Abstract
Background and aims
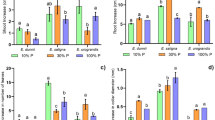
Twenty-four species of eucalypts were studied regarding their ability to grow under low P and their responsiveness to P inputs.
Methods
Growth and photosynthesis-related parameters were evaluated.
Results
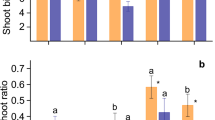
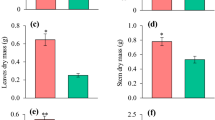
Growth of all species was influenced by low P availability. No significant correlation was found between leaf P concentration and biomass, indicating that P concentrations in leaves cannot be solely considered an indication of the responsiveness to P in eucalypts. Species responsive to P-input (high agronomic P efficiency values, APE) were those with low P use efficiency - PUE (here assessed as relative efficiency of P-use, REP) and low P uptake efficiency (PUpE). But, non-responsive species were related to higher P-efficiency under low soil P-availability. Eucalyptus tereticornis, E. cladocalyx, E. globulus and E. camaldulensis were efficient under low-P availability. Whereas, E. crebra and E acmenoides were the most responsive species, with high APE, suggesting that for these species P-inputs are needed to guarantee plant growth. The root:shoot ratio remained constant at different P availabilities, suggesting that biomass allocation towards the root in response to P and greater investment in roots were not correlated with greater PUE. Under limited P, E. robusta and E. botryoides exhibited low foliar P contents and higher root:shoot ratios than those of other species with higher P contents, indicating that greater root investment does not necessarily result in greater PUE.
Conclusion
The results suggest that the divergence among species is probably related to different mechanisms, which may improve P-use efficiency.






Similar content being viewed by others
References
Adams F, Reddell P, Webb MJ, Shipton WA (2006) Arbuscular mycorrhizas and ectomycorrhizas on Eucalyptus grandis (Myrtaceae) trees and seedlings in native forests of tropical North-Eastern Australia. Aust J Bot 54:271–281. https://doi.org/10.1071/BT05028
Bahar NHA, Gauthier PPG, O’Sullivan OS et al (2018) Phosphorus deficiency alters scaling relationships between leaf gas exchange and associated traits in a wide range of contrasting Eucalyptus species. Funct Plant Biol 45:813–826. https://doi.org/10.1071/FP17134
Bhering LL (2017) Rbio: a tool for biometric and statistical analysis using the R platform. Crop Breed Appl Biotechnol 17:187–190. https://doi.org/10.1590/1984-70332017v17n2s29
Bieleski RL (1973) Phosphate pools, phosphate transport, and phosphate availability. Annu Rev Plant Physiol 24:225–252. https://doi.org/10.1146/annurev.pp.24.060173.001301
Blair G (1993) Nutrient efficiency — what do we really mean?. In: Randall P.J., Delhaize E., Richards R.A., Munns R. (eds) Genetic Aspects of Plant Mineral Nutrition. Developments in Plant and Soil Sciences, vol 50. Springer, Dordrecht. pp 205–213
Bloomfield KJ, Farquhar GD, Lloyd J (2014) Photosynthesis–nitrogen relationships in tropical forest tree species as affected by soil phosphorus availability: a controlled environment study. Funct Plant Biol 41:820–832. https://doi.org/10.1071/FP13278
Bremner JM (1965) Total nitrogen. In: Black CA (ed) Methods of soil analysis. American Society of Agronomy, Madison, pp 1149–1178
Chandler JW, Dale JE (1993) Photosynthesis and nutrient supply in needles of Sitka spruce [Picea sitchensis (bong.) Carr.]. New Phytol 125:101–111. https://doi.org/10.1111/j.1469-8137.1993.tb03868.x
Cheng L, Bucciarelli B, Liu J et al (2011) White lupin cluster root acclimation to phosphorus deficiency and root hair development involve unique glycerophosphodiester phosphodiesterases. Plant Physiol 156:1131–1148. https://doi.org/10.1104/pp.111.173724
