Abstract
Background and aim
Saccharothrix algeriensis NRRL B-24137, isolated from a Saharan soil, has been described as a potential biocontrol agent against Botrytis cinerea and other phytopathogens. However, the plant protection mechanisms involved still need to be described. The aim of this study was to determine this protection phenomenon as well as parts of the mechanisms involved, using Arabidopsis thaliana seedlings and B. cinerea.
Methods
The bacterial colonization process was evaluated on A. thaliana seedlings using fluorescence in situ hybridization. Protection of A. thaliana seedlings inoculated with NRRL B-24137 against B. cinerea was then evaluated. Parts of the mechanisms involved in the systemic protection against B. cinerea were evaluated using known mutants of genes involved in jasmonate (JA)/ethylene (ET)/salicylic acid (SA) signaling. Other Arabidopsis mutants, AtrhbohD-3, AtrhbohF-3, and ups1-1 were also screened to determine other parts of the mechanisms involved.
Results
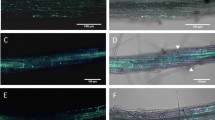
The results showed that the strain NRRL B-24137 colonized, epi- and endophytically, the roots of Arabidopsis seedlings but the strain was not a systemic colonizer during the time of the experiment. The strain NRRL B-24137 also reduced B. cinerea symptoms and the protection was linked to known mechanisms of induced systemic resistance (ISR; JA/ET signaling), as well as to functionality of AtrbohF oxidase and of UPS1. Crosstalk between ET/JA and SA signaling could also be involved.
Conclusions
The isolate NRRL B-24137, after colonizing the root systems of A. thaliana, induces an ISR against B. cinerea, which is JA/ET dependent, but could also require SA crosstalk and protection could also require NAPDH oxidases and UPS1 functionalities.





Similar content being viewed by others
References
Amann RI, Binder BJ, Olson RJ, Chisholm SW, Devereux R, Stahl DA (1990) Combination of 16S rRNA-targeted oligonucleotide probes with flow cytometry for analyzing mixed microbial populations. Appl Environ Microbiol 56:1919–1925
Audenaert K, Pattery T, Cornelis P, Höfte M (2002) Induction of systemic resistance to Botrytis cinerea in tomato by Pseudomonas aeruginosa 7NSK2: role of salicylic acid, pyochelin, and pyocyanin. Mol Plant Microbe Interact 15:1147–1156
Bakker PAHM, Pieterse CMJ, Van Loon LC (2007) Induced systemic resistance by fluorescent Pseudomonas spp. Phytopathology 97:239–243
Barriuso J, Solano BR, Gutiérrez Mañero FJ (2008) Protection against pathogen and salt stress by four plant growth-promoting rhizobacteria isolated from Pinus sp. on Arabidopsis thaliana. Phytopathology 98:666–672
Barrow JR, Lucero ME, Reyes-Vera I, Havstad KM (2008) Do symbiotic microbes have a role in plant evolution, performance and response to stress? Com Integr Biol 1:69–73
Brandam C, Lebrihi A, Meyer XM, Strub C (2008) Saccharothrix algeriensis, a new antibiotic producer: investigations on its secondary metabolism. In: Industrial Biotechnology International Conferences, 8–11 June 2008, Italy.
