Abstract
Aims
Culture media compositions and bioprocess conditions were studied to improve the production of cell biomass and indolic phytohormones by Herbaspirillum seropedicae BR11471, a plant growth promoting bacterium, and different inoculant formulations were also produced and tested for their stability and shelf life.
Methods
Response surface methodology (RSM) based on central composite rotation designs (CCRD) was used to find bioprocess variables that lead to an increase in bacterial biomass and yield of indolic compounds. The major components of DYGS medium were optimized in small-scale shaken cultivations, in two sets of CCRD. High performance liquid chromatography was used to determine nutrient consumption and to correlate it with cell biomass production, and the Salkowski method was used to quantify indoles. Hydrolytic activity in the formulations was quantified with the fluorescein diacetate assay.
Results
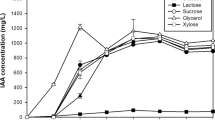
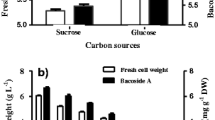
Glycerol (5.5 g L−1) and yeast extract (2.8 g L−1), as the main carbon and nitrogen sources, respectively, increased biomass production by 87.5% when compared to original DYGS medium, reaching 3.0 g L−1 of dry cell weight (DCW). In a 2.0 L bioreactor, the optimized medium was used to enhance process conditions for DCW and indole-3-acetic acid (IAA). Biomass production reached 3.4 g L−1 and was restrained at highest air flow levels. The conditions of 34-36 °C, 150 rpm and 4.0 L min−1 of air flow rate resulted in 11.97 mg L−1 of IAA, an increase of 370% over original DYGS at 30 °C. Peat can still be regarded as a good cell carrier for solid state inoculants, whilst the additives tested for liquid formulations are individually more efficient than the mixture.
Conclusions
The production of inoculants containing H. seropedicae strain BR11471 can be efficiently improved with the use of the RSM approach i.e. it maximizes the production of biomass and indolic compounds, and reduces culture media components, both key factors for large-scale industrial production.





Similar content being viewed by others
References
Adesemoye AO, Torbert HA, Kloepper JW (2009) Plant Growth-Promoting Rhizobacteria Allow Reduced Application Rates of Chemical Fertilizers. Microb Ecol 58:921–929. https://doi.org/10.1007/s00248-009-9531-y
Adnan NAA, Suhaimi SN, Abd-Aziz S, Hassan MA, Phang L-Y (2014) Optimization of bioethanol production from glycerol by Escherichia coli SS1. Renew Energy 66:625–633. https://doi.org/10.1016/j.renene.2013.12.032
Alves GC, Videira SS, Urquiaga S, Reis V (2015) Differential plant growth promotion and nitrogen fixation in two genotypes of maize by several Herbaspirillum inoculants. Plant Soil 387:307–321. https://doi.org/10.1007/s11104-014-2295-2
Amadeo I, Mauro LV, Ortí E, Forno G (2011) Determination of robustness and optimal work conditions for a purification process of a therapeutic recombinant protein using response surface methodology. Biotechnol Prog 27:724–732. https://doi.org/10.1002/btpr.588
Amiri S, Shakeri A, Sohrabi MR, Khalajzadeh S, Ghasemi E (2019) Optimization of ultrasonic assisted extraction of fatty acids from Aesculus hippocastanum fruit by response surface methodology. Food Chem 271:762–766. https://doi.org/10.1016/j.foodchem.2018.07.144
Baldani JI, Baldani VLD, Seldin L, Dobereiner J (1986) Characterization of Herbaspirillum seropedicae gen. Nov. sp. nov. a root-associated nitrogen-fixing bacterium. Int J Syst Bacteriol 36:86–93
Baldani JI, Reis VM, Videira SS, Boddey LH, Baldani VLD (2014) The art of isolating nitrogen-fixing bacteria from non-leguminous plants using N-free semi-solid media: a practical guide for microbiologists. Plant Soil 384:413–431. https://doi.org/10.1007/s11104-014-2186-6