Chowdhury RB, Moore GA, Weatherley AJ, Arora M (2017) Key sustainability challenges for the global phosphorus resource, their implications for global food security, and options for mitigation. J Clean Prod 140:945–963. https://doi.org/10.1016/j.jclepro.2016.07.012
Clarkson DT (1981) Nutrient interception and transport by root systems. In: Johnson, C.B. ed. Physiological processes limiting plant productivity. Butterworths, London, 307–330
Clarkson DT, Hanson JB (1980) The mineral nutrition of higher plants. Annu Rev Plant Physiol 31:239–298. https://doi.org/10.1146/annurev.pp.31.060180.001323
Crous KY, Ósvaldsson A, Ellsworth DS (2015) Is phosphorus limiting in a mature Eucalyptus woodland? Phosphorus fertilisation stimulates stem growth. Plant Soil 391:293–305. https://doi.org/10.1007/s11104-015-2426-4
Del-Saz NF, Romero-Munar A, Cawthray GR et al (2018) Phosphorus concentration coordinates a respiratory bypass, synthesis and exudation of citrate, and the expression of high-affinity phosphorus transporters in Solanum lycopersicum. Plant Cell Environ 41:865–875. https://doi.org/10.1111/pce.13155
Dörmann P, Benning C, Joyard J et al (2002) Galactolipids rule in seed plants. Trends Plant Sci 7:112–118. https://doi.org/10.1016/S1360-1385(01)02216-6
Elser JJ, Bracken MES, Cleland EE et al (2007) Global analysis of nitrogen and phosphorus limitation of primary producers in freshwater, marine and terrestrial ecosystems. Ecol Lett 10:1135–1142. https://doi.org/10.1111/j.1461-0248.2007.01113.x
Ferreira DF (2011) Sisvar: a computer statistical analysis system. Ciência e Agrotecnologia 35:1039–1042. https://doi.org/10.1590/S1413-70542011000600001
Fixen PE, Johnston AM (2012) World fertilizer nutrient reserves: a view to the future. J Sci Food Agric 92:1001–1005. https://doi.org/10.1002/jsfa.4532
Fredeen AL, Rao IM, Terry N (1989) Influence of phosphorus nutrition on growth and carbon partitioning in Glycine max. Plant Physiol 89:225–230. https://doi.org/10.1104/pp.89.1.225
Friesen DK, Rao IM, Thomas RJ et al (1997) Phosphorus acquisition and cycling in crop and pasture systems in low fertility tropical soils. Plant Soil 196:289–294. https://doi.org/10.1023/A:1004226708485
Gamuyao R, Chin JH, Pariasca-Tanaka J et al (2012) The protein kinase Pstol1 from traditional rice confers tolerance of phosphorus deficiency. Nature 488:535–539. https://doi.org/10.1038/nature11346
Garrish V, Cernusak LA, Winter K, Turner BL (2010) Nitrogen to phosphorus ratio of plant biomass versus soil solution in a tropical pioneer tree, Ficus insipida. J Exp Bot 61:3735–3748. https://doi.org/10.1093/jxb/erq183
Gerloff GC (1977) Plant efficiencies in the use of nitrogen, phosphorus and potassium. In: Wright MJ (ed) Plant adaptation to mineral stress in problem soils. Cornell University Press, New York, pp 161–174
Gomez-Roldan V, Fermas S, Brewer PB et al (2008) Strigolactone inhibition of shoot branching. Nature 455:189–194. https://doi.org/10.1038/nature07271
Gotore T, Murepa R, Gapare WJ (2014) Effects of nitrogen, phosphorus and potassium on the early growth of Pinus patula and Eucalyptus grandis. J Trop For Sci 26:22–31. https://doi.org/10.2307/23617010
Grattapaglia D, Kirst M (2008) Eucalyptus applied genomics: from gene sequences to breeding tools. New Phytol 179:911–929. https://doi.org/10.1111/j.1469-8137.2008.02503.x
Güeswell S (2004) N: P ratios in terrestrial plants: Variation and functional significance. New Phytologist 164:243–266
Hammond JP, Broadley MR, White PJ et al (2009) Shoot yield drives phosphorus use efficiency in Brassica oleracea and correlates with root architecture traits. J Exp Bot 60:1953–1968. https://doi.org/10.1093/jxb/erp083
Hayes JE, Zhu YG, Mimura T, Reid RJ (2004) An assessment of the usefulness of solution culture in screening for phosphorus efficiency in wheat. Plant Soil 261:91–97. https://doi.org/10.1023/B:PLSO.0000035561.00460.8b
