Abstract
Background and aims
The canopies in evergreen coniferous plantations are often composed of various-aged needles. Plantation management, such as thinning, produced abundant harvest residue, including needles with different needle ages. However, little attention was paid to the effect of needle age on decomposition, although the needle chemical properties varied substantially with leaf ages.
Methods
A field experiment was conducted for 3 years to investigate the decomposition of harvest residue needles at different needle ages, and determine the main controlling factors in different stages of decomposition in a Chinese fir plantation.
Results
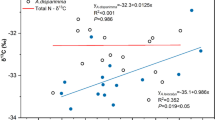
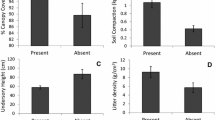
We found that the initial decomposition rate varied 5-fold among different needle ages in a Chinese fir plantation. Litter quality controlled the overall litter decomposition rate, especially the initial decomposition rate. Needle nitrogen content was positively correlated to decomposition rate during the early stage of decomposition. However, it was negatively correlated to decomposition rate during the later stage of decomposition. The contents of needle tannins increased the asymptotic mass remaining (A, proportion of mass remaining at which decomposition approaches zero, i.e., the fraction of slowly decomposing litter). We also found that the initial litter decomposition rates in soil fauna presence were significantly higher than those in soil fauna absence across different needle ages. Moreover, the effect of soil fauna on initial litter decomposition is independent of needle quality.
Conclusions
Our results suggest that needle age and plant secondary metabolites should be considered to understand the response of litter decomposition and nutrient cycling to management practices, such as thinning, in conifer plantations.





Similar content being viewed by others
References
Berg B (2000) Initial rates and limit values for decomposition of scots pine and Norway spruce needle litter: a synthesis for N-fertilized forest stands. Can J For Res 30:122–135
Berg B (2014) Decomposition patterns for foliar litter–a theory for influencing factors. Soil Biol Biochem 78:222–232
Berg B, Ekbohm G (1991) Litter mass-loss rates and decomposition patterns in some needle and leaf litter types. Long-term decomposition in a scots pine forest. VII. Can J Bot 69:1449–1456
Berg B, Meentemeyer V (2002) Litter quality in a north European transect versus carbon storage potential. Plant Soil 242:83–92
Berg B, Steffen K, McClaugherty C (2007) Litter decomposition rate is dependent on litter Mn concentrations. Biogeochemistry 82:29–39
Berg B, Davey MP, De Marco A, Emmett B, Faituri M, Hobbie SE, Johansson MB, Liu C, McClaugherty C, Norell L, Rutigliano FA, Vesterdal L, Virzo De Santo A (2010) Factors influencing limit values for pine needle litter decomposition: a synthesis for boreal and temperate pine forest systems. Biogeochemistry 100:57–73
Chomel M, Fernandez C, Bousquet-Mélou A, Gers C, Monnier Y, Santonja M, Gauquelin T, Gros R, Lecareux C, Baldy V (2014) Secondary metabolites of Pinus halepensis Alter decomposer organisms and litter decomposition during afforestation of abandoned agricultural zones. J Ecol 102:411–424
Chomel M, Guittonny-Larchevêque M, Fernandez C, Gallet C, DesRochers A, Paré D, Jackson BG, Baldy V (2016) Plant secondary metabolites: a key driver of litter decomposition and soil nutrient cycling. J Ecol 104:1527–1541
Coq S, Souquet JM, Meudec E, Cheynier V, Hättenschwiler S (2010) Interspecific variation in leaf litter tannins drives decomposition in a tropical rain forest of French Guiana. Ecology 91:2080–2091
Cornelissen JHC, Perez-Harguindeguy N, Diaz S, Grime JP, Marzano B, Cabido M, Vendramini F, Cerabolini B (1999) Leaf structure and defence control litter decomposition rate across species and life forms in regional floras on two continents. New Phytol 143:191–200
Cornwell WK, Cornelissen JHC, Amatangelo K, Dorrepaal E, Eviner VT, Godoy O, Hobbie SE, Hoorens B, Kurokawa H, Pérez-Harguindeguy N, Quested HM, Santiago LS, Wardle DA, Wright IJ, Aerts R, Allison SD, van Bodegom P, Brovkin V, Chatain A, Callaghan TV, Díaz S, Garnier E, Gurvich DE, Kazakou E, Klein JA, Read J, Reich PB, Soudzilovskaia NA, Vaieretti MV, Westoby M (2008) Plant species traits are the predominant control on litter decomposition rates within biomes worldwide. Ecol Lett 11:1065–1071
