobacteria and the contribution of magnesium to the observed responses. Appl Soil Ecol 87:49–55. https://doi.org/10.1016/j.apsoil.2014.11.005
Monteiro-vitorello CB, Camargo LEA, Van Sluys MA et al (2004) The genome sequence of the gram-positive sugarcane pathogen leifsonia xyli subsp. xyli. Mol Plant-Microbe Interact 17:827–836. https://doi.org/10.1094/MPMI.2004.17.8.827
Quecine MC, Silva TM, Carvalho G, Quecine MC, Silva TM, Carvalho G, Saito S, Mondin M, Teixeira-Silva NS, Camargo LEA, Monteiro-Vitorello CB (2016) A stable Leifsonia xyli subsp. xyli GFP-tagged strain reveals a new colonization niche in sugarcane tissues. Plant Pathol 65:154–162. https://doi.org/10.1111/ppa.12397
Saez JV, Mariotti JA, Vega CRC (2019) Source-sink relationships during early crop development influence earliness of sugar accumulation in sugarcane. J Exp Bot 70:5157–5171. https://doi.org/10.1093/jxb/erz251
Sánchez-Elordi E, de los Ríos LM, Vicente C, Legaz ME (2019) Polyamines levels increase in smut teliospores after contact with sugarcane glycoproteins as a plant defensive mechanism. J Plant Res 132:405–417. https://doi.org/10.1007/s10265-019-01098-7
Santos EF, Mateus NK, Rosario MO, Garcez TB, Mazzafera P, Lavres (2021) Enhancing potassium content in leaves and stems improves drought tolerance of eucalyptus clones. Physiol Plant 172:552–563. https://doi.org/10.1111/ppl.13228
Takahashi H, Kopriva S, Giordano M, Takahashi H, Kopriva S, Giordano M, Saito K, Hell R (2011) Sulfur assimilation in photosynthetic organisms: molecular functions and regulations of transporters and assimilatory enzymes. Annu Rev Plant Biol 62:157–184. https://doi.org/10.1146/annurev-arplant-042110-103921
Urashima AS, Silva MF, Correa JJ, Moraes MC, Singh AV, Smith EC, Sainz MB (2017) Prevalence and severity of ratoon stunt in commercial Brazilian sugarcane fields. Plant Dis 101(5):815–821
Vilas JM, Romero FM, Rossi FR, Vilas JM, Romero FM, Rossi FR, Marina M, Maiale SJ, Calzadilla PI, Pieckenstain FL, Ruiz OA, Gárriz A (2018) Modulation of plant and bacterial polyamine metabolism during the compatible interaction between tomato and Pseudomonas syringae. J Plant Physiol 231:281–290. https://doi.org/10.1016/j.jplph.2018.09.014
Weckopp SC, Kopriva S (2014) Are changes in sulfate assimilation pathway needed for evolution of C4 photosynthesis? Front Plant Sci 5:773. https://doi.org/10.3389/fpls.2014.00773
Williams JS, Cooper RM (2003) Elemental sulphur is produced by diverse plant families as a component of defence against fungal and bacterial pathogens. Physiol Mol Plant Pathol 63:3–16. https://doi.org/10.1016/j.pmpp.2003.08.003
Yang Z, Hui S, Lv Y, Yang Z, Hui S, Lv Y, Zhang M, Chen D, Tian J, Zhang H, Liu H, Cao J, Xie W, Wu C, Wang S, Yuan M (2022) miR395-regulated sulfate metabolism exploits pathogen sensitivity to sulfate to boost immunity in rice. Mol Plant 15:671–688. https://doi.org/10.1016/j.molp.2021.12.013
Young AJ (2016) Possible origin of ratoon stunting disease following interspecific hybridization of Saccharum species. Plant Patholgoy 65:1403–1410. https://doi.org/10.1111/ppa.12545
Young A (2018) Turning a blind eye to ratoon stunting disease of sugarcane in Australia. Plant Dis 102:473–482. https://doi.org/10.1094/PDIS-06-17-0911-FE
Young AJ, Health C, Queensland S, Nock CJ (2017) Molecular detection of diverse Leifsonia strains Associated with Sugarcane. Plant Dis 101:1422–1431. https://doi.org/10.1094/PDIS-01-17-0016-RE
Zechmann B (2020) Subcellular roles of glutathione in mediating plant defense during biotic stress. Plants 9:1–21. https://doi.org/10.3390/plants9091067
Zhang X, Chen M, Liang Y et al (2016a) Morphological and physiological responses of sugarcane to Leifsonia xyli subsp. xyli infection. Plant Dis 100:1–8. https://doi.org/10.1094/PDIS-10-15-1134-RE
Zhang X, Liang YZ, Zhu K et al (2016b) Influence of Inoculation of Leifsonia xyli subsp. xyli on photosynthetic parameters and activities of defense enzymes in sugarcane. Sugar Tech 19:394–401. https://doi.org/10.1007/s12355-016-0479-1
Zhang Z, Long Y, Yin X, Yang S (2021) Sulfur-induced resistance against Pseudomonas syringae pv. Actinidiae via triggering salicylic acid signaling pathway in kiwifruit. Int J Mol Sci 22:12710. https://doi.org/10.3390/ijms222312710
Zhu K, Yuan D, Zhang X et al (2017) The physiological characteristics and associated gene expression of sugar cane inoculated with Leifsonia xyli subsp. xyli. J Phy 1–9. https://doi.org/10.1111/jph.12659
Acknowledgements
This study was financed in part by the Coordination for the Improvement of Higher Education Personnel (CAPES) - Brazil (CAPES) - Finance Code 001 and The Brazilian National Council for Scientific and Technological Development (CNPQ), through a scholarship (# 169260/2017-8). JL is grateful to CNPq for the productivity grant # 303718/2020-0. We would also like to thank the technicians Pedro Conceição Arthuso, Salete Aparecida Gaziola and Cleusa Pereira Cabral for their technical support in this study.
Author information
Authors and Affiliations
Contributions
FHSG designed the experiments, FHSG, APDJ, MN, SP, and JF performed the experiments, FHSG and SJM wrote the manuscript, FHSG and SP analyzed the data, FHSG, SP, and SJM created and edited figures, FHSG, APDJ, MN, SP, JL, SJM, ARF, and RAK revised this draft by rewriting, discussing and commenting. All authors read and approved the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The manuscript does not present any kind of conflict of interest.
Additional information
Responsible Editor: Christopher Guppy.
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Garcia, F.H.S., Domingues-Júnior, A.P., Lima Nogueira, M. et al. Impact of Leifsonia xyli subsp. xyli titer on nutritional status, and metabolism of sugar cane. Plant Soil 493, 341–354 (2023). https://doi.org/10.1007/s11104-023-06230-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-023-06230-0