He H, Peng Q, Wang X et al (2017) Growth, morphological and physiological responses of alfalfa (Medicago sativa) to phosphorus supply in two alkaline soils. Plant Soil 416:565–584. https://doi.org/10.1007/s11104-017-3242-9
Hidaka A, Kitayama K (2011) Allocation of foliar phosphorus fractions and leaf traits of tropical tree species in response to decreased soil phosphorus availability on mount Kinabalu, Borneo. J Ecol 99:849–857. https://doi.org/10.1111/j.1365-2745.2011.01805.x
Huang G, Hayes PE, Ryan MH et al (2017) Peppermint trees shift their phosphorus-acquisition strategy along a strong gradient of plant-available phosphorus by increasing their transpiration at very low phosphorus availability. Oecologia 185:387–400. https://doi.org/10.1007/s00442-017-3961-x
Ikka T, Ogawa T, Li D et al (2013) Effect of aluminum on metabolism of organic acids and chemical forms of aluminum in root tips of Eucalyptus camaldulensis Dehnh. Phytochemistry 94:142–147. https://doi.org/10.1016/j.phytochem.2013.06.016
IUSS Working Group WRB (2015) World Reference Base for Soil Resources 2014, update 2015. International soil classification system for naming soils and creating legends for soil maps
Kelly AA, Froehlich JE, Dörmann P (2003) Disruption of the two digalactosyldiacylglycerol synthase genes DGD1 and DGD2 in Arabidopsis reveals the existence of an additional enzyme of galactolipid synthesis. Plant Cell 15:2694–2706. https://doi.org/10.1105/tpc016675
Koerselman W, Meuleman AFM (1996) The vegetation N:P ratio: a new tool to detect the nature of nutrient limitation. J Appl Ecol 33:1441–1450. https://doi.org/10.2307/2404783
Kramer-Walter KR, Laughlin DC (2017) Root nutrient concentration and biomass allocation are more plastic than morphological traits in response to nutrient limitation. Plant Soil 416:539–550. https://doi.org/10.1007/s11104-017-3234-9
Lambers H, Plaxton WC (2018) Phosphorus: back to the roots. Annu Plant Rev:3–22. https://doi.org/10.1002/9781119312994.apr0516
Lambers H, Brundrett MC, Raven JA, Hopper SD (2011) Plant mineral nutrition in ancient landscapes: high plant species diversity on infertile soils is linked to functional diversity for nutritional strategies. Plant Soil 348:7–27. https://doi.org/10.1007/s11104-011-0977-6
Lambers H, Hayes PE, Laliberté E et al (2015) Leaf manganese accumulation and phosphorus-acquisition efficiency. Trends Plant Sci 20:83–90
Lloyd J, Bloomfield K, Domingues TF, Farquhar GD (2013) Photosynthetically relevant foliar traits correlating better on a mass vs an area basis: of ecophysiological relevance or just a case of mathematical imperatives and statistical quicksand? New Phytol 199:311–321. https://doi.org/10.1111/nph.12281
Lynch JP (2011) Root phenes for enhanced soil exploration and phosphorus acquisition: tools for future crops. Plant Physiol 156:1041–1049. https://doi.org/10.1104/pp.111.175414
Marschner H (1995) The mineral nutrition of higher plants. Academic Press, London
Martinez HEP, Novais RF, Rodrigues LA, de Sacramento LVS (2005) Phosphate forms in plant and their internal buffering in five soybean cultivars. Rev Bras Ciência do Solo 29:249–257. https://doi.org/10.1590/S0100-06832005000200010
Morcuende R, Bari R, Gibon Y et al (2007) Genome-wide reprogramming of metabolism and regulatory networks of Arabidopsis in response to phosphorus. Plant Cell Environ 30:85–112. https://doi.org/10.1111/j.1365-3040.2006.01608.x
Moura JCMS, Bonine CAV, de Oliveira Fernandes Viana J et al (2010) Abiotic and biotic stresses and changes in the lignin content and composition in plants. J Integr Plant Biol 52:360–376. https://doi.org/10.1111/j.1744-7909.2010.00892.x
Mullette KJ, Hannon NJ, Elliott AGL (1974) Insoluble phosphorus usage by Eucalyptus. Plant Soil 41:199–205. https://doi.org/10.1007/BF00017956