Cao H, Bowling SA, Gordon AS, Dong X (1994) Characterization of an Arabidopsis mutant that is nonresponsive to inducers of systemic acquired resistance. Plant Cell 6:1583–1592
Cao H, Glazebrook J, Clarke JD, Volko S, Dong X (1997) The Arabidopsis NPR1 gene that controls systemic acquired resistance encodes a novel protein containing ankyrin repeats. Cell 88:57–63
Compant S, Clément C, Sessitsch A (2010a) Plant growth-promoting bacteria in the rhizo- and endosphere of plants. Their role, colonization, mechanisms involved and prospects for utilization. Soil Biol Biochem 42:669–678
Compant S, Duffy B, Nowak J, Clément C, Ait Barka E (2005) Use of plant growth-promoting bacteria for biocontrol of plant diseases: principles, mechanisms of action, and future prospects. Appl Environ Microbiol 71:4951–4959
Compant S, Reiter B, Colli-Mull JG, Gangl H, Sessitsch A (2011) Endophytes of grapevine flowers, berries and seeds: identification of cultivable bacteria, comparison with other plant parts, and visualization of niches of colonization. Microb Ecol 62:188–197
Compant S, van der Heijden M, Sessitsch A (2010b) Climate change effects on beneficial plant–microbes interactions. FEMS Microbiol Ecol 73:197–214
Conn VM, Walker AR, Franco CMM (2008) Endophytic actinobacteria induce defense pathways in Arabidopsis thaliana. Mol Plant-Microbe Interact 21:208–218
Daims H, Brühl A, Amann R, Schleifer K-H, Wagner M (1999) The domain-specific probe EUB338 is insufficient for the detection of all bacteria: development and evaluation of a more comprehensive probe set. Syst Appl Microbiol 22:434–444
De Meyer G, Capieau K, Audenaert K, Buchala A, Métraux JP, Höfte M (1999) Nanogram amounts of salicylic acid produced by the rhizobacterium Pseudomonas aeruginosa 7NSK2 activate the systemic acquired resistance pathway in bean. Mol Plant Microbe Interact 12:450–458
De Meyer G, Höfte M (1997) Salicylic acid produced by the rhizobacterium Pseudomonas aeruginosa 7NSK2 induces resistance to leaf infection by Botrytis cinerea on bean. Phytopathology 87:588–593
Denby KJ, Jason LJM, Murray SL, Last RL (2005) ups1, an Arabidopsis thaliana camalexin accumulation mutant defective in multiple defence signaling pathways. Plant J 41:673–684
Ferrari S, Galletti R, Denoux C, De Lorenzo G, Ausubel FM, Dewdney J (2007) Resistance to Botrytis cinerea induced in Arabidopsis by elicitors is independent of salicylic acid, ethylene, or jasmonate signaling but requires PHYTOALEXIN DEFICIENT3. Plant Physiol 144:367–379
Glazebrook J, Chen W, Estes B, Chang H-S, Nawrath C, Métraux J-P, Zhu T, Katagiri F (2003) Topology of the network integrating salicylate and jasmonate signal transduction derived from global expression phenotyping. Plant J 34:217–228
Hallmann J (2001) Plant interactions with endophytic bacteria. In: Jeger MJ, Spence NJ (eds) Biotic interactions in plant–pathogen associations. CABI, Wallingford, pp 87–119
Hallmann J, Berg B (2007) Spectrum and population dynamics of bacterial root endophytes. In: Schulz BJE, Boyle CJC, Sieber TN (eds) Microbial root endophytes. Springer, Berlin, pp 15–31
Hammerschmidt R (1999) Induced disease resistance: how do induced plants stop pathogens. Physiol Mol Plant Pathol 55:77–84
Hann J, Kamber M (2000) Data mining: concepts and techniques. Morgan Kaufman, San Francisco
Heck S, Grau T, Buchala AJ, Metraux JP, Nawrath C (2003) Genetic evidence that expression of NahG modifies defence pathways independent of salicylic acid biosynthesis in the Arabidopsis–Pseudomonas syringae pv. tomato interaction. Plant J 36:342–352
Kloepper JW, Ryu C-M, Zhang S (2004) Induced systemic resistance and promotion of plant growth by Bacillus spp. Phytopathology 94:1259–1266
Köberl M, Müller H, Ramadan EM, Berg G (2011) Desert farming benefits from microbial potential in arid soils and promotes diversity and plant health. PLoS ONE 6(9):e24452
Lamari L, Zitouni A, Boudjella H, Badjin B, Sabaou N, Lebrihi A, Lefebvre G, Seguin E, Tillequin F (2002a) New dithiolopyrrolones antibiotics from Saccharothrix sp. SA 233: I. Taxonomy, fermentation, isolation and biological activities. J Antibiot 55:696–701
Lamari L, Zitouni A, Dob T, Sabaou N, Lebrihi A, Germain P, Seguin E, Tillequin F (2002b) New dithiolopyrrolone antibiotics from Saccharothrix sp. SA 233. II. Physiochemical properties and structure elucidation. J Antibiot 55:702–707
Lamari L. (2006) Production de nouveaux antibiotiques du groupe des pyrrothines par une nouvelle espèce dactinomycète, Saccharothrix algeriensis. Thèse de doctorat, E.N.S. de Kouba (Algérie), pp 1–177.