DeForest JL, Zak DR, Pregitzer KS, Burton AJ (2004) Atmospheric nitrate deposition, microbial community composition, and enzyme activity in northern hardwood forests. Soil Sci Soc Am J 68:132–138
Escudero A, Mediavilla S (2003) Decline in photosynthetic nitrogen use efficiency with leaf age and nitrogen resorption as determinants of leaf life span. J Ecol 91:880–889
Forkner RE, Marquis RJ, Lill JT (2004) Feeny revisited: condensed tannins as anti-herbivore defences in leaf-chewing herbivore communities of Quercus. Ecol Entomol 29:174–187
Frost CJ, Dean JM, Smyers EC, Mescher MC, Carlson JE, De Moraes CM, Tooker JF (2012) A petiole-galling insect herbivore decelerates leaf lamina litter decomposition rates. Funct Ecol 26:628–636
Fukushima RS, Hatfield RD (2001) Extraction and isolation of lignin for utilization as a standard to determine lignin concentration using the acetyl bromide spectrophotometric method. J Agric Food Chem 49:3133–3139
González G, Seastedt TR (2001) Soil fauna and plant litter decomposition in tropical and subalpine forests. Ecology 82:955–964
Hobbie SE (2005) Contrasting effects of substrate and fertilizer nitrogen on the early stages of decomposition. Ecosystems 8:644–656
Hobbie SE (2008) Nitrogen effects on litter decomposition: a five-year experiment in eight temperate grassland and forest sites. Ecology 89:2633–2644
Hobbie SE, Eddy WC, Buyarski CR, Adair CE, Ogdahl ML, Weisenhorn P (2012) Response of decomposing litter and its microbial community to multiple forms of nitrogen enrichment. Ecol Monogr 82:389–405
Howell CR, Bell AA, Stipanovic RD (1976) Effect of aging on flavonoid content and resistance of cotton leaves to verticillium wilt. Physiol Plant Pathol 8:181–188
Hyvönen R, Olsson BA, Lundkvist H, Staaf H (2000) Decomposition and nutrient release from Picea abies (L.) karst. And Pinus sylvestris L. logging residues. For Ecol Manag 126:97–112
Larcher W (2003) Physiological plant ecology. Springer, Heidelberg
Li RS, Yang QP, Zhang WD, Zheng WH, Chi YG, Xu M, Fang YT, Gessler A, Li MH, Wang SL (2017) Thinning effect on photosynthesis depends on needle ages in a Chinese fir (Cunninghamia lanceolata) plantation. Sci Total Environ 580:900–906
Liu ZJ, Carpenter SB, Bourgeois WJ, Yu Y, Constantin RJ, Falcon MJ, Adams JC (1998) Variations in the secondary metabolite camptothecin in relation to tissue age and season in Camptotheca acuminata. Tree Physiol 18:265–270
Loranger G, Ponge JF, Imbert D, Lavelle P (2002) Leaf decomposition in two semi-evergreen tropical forests: influence of litter quality. Biol Fertil Soils 35:247–252
Makkonen M, Berg MP, Handa IT, Hättenschwiler S, van Ruijven J, van Bodegom PM, Aerts R (2012) Highly consistent effects of plant litter identity and functional traits on decomposition across a latitudinal gradient. Ecol Lett 15:1033–1041
Mediavilla S, González-Zurdo P, García-Ciudad A, Escudero A (2011) Morphological and chemical leaf composition of Mediterranean evergreen tree species according to leaf age. Trees 25:669–677
Moorhead DL, Sinsabaugh RL (2006) A theoretical model of litter decay and microbial interaction. Ecol Monogr 76:151–174
Nierop KGJ, Preston CM, Verstraten JM (2006) Linking the B ring hydroxylation pattern of condensed tannins to C, N and P mineralization. A case study using four tannins. Soil Biol Biochem 38:2794–2802
Olsen SR, Sommers LE (1982) Phosphorus. In: Page, A.L., Miller, R.H. and Keeney, D.R., Eds., Methods of soil analysis. Part 2: chemical and microbiological properties, American Society of Agronomy, Inc., Madison, pp 403–430
Olson JS (1963) Energy storage and the balance of producers and decomposers in ecological systems. Ecology 44:322–331
Page V, Feller U (2005) Selective transport of zinc, manganese, nickel, cobalt and cadmium in the root system and transfer to the leaves in young wheat plants. Ann Bot 96:425–434
Palviainen M, Finér L, Kurka AM, Mannerkoski H, Piirainen S, Starr M (2004) Decomposition and nutrient release from logging residues after clear-cutting of mixed boreal forest. Plant Soil 263:53–67