Balsanelli E, Tadra-Sfeir MZ, Faoro H, Pankievicz VCS, de Baura VA, Pedrosa FO, de Souza EM, Dixon R, Monteiro RA (2015) Molecular adaptations of Herbaspirillum seropedicae during colonization of the maize rhizosphere. Environ Microbiol 18:2343–2356. https://doi.org/10.1111/1462-2920.12887
Bashan Y, Trejo A, de-Bashan LE (2011) Development of two culture media for mass cultivation of Azospirillum spp. and for production of inoculants to enhance plant growth. Biol Fertil Soils 47:963–969. https://doi.org/10.1007/s00374-011-0555-3
Bashan Y, de-Bashan LE, Prabhu SR, Hernandez J-P (2014) Advances in plant growth-promoting bacterial inoculant technology: formulations and practical perspectives (1998–2013). Plant Soil 378:1–33. https://doi.org/10.1007/s11104-013-1956-x
Bastián F, Cohen A, Piccoli P, Luna V, Baraldi R, Bottini R (1998) Production of indole-3-acetic acid and gibberellins A1 and A3 by Acetobacter diazotrophicus and Herbaspirillum seropedicae in chemically-defined culture media. Plant Growth Regul 24:7–11
Bernabeu PR, García SS, López AC, Vio SA, Carrasco N, Boiardi JL, Luna MF (2018) Assessment of bacterial inoculant formulated with Paraburkholderia tropica to enhance wheat productivity. W J Microbiol Biotechnol 34:81–91. https://doi.org/10.1007/s11274-018-2461-4
Berninger T, González-López O, Bejarano A, Preininger C, Sessitsch A (2018) Maintenance and assessment of cell viability in formulation of non-sporulating bacterial inoculants. Microb Biotechnol 11:277–301. https://doi.org/10.1111/1751-7915.12880
Canellas LP, Balmori DM, Médici LO, Aguiar NO, Campostrini E, Rosa RCC, Façanha AR, Olivares FL (2013) A combination of humic substances and Herbaspirillum seropedicae inoculation enhances the growth of maize (Zea mays L). Plant Soil 366:119–132. https://doi.org/10.1007/s11104-012-1382-5
Cappuyns AM, Bernaerts K, Smets IY, Ona O, Prinsen E, Vanderleyden J, Van Impe JF (2007) Optimal fed batch experiment design for estimation of monod kinetics of Azospirillum brasilense: from theory to practice. Biotechnol Prog 23:1074–1081
Chebotar VK, Malfanova NV, Shcherbakov AV, Ahtemova GA, Borisov AY, Lugtenberg B, Tikhonovich IA (2015) Endophytic Bacteria in microbial preparations that improve plant development (review). Appl Biochem Microbiol 51(3):271–277. https://doi.org/10.1134/S0003683815030059
Chen J, Huang PT, Zhang KY, Ding FR (2012) Isolation of biosurfactant producers, optimization and properties of biosurfactant produced by Acinetobacter sp. from petroleum-contaminated soil. J Appl Microbiol 112:660–671. https://doi.org/10.1111/j.1365-2672.2012.05242.x
Compant S, Clément C, Sessitsch A (2010) Plant growth-promoting bacteria in the rhizo- and endosphere of plants: their role, colonization, mechanisms involved and prospects for utilization. Soil Biol Biochem 42:669–678. https://doi.org/10.1016/j.soilbio.2009.11.024
Estrada GA, Baldani VLD, Oliveira DM, Urquiaga S, Baldani JI (2013) Selection of phosphate-solubilizing diazotrophic Herbaspirillum and Burkholderia strains and their effect on rice crop yield and nutrient uptake. Plant Soil 369:115–129. https://doi.org/10.1007/s11104-012-1550-7
Green VS, Stott DE, Diack M (2006) Assay for fluorescein diacetate hydrolytic activity: Optimization for soil samples. Soil Biol Biochem 38:693701. https://doi.org/10.1016/j.soilbio.2005.06.020
Hajji M, Rebai A, Gharsallah N, Nasri M (2008) Optimization of alkaline protease production by Aspergillus clavatus ES1 in Mirabilis jalapa tuber powder using statistical experimental design. Appl Microbiol Biotechnol 79:915–923. https://doi.org/10.1007/s00253-008-1508-0