Lugtenberg B, Kamilova F (2009) Plant-growth-promoting rhizobacteria. Annu Rev Microbiol 63:541–556
Marasco R, Rolli E, Ettoumi B, Vigani G, Mapelli F, Borin S, Abou-Hadid AF, El-Behairy UA, Sorlini C, Cherif A, Zocchi G, Daffonchio D (2012) A drought resistance-promoting microbiome is selected by root system under desert farming. PLOS ONE 7(10):e48479
Maurhofer M, Hase C, Meuwly P, Métraux JP, Défago G (1994) Induction of systemic resistance of tobacco to tobacco necrosis virus by the root-colonizing Pseudomonas fluorescens strain CHA0: influence of the gacA gene and of pyoverdine production. Phytopathology 84:139–146
Maurhofer M, Reimmann C, Schmidli-Sacherer P, Heeb S, Haas D, Défago G (1998) Salicylic acid biosynthetic genes expressed in Pseudomonas fluorescens strain P3 improve the induction of systemic resistance in tobacco against tobacco necrosis virus. Phytopathology 88:678–684
Mitter B, Brader G, Afzal M, Compant S, Naveed M, Trognitz F, Sessitsch A (2013) Advances in elucidating plant–soil–microbe (bacteria) interactions. Adv Agron 121:381–445
Muzammil S, Compant S, Yu Z, Mathieu F, Lebrihi A (2011) Saccharothrix algeriensis NRRL B-24137: a new endophyte with high potential to protect grapevine towards Botrytis cinerea in case of high temperature conditions. In: Oeno 2011 - Actes de colloques du 9e symposium international d’oenologie de Bordeaux. Dunod (in press)
Oelmüller R, Sherameti I, Tripathi S, Varma A (2009) Piriformospora indica, a cultivable root endophyte with multiple biotechnological applications. Symbiosis 49:1–17
Pieterse CM, van der Does D, Zamioudis C, Leon-Reyes A, van Wees SC (2012) Hormonal modulation of plant immunity. Annu Rev Cell Dev Biol 28:28–33
Pieterse CMJ, Van Loon LC (1999) Salicylic acid-independent plant defence pathways. Trends Plant Sci 4:52–58
Pieterse CMJ, Van Loon LC (2004) NPR1: the spider in the web of induced resistance signaling pathways. Curr Opinion Plant Biol 7:456–464
Pieterse CMJ, Van Wees SCM, Hoffland E, Van Pelt JA, Van Loon LC (1996) Systemic resistance in Arabidopsis induced by biocontrol bacteria is independent of salicylic acid accumulation and pathogenesis-related gene expression. Plant Cell 8:1225–1237
Pieterse CMJ, Van Wees SCM, Van Pelt JA, Knoester M, Laan R, Gerrits H, Weisbeek PJ, Van Loon LC (1998) A novel signaling pathway controlling induced systemic resistance in Arabidopsis. Plant Cell 10:1571–1580
Pineda A, Zheng SJ, van Loon JJA, Pieterse CM, Dicke M (2010) Helping plants to deal with insects: the role of beneficial soil-borne microbes. Trends Plant Sci 15:507–514
Reinhold-Hurek B, Hurek T (2011) Living inside plants: bacterial endophytes. Curr Opinion Plant Biol 14(4):435–443
Roller C, Wagner M, Amann R, Ludwig W, Schleifer KH (1994) In situ probing of gram-positive bacteria with high DNA G+C content using 23S rRNA-targeted oligonucleotides. Microbiology 140:2849–2858
Rosenblueth M, Martínez-Romero E (2006) Bacterial endophytes and their interaction with hosts. Mol Plant-Microbe Interact 19:827–837
Ryu C-M, Murphy JR, Mysore K, Kloepper JW (2004) Plant growth-promoting rhizobacteria systemically protect Arabidopsis thaliana against cucumber mosaic virus by a salicylic acid and NPR-1 independent and jasmonic acid-dependent pathway. Plant J 39:381–392
Ton J, De Vos M, Robben C, Buchala AJ, Métraux J-P, Van Loon LC, Pieterse CMJ (2002) Characterisation of Arabidopsis enhanced disease susceptibility mutants that are affected in systemically induced resistance. Plant J 29:11–21
Torres MA, Dangl J (2005) Functions of the respiratory burst oxidase in biotic interactions, abiotic stress and development. Curr Opinion Plant Biol 8:397–403