Pérez-Harguindeguy N, Díaz S, Cornelissen J, Vendramini F, Cabido M, Castellanos A (2000) Chemistry and toughness predict leaf litter decomposition rates over a wide spectrum of functional types and taxa in central Argentina. Plant Soil 218:21–30
Powers JS, Montgomery RA, Adair EC, Brearley FQ, DeWalt SJ, Castanho CT, Chave J, Deinert E, Ganzhorn JU, Gilbert ME, Gonzalez-Iturbe JA, Bunyavejchewin S, Grau HR, Harms KE, Hiremath A, Iriarte-Vivar S, Manzane E, de Oliveira AA, Poorter L, Ramanamanjato JB, Salk C, Varela A, Weiblen GD, Lerdau MT (2009) Decomposition in tropical forests: a pan-tropical study of the effects of litter type, litter placement and meso-faunal exclusion across a precipitation gradient. J Ecol 97:801–811
Rodríguez-Gutiérrez G, Rubio-Senent F, Lama-Muñoz A, García A, Fernández-Bolaños J (2014) Properties of lignin, cellulose, and hemicelluloses isolated from olive cake and olive stones: binding of water, oil, bile acids, and glucose. J Agric Food Chem 62:8973–8981
Schweitzer JA, Bailey JK, Hart SC, Wimp GM, Chapman SK, Whitham TG (2005) The interaction of plant genotype and herbivory decelerate leaf litter decomposition and alter nutrient dynamics. Oikos 110:133–145
Smith VC, Bradford MA (2003) Litter quality impacts on grassland litter decomposition are differently dependent on soil fauna across time. Appl Soil Ecol 24:197–203
State Forestry Administration of China (2014) Forest resources report in China (2009–2013). China Forestry Publishing House, Beijing
Sterner RW, Elser J (2002) Ecological stoichiometry. Princeton University Press, Princeton
Szymura TH (2009) Concentration of elements in silver fir (Abies alba Mill.) needles as a function of needles’ age. Trees 23:211–217
Taylor BR, Parkinson D, Parsons WFJ (1989) Nitrogen and lignin content as predictors of litter decay-rates – a microcosm test. Ecology 70:97–104
Updegraff DM (1969) Semimicro determination of cellulose in biological materials. Anal Biochem 32:420–424
Wright IJ, Leishman MR, Read C, Westoby M (2006) Gradients of light availability and leaf traits with leaf age and canopy position 28 Australian shrubs and trees. Funct Plant Biol 33:407–419
Yang XD, Chen J (2009) Plant litter quality influences the contribution of soil fauna to litter decomposition in humid tropical forests, southwestern China. Soil Biol Biochem 41:910–918
Yang QP, Xu M, Chi YG, Zheng YP, Shen RC, Wang SL (2014) Effects of freeze damage on litter production, quality and decomposition in a loblolly pine forest in central China. Plant Soil 374:449–458
Yang QP, Liu LL, Zhang WD, Xu M, Wang SL (2015) Different responses of stem and soil CO2 efflux to pruning in a Chinese fir (Cunninghamialanceolata) plantation. Trees 29:1207–1218
Yue K, Yang WQ, Peng CH, Peng Y, Zhang C, Huang CP, Tan Y, Wu FZ (2016) Foliar litter decomposition in an alpine forest meta-ecosystem on the eastern Tibetan plateau. Sci Total Environ 566–567:279–287
Zhang WD, Yuan SF, Hu N, Lou YL, Wang SL (2015) Predicting soil fauna effect on plant litter decomposition by using boosted regression trees. Soil Biol Biochem 82:81–86
Zhang WD, Chao L, Yang QP, Wang QK, Fang YT, Wang SL (2016) Litter quality mediated nitrogen effect on plant litter decomposition regardless of soil fauna presence. Ecology 97:2834–2843
Zhou HR, Xu M, Pan HL, XB Y (2015) Leaf-age effects on temperature responses of photosynthesis and respiration of an alpine oak, Quercus aquifolioides, in southwestern China. Tree Physiol 35:1236–1248
Acknowledgments
This work was supported by the National Key Research and Development Program of China (Grant no. 2016YFD0600204), the National Natural Science Foundation of China (Grant Nos. 41630755 and 31570402), and the National Basic Research Program of China (973 Program, Grant No. 2012CB416905). We also thank Xiuyong Zhang, Zhengqi Shen, Xiaojun Yu, Ke Huang and Munan Zhu for their invaluable assistance in the laboratory and the field experiments.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Responsible Editor: Alfonso Escudero.
Electronic supplementary material
ESM 1
(DOCX 19 kb)
Rights and permissions
About this article
Cite this article
Yang, Q., Li, R., Zhang, W. et al. Decomposition of harvest residue needles of different needle ages in a Chinese fir (Cunninghamia lanceolata) plantation. Plant Soil 423, 273–284 (2018). https://doi.org/10.1007/s11104-017-3515-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-017-3515-3