Hallenbeck PC, Grogger M, Mraz M, Veverka D (2015) The use of design of experiments and response surface methodology to optimize biomass and lipid production by the oleaginous marine green alga, Nannochloropsis gaditana in response to light intensity, inoculum size and CO2. Bioresour Technol 184:161–168. https://doi.org/10.1016/j.biortech.2014.09.022
Hayat R, Ali S, Amara U, Khalid R, Ahmed I (2010) Soil beneficial bacteria and their role in plant growth promotion: a review. Ann Microbiol 60:579–598. https://doi.org/10.1007/s13213-010-0117-1
Heck JX, Soares LHB, Ayub MAZ (2005) Optimization of xylanase and mannanase production by Bacillus circulans strain BL53 on solid-state cultivation. Enz Microb Technol 37:417–423. https://doi.org/10.1016/j.enzmictec.2005.02.015
James EK, Gyaneshwar P, Mathan N, Barraquio QL, Reddy PM, Iannetta PPM, Olivares FL, Ladha JK (2002) Infection and colonization of rice seedlings by the plant growth-promoting bacterium Herbaspirillum seropedicae Z67. Mol Plant Microbe Interact 15:894–906. https://doi.org/10.1094/Mpmi.2002.15.9.894
Liu J, Li G, Sui Y (2017) Optimization of culture medium enhances viable biomass production and biocontrol efficacy of the antagonistic yeast Candida diversa. Front Microbiol 8:1–7. https://doi.org/10.3389/fmicb.2017.02021
Lobo CB, Tomás MSJ, Viruel E, Ferrero MA, Lucca ME (2018) Development of low-cost formulations of plant growth-promoting bacteria to be used as inoculants in beneficial agricultural technologies. Microbiol Res 219:12–25. https://doi.org/10.1016/j.micres.2018.10.012
Lotfy WA, Ghanem KM, El-Helow ER (2007) Citric acid production by a novel Aspergillus niger isolate: II. Optimization of process parameters through statistical experimental designs. Bioresour Technol 98:3470–3477. https://doi.org/10.1016/j.biortech.2006.11.032
Lucy M, Reed E, Glick BR (2004) Applications of free living plant growth-promoting rhizobacteria. Antonie Van Leeuwenhoek 86:1–25. https://doi.org/10.1023/B:ANTO.0000024903.10757.6e
Monteiro RA, Balsanelli E, Wassem R, Marin AM, Brusamarello-Santos LCC, Schmidt MA, Tadra-Sfeir MZ, Pankievicz VCS, Cruz LM, Chubatsu LS, Pedrosa FO, Souza EM (2012) Herbaspirillum-plant interactions: microscopical, histological and molecular aspects. Plant Soil 356:175–196. https://doi.org/10.1007/s11104-012-1125-7
Mutalik SR, Vaidya BK, Joshi RM, Desai KM, Nene SN (2008) Use of response surface optimization for the production of biosurfactant from Rhodococcus spp. MTCC 2574. Bioresour Technol 99:7875–7880. https://doi.org/10.1016/j.biortech.2008.02.027
Myers RH, Montgomery DC, Anderson-Cook CM (2016) Response surface methodology: process and product optimization using designed experiments, 4th Edition. John Wiley & Sons. ISBN: 978-1-118-91601-8
O'Callaghan M (2016) Microbial inoculation of seed for improved crop performance: issues and opportunities. Appl Microbiol Biotechnol 100:5729–5746. https://doi.org/10.1007/s00253-016-7590-9
Ona O, Van IJ, Prinsen E, Vanderleyden J (2005) Growth and indole-3-acetic acid biosynthesis of Azospirillum brasilense Sp245 is environmentally controlled. FEMS Microbiol Lett 246:125–132
Pandey A, Belwal T, Sekar KC, Bhat ID, Rawal RS (2018) Optimization of ultrasonic-assisted extraction (UAE) of phenolics and antioxidant compounds from rhizomes of Rheum moorcroftianum using response surface methodology (RSM). Ind Crop Prod 119:218–225. https://doi.org/10.1016/j.indcrop.2018.04.019
Pedraza RO, Ramirez-Mata A, Xiqui ML, Baca ME (2004) Aromatic amino acid aminotransferase activity and indole-3-acetic acid production by associative nitrogen-fixing bacteria. FEMS Microbiol Lett 233:15–21