Neto AP, Favarin JL, Hammond JP et al (2016) Analysis of phosphorus use efficiency traits in Coffea genotypes reveals Coffea arabica and Coffea canephora have contrasting phosphorus uptake and utilization efficiencies. Front Plant Sci 7:408. https://doi.org/10.3389/fpls.2016.00408
Neumann G (2016) The role of ethylene in plant adaptations for phosphate acquisition in soils – a review. Front Plant Sci 6:1–9. https://doi.org/10.3389/fpls.2015.01224
Niu YF, Chai RS, Jin GL et al (2013) Responses of root architecture development to low phosphorus availability: a review. Ann Bot 112:391–408. https://doi.org/10.1093/aob/mcs285
Novaes E (2010) Integrating genomics and quantitative genetics for discovery of genes that regulate bioenergy traits in woody species. University of Florida, Gainesville
Ozturk L, Eker S, Torun B, Cakmak I (2005) Variation in phosphorus efficiency among 73 bread and durum wheat genotypes grown in a phosphorus-deficient calcareous soil. Plant Soil 269:69–80. https://doi.org/10.1007/s11104-004-0469-z
Pérez-Cruzado C, Merino A, Rodríguez-Soalleiro R (2011) A management tool for estimating bioenergy production and carbon sequestration in Eucalyptus globulus and Eucalyptus nitens grown as short rotation woody crops in north-West Spain. Biomass Bioenergy 35:2839–2851. https://doi.org/10.1016/j.biombioe.2011.03.020
Poorter H, Niklas KJ, Reich PB et al (2012) Biomass allocation to leaves, stems and roots: meta-analyses of interspecific variation and environmental control. New Phytol 193:30–50. https://doi.org/10.1111/j.1469-8137.2011.03952.x
Raij B, van & Quaggio JA (2001) Determinação de fósforo, cálcio, magnésio e potássio extraídos com resina trocadora de íons. In: Raij, B.van; Andrade, J.C.; Cantarella, H. & Quaggio, J.A., eds. Análise química para avaliação da fertilidade de solos tropicais. Campinas, Secretaria de Agricultura e Abastecimento, APTA/IAC, 2001. p.189–199
Raij BV, Cantarella H, Quaggio JA, Furlani AMC (1997) Recomendações de adubação e calagem para o estado de São Paulo. Bol Técnico 100
Richardson AE, Simpson RJ (2011) Soil microorganisms mediating phosphorus availability update on microbial phosphorus. Plant Physiol 156:989–996. https://doi.org/10.1104/pp.111.175448
Robinson WD, Park J, Tran HT et al (2012) The secreted purple acid phosphatase isozymes AtPAP12 and AtPAP26 play a pivotal role in extracellular phosphate-scavenging by Arabidopsis thaliana. J Exp Bot 63:6531–6542. https://doi.org/10.1093/jxb/ers309
Ryser P, Lambers H (1995) Root and leaf attributes accounting for the performance of fast- and slow-growing grasses at different nutrient supply. Plant Soil 170:251–265. https://doi.org/10.1007/BF00010478
Salazar-Henao JE, Lin W-D, Schmidt W et al (2016) Discriminative gene co-expression network analysis uncovers novel modules involved in the formation of phosphate deficiency-induced root hairs in Arabidopsis. Sci Rep 6:26820. https://doi.org/10.1038/srep26820
Santana RC, Barros NF, Neves JCL (2002) Eficiência de utilização de nutrientes e sustentabilidade da produção em procedências de Eucalyptus grandis e Eucalyptus saligna em sítios florestais do Estado de São Paulo. Rev Árvore 26:447–457
Santiago LS, Wright SJ, Harms KE et al (2012) Tropical tree seedling growth responses to nitrogen, phosphorus and potassium addition. J Ecol 100:309–316. https://doi.org/10.1111/j.1365-2745.2011.01904.x
Smith S, Read D (2010) Mycorrhizal Symbiosis. Academic Press, London
Suriyagoda LDB, Ryan MH, Renton M, Lambers H (2010) Multiple adaptive responses of Australian native perennial legumes with pasture potential to grow in phosphorus- and moisture-limited environments. Ann Bot 105:755–767. https://doi.org/10.1093/aob/mcq040
Teng W, Kang Y, Hou W et al (2018) Phosphorus application reduces aluminum toxicity in two Eucalyptus clones by increasing its accumulation in roots and decreasing its content in leaves. PLoS One 13:1–20. https://doi.org/10.1371/journal.pone.0190900