van de Mortel JE, de Vos RC, Dekkers E, Pineda A, Guillod L, Bouwmeester K, van Loon JJ, Dicke M, Raaijmakers JM (2012) Metabolic and transcriptomic changes induced in Arabidopsis by the rhizobacterium Pseudomonas fluorescens SS101. Plant Physiol 160(4):2173–2188
Van der Ent S, Van Wees SCM, Pieterse CMJ (2009) Jasmonate signaling in plant interactions with resistance-inducing beneficial microbes. Phytochemistry 70:1581–1588
Van Loon LC, Bakker PAHM (2004) Signaling in rhizobacteria–plant interactions. In: De Kroon J, Visser EJW (eds) Ecological studies, vol 168, Root ecology. Springer, Berlin, pp 287–330
Van Loon LC, Bakker PAHM (2005) Induced systemic resistance as a mechanism of disease suppression by rhizobacteria. In: Siddiqui ZA (ed) PGPR: biocontrol and biofertilization. Springer, Dordrecht, pp 39–66
Van Loon LC, Bakker PAHM, Pieterse CMJ (1998) Systemic resistance induced by rhizosphere bacteria. Annu Rev Phytopathol 36:453–483
van Loon LC, Van Strien EA (1999) The families of pathogenesis-related proteins, their activities, and comparative analysis of PR-1 type proteins. Physiol Mol Plant Pathol 55:85–97
Van Wees SCM, Van der Ent S, Pieterse CMJ (2008) Plant immune responses triggered by beneficial microbes. Curr Opinion Plant Biol 11:443–448
Ward ER, Uknes SJ, Williams SC, Dincher SS, Wiederhold DL, Alexander DC, Ahl-Goy P, Métraux J-P, Ryals JA (1991) Coordinate gene activity in response to agents that induce systemic acquired resistance. Plant Cell 3:1085–1094
Yekkour A, Sabaou N, Zitouni A, Errakhi R, Mathieu F, Lebrihi A (2012) Characterization and antagonistic properties of Streptomyces strains isolated from Saharan soils, and evaluation of their ability to control seedling blight of barley caused by Fusarium culmorum. Lett Appl Microbiol 55:427–435
Zamioudis C, Pieterse CMJ (2012) Modulation of host immunity by beneficial microbes. Mol Plant-Microbe Interact 25:139–150
Zhou N, Tootle TL, Tsui F, Klessig DF, Glazebrook J (1998) PAD4 functions upstream from salicylic acid to control defense responses in Arabidopsis. Plant Cell 10:1021–1030
Zitouni A (1995) Les genres Nocardiopsis et Saccharothrix (Actinomycetales) dans les sols sahariens: taxonomie numérique, extraction, purification et caractérisation de quelques antibiotiques synthétisés. Magister de microbiologie, E.N.S. de Kouba, Algeria
Zitouni A, Boudjella H, Lamari L, Badji B, Mathieu F, Lebrihi A, Sabaou N (2005) Nocardiopsis and Saccharothrix genera in Saharan soils in Algeria: isolation, biological activities and partial characterisation of antibiotics. Res Microbiol 156:984–993
Zitouni A, Lamari L, Boudjella H, Badji B, Sabaou N, Gaouar A, Mathieu F, Lebrihi A, Labeda DP (2004) Saccharothrix algeriensis sp. nov., isolated from Saharan soil. Int J Syst Evol Microbiol 54:1377–1381
Acknowledgments
This work was supported by a grant from HEC Pakistan. Additional funding came from CNRS. We would like to thank Ms. Helen Smith (AIT, Wien, Austria) and Dr. Brian Murphy (Trinity College Dublin, Ireland) for the proof reading of the manuscript. Also, special thanks to Livio Antonielli (AIT, Tulln, Austria) for his comments. Additional thanks to the reviewers for their valuable comments.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible Editor: Jesus Mercado-Blanco.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Table Suppl 1
Results of detection of NRRL B-24137 in WT Col N39005 on and inside roots and leaves with or without Botrytis cinerea. + presence, ND not detectable. (PPT 80 kb)
Rights and permissions
About this article
Cite this article
Muzammil, S., Graillon, C., Saria, R. et al. The Saharan isolate Saccharothrix algeriensis NRRL B-24137 induces systemic resistance in Arabidopsis thaliana seedlings against Botrytis cinerea . Plant Soil 374, 423–434 (2014). https://doi.org/10.1007/s11104-013-1864-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-013-1864-0