Richardson AE, Barea J-M, McNeill AM, Prigent-Combaret C (2009) Acquisition of phosphorus and nitrogen in the rhizosphere and plant growth promotion by microorganisms. Plant Soil 321:305–339. https://doi.org/10.1007/s11104-009-9895-2
Rosconi F, Davyt D, Martínez V, Martínez M, Abin-Carriquiry JA, Zane H, Butler A, de Souza EM, Fabiano E (2013) Identification and structural characterization of serobactins, a suite of lipopeptide siderophores produced by the grass endophyte Herbaspirillum seropedicae. Environ Microbiol 15:916–927. https://doi.org/10.1111/1462-2920.12075
Rothballer M, Eckert B, Schmid M, Fekete A, Schloter M, Lehner A, Pollmann S, Hartmann A (2008) Endophytic root colonization of gramineous plants by Herbaspirillum frisingense. FEMS Microbiol Ecol 66:85–95. https://doi.org/10.1111/j.1574-6941.2008.00582.x
Sarwar M, Kremer RJ (1995) Determination of bacterially derived auxins using a microplate method. Lett Appl Microbiol 20:282–285
Sharma D, Yadav KD, Kumar S (2018) Biotransformation of flower waste composting: optimization of waste combinations using response surface methodology. Bioresour Technol 270:198–2017. https://doi.org/10.1016/j.biortech.2018.09.036
Silva MF, Antônio CS, Oliveira PJ, Xavier GR, Rumjanek NG, Soares LHB, Reis VM (2012) Survival of endophytic bacteria in polymer-based inoculants and efficiency of their application to sugarcane. Plant Soil 356:231–243. https://doi.org/10.1007/s11104-012-1242-3
Spaepen S, Vanderleyden J, Remans R (2007) Indole-3-acetic acid in microbial and microorganism-plant signaling. FEMS Microbiol Rev 31:425–448
Stephens JHG, Rask HM (2000) Inoculant production and formulation. Field Crop Res 65:249–258. https://doi.org/10.1016/S0378-4290(99)00090-8
Trovero MF, Scavone P, Platero R, Souza EM, Fabiano E, Rosconi F (2018) Herbaspirillum seropedicae differentially expressed genes in response to iron availability. Front Microbiol 9:1–12. https://doi.org/10.3389/fmicb.2018.01430
Trujillo-Roldán MA, Valdez-Cruz NA, Gonzalez-Monterrubio CF, Acevedo-Sánchez EV, Martínez-Salinas C, García-Cabrera RI, Gamboa-Suasnavart RA, Marín-Palacio LD, Villegas J, Blancas-Cabrera A (2013) Scale-up from shake flasks to pilot-scale production of the plant growth-promoting bacterium Azospirillum brasilense for preparing a liquid inoculant formulation. Appl Microbiol Biotechnol 97:9665–9677. https://doi.org/10.1007/s00253-013-5199-9
Wagh J, Bhandari P, Shah S, Archana G, Kumar GN (2014) Overexpression of citrate operon in Herbaspirillum seropedicae Z67 enhances organic acid secretion, mineral phosphate solubilization and growth promotion of Oryza sativa. Plant Soil 383:73–86. https://doi.org/10.1007/s11104-014-2161-2
Xie T, Sun Y, Du K, Liang B, Cheng R, Zhang Y (2012) Optimization of heterotrophic cultivation of Chlorella sp. for oil production. Bioresour Technol 118:235–242. https://doi.org/10.1016/j.biortech.2012.05.004
Acknowledgements
The authors would like to thank the financial support from Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES – Grant number 001), Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq), and Fundação Carlos Chagas Filho de Amparo à Pesquisa do Estado do Rio de Janeiro (FAPERJ).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Responsible Editor: Euan K. James.
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(DOCX 24 kb)
Rights and permissions
About this article
Cite this article
Scheidt, W., dos Santos Pedroza, I.C.P., Fontana, J. et al. Optimization of culture medium and growth conditions of the plant growth-promoting bacterium Herbaspirillum seropedicae BR11417 for its use as an agricultural inoculant using response surface methodology (RSM). Plant Soil 451, 75–87 (2020). https://doi.org/10.1007/s11104-019-04172-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-019-04172-0