Thomas DS, Montagu KD, Conroy JP (2006) Leaf inorganic phosphorus as a potential indicator of phosphorus status, photosynthesis and growth of Eucalyptus grandis seedlings. For Ecol Manag 223:267–274. https://doi.org/10.1016/j.foreco.2005.11.006
Turnbull TL, Adams MA, Warren CR (2007) Increased photosynthesis following partial defoliation of field-grown Eucalyptus globulus seedlings is not caused by increased leaf nitrogen. Tree Physiol 27:1481–1492. https://doi.org/10.1093/treephys/27.10.1481
Turner BL, Brenes-Arguedas T, Condit R (2018) Pervasive phosphorus limitation of tree species but not communities in tropical forests. Nature 555:367–370. https://doi.org/10.1038/nature25789
Uhde-Stone C, Zinn KE, Ramirez-Yáñez M et al (2003) Nylon filter arrays reveal differential gene expression in proteoid roots of white lupin in response to phosphorus deficiency. Plant Physiol 131:1064–1079. https://doi.org/10.1104/pp.102.016881
van de Wiel CCM, van der Linden CG, Scholten OE (2016) Improving phosphorus use efficiency in agriculture: opportunities for breeding. Euphytica 207:1–22. https://doi.org/10.1007/s10681-015-1572-3
Vance CP, Uhde-Stone C, Allan DL (2003) Phosphorus acquisition and use: critical adaptations by plants for securing a nonrenewable resource. New Phytol 157:423–447. https://doi.org/10.1046/j.1469-8137.2003.00695.x
Veneklaas EJ, Lambers H, Bragg J et al (2012) Opportunities for improving phosphorus-use efficiency in crop plants. New Phytol 195:306–320. https://doi.org/10.1111/j.1469-8137.2012.04190.x
Waddell HA, Simpson RJ, Lambers H et al (2016) Phosphorus-utilisation efficiency and leaf-morphology traits of Rytidosperma species (wallaby grasses) that differ in their growth response to phosphorus fertilisation. Aust J Bot 64:65–76. https://doi.org/10.1071/BT15202
Wang YP, Law RM, Pak B (2010) A global model of carbon, nitrogen and phosphorus cycles for the terrestrial biosphere. Biogeosciences 7:2261–2282. https://doi.org/10.5194/bg-7-2261-2010
Warcup JH (1980) Ectomycorrhizal associations of Australian indigenous plants. New Phytol 85:531–535. https://doi.org/10.1111/j.1469-8137.1980.tb00768.x
Warren CR (2011) How does P affect photosynthesis and metabolite profiles of Eucalyptus globulus? Tree Physiol 31:727–739. https://doi.org/10.1093/treephys/tpr064
Wrage N, Chapuis-Lardy L, Isselstein J (2010) Phosphorus, plant biodiversity and climate change. In: Lichtfouse E (ed) Sociology, organic farming, climate change and soil science. Sustainable agriculture reviews. Springer, New York, pp 147–169
Wright IJ, Reich PB, Cornelissen JHC et al (2005) Assessing the generality of global leaf trait relationships. New Phytol 166:485–496. https://doi.org/10.1111/j.1469-8137.2005.01349.x
Zavišić A, Nassal P, Yang N et al (2016) Phosphorus availabilities in beech (Fagus sylvatica L.) forests impose habitat filtering on ectomycorrhizal communities and impact tree nutrition. Soil Biol Biochem 98:127–137. https://doi.org/10.1016/j.soilbio.2016.04.006
Acknowledgements
This work was supported by the São Paulo Research Foundation (FAPESP – Grant number 2016/25498-0). We thank FAPESP, CAPES (Coordenação de Aperfeiçoamento de Pessoal de Nível Superior - Brasil) and Conselho Nacional de Desenvolvimento Científico e Tecnológico – Brasil (CNPq) for post-doctoral, doctoral and research fellowships to FMSO (Grant number 2018/09624-0), RGB and PM, respectively, and FAPESP for undergraduate fellowship to SB (Grant Number 2017/21738-9).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Responsible Editor: Hans Lambers.
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Bulgarelli, R.G., de Oliveira Silva, F.M., Bichara, S. et al. Eucalypts and low phosphorus availability: between responsiveness and efficiency. Plant Soil 445, 349–368 (2019). https://doi.org/10.1007/s11104-019-04316-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-019-04316-2