Abstract
Aims
We used a Procrustean superimposition approach associated with regression analysis to test hypotheses regarding the relationship between plant communities and distinct below-ground compartments—soil chemistry (SC) and soil microbial activity (SMA). Additionally, we evaluated litter chemical quality as an interface between the above and below-ground compartments.
Methods
Plant community, and soil chemical and biochemical data from three post-mining degraded sites under reclamation and from one nearby forest site in the Brazilian Amazon Basin were analyzed.
Results
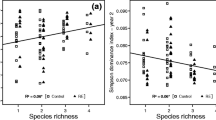
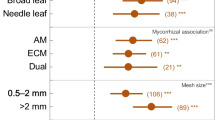
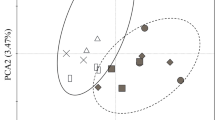
All studied sites presented distinct plant community, litter quality, SC and SMA. Plant community consistently affected the below-ground variation in both SC and SMA compartments. The influence of litter quality was greater in the plant community versus SMA relationship than in the plant community versus SC. Nevertheless, the SC affected significantly the SMA, but without influence of litter quality.
Conclusions
Differently from previous studies, our findings suggest that plant community and soil chemistry can affect the soil microbial activity independently. Specifically for our study area, these results point to a rupture of the ‘in nested’ structure of the causal relationship between changes in vegetation, changes in the chemical litter quality, changes in the SC and the response of SMA.






Similar content being viewed by others
References
Anderson JD, Ingram LJ, Stahl PD (2008) Influence of reclamation management practices on microbial biomass carbon and soil organic carbon accumulation in semiarid mined lands of Wyoming. Appl Soil Ecol 40:387–397. doi:10.1016/j.apsoil.2008.06.008
Arato HD, Martins SV, Ferrari SHS (2003) Litterfall and litter decomposition in an agroforestry system established for reclamation of a degraded area in Viçosa- MG. Rev Arv 27:715–721. doi:10.1590/S0100
Bardgett RD, Wardle DA (2010) Aboveground-belowground linkages. Biotic interactions, ecosystem processes, and global change. Oxford Univeristy Press, Oxford, p 301
Bartlett RJ, Ross DS (1988) Colorimetric determination of oxidizable carbon in acid soil solutions. Soil Sci Soc Am J 52:1191–1192
Berg G, Smalla K (2009) Plant species and soil type cooperatively shape structure and function of microbial communities in the rhizosphere. FEMS Microb Ecol 68:1–13. doi:10.1111/j.1574-6941.2009.00654.x
Chaer GM, Fernandes MF, Myrold DD, Bottomley PJ (2009) Comparative resistance and resilience of soil microbial communities and enzyme activities in adjacent native forest and agricultural soils. Microb Ecol 58:414–424. doi:10.1007/s00248-009-9508-x
Chen D, Zhou L, Wu J, Hsu J, Lin Y, Fu S (2012) Tree girdling affects the soil microbial community by modifying resource availability in two subtropical plantations. Appl Soil Ecol 53:108–115. doi:10.1016/j.apsoil.2011.10.014
Cleary DFR, Smalla K, Mendonça-Hagler LCS, Gomes NCM (2012) Assessment of variation in bacterial composition among microhabitats in a mangrove environment using DGGE fingerprints and barcode pyrosequencing. PlosOne e29380 doi:10.1371/journal.pone.0029380
Eivazi F, Tabatabai MA (1988) Glucosidases and galactosidases in soils. Soil Biol Biochem 20:601–606
Embrapa—Centro Nacional de Pesquisa de Solos (1997) Manual de métodos de análise de solo. Centro Nacional de Pesquisa de Solos, Rio de Janeiro
Embrapa—Centro Nacional de Pesquisa Agropecuária (1999) Manual de análises químicas em solos, plantas e fertilizantes. Embrapa Solos/Embrapa informática/Embrapa Comunicação para Transferência de Tecnologia, Brasília
García-Palacios P, Maestre FT, Gallardo A (2010) Soil nutrient heterogeneity modulates ecosystem responses to changes in the identity and richnnes of plant functional groups. J Ecol 99:551–562. doi:10.1111/j.1365-2745.2010.01765.x
Green VS, Stott DE, Diack M (2006) Assay for fluorescein diacetate hydrolytic activity: optimization for soil samples. Soil Biol Biochem 38:693–701. doi:10.1016/j.soilbio.2005.06.020
Grime JP (1998) Benefits of plant diversity to ecosystems: immediate, filter and founder effects. J Ecol 86:902–910. doi:10.1046/j.1365-2745.1998.00306.x
Harris JA (2003) Measurements of the soil microbial community for estimating the success of restoration. Euro J Soil Sci 54:801–808. doi:10.1046/j.1351-0754.2003.0559.x
Högberg MN, Högbeg P, Myrold DD (2007) Is microbial community composition in boreal forest determined by pH, C to N ratio, the trees or all three? Oecologia 150:590–601. doi:10.1007/s00442-006-0562-5
Hooper DU, Vitousek PM (1997) The effects of plant composition and diversity on ecosystem processes. Science 227:1302–1305. doi:10.1126/science.277.5330.1302
Ingram LJ, Schuman GE, Stahl PD, Spackman LK (2005) Microbial respiration and organic carbon cycling recovery in reclaimed soils. Soil Sci Soc Am J 69:1737–1745. doi:10.2136/sssaj2004.0371
Jesus EC, Marsh TL, Tiedje JM, Moreira FMS (2009) Changes in land use alter the structure of bacterial communities in Western Amazon soils. Inter Soc Microb Ecol 3:1004–1011. doi:10.1038/ismej.2009.98
Lamb EG, Kennedy N, Siciliano SF (2011) Effects of plant species richness and evenness on soil microbial community diversity and function. Plant Soil 338:483–495. doi:10.1007/s11104-010-0560-6
Legendre P, Gallagher ED (2001) Ecologically meaningful transformations for ordination of species data. Oecologia 129:271–280. doi:10.1007/s004420100716
Legendre P, Legendre L (1998) Numerical Ecology, 2nd edn. Elsevier, Amsterdam
McLaren JR, Turkington R (2010) Ecosystem properties determined by plant functional group identify. J Ecol 98:459–469. doi:10.1111/j.1365-2745.2009.01630.x
Meisner A, de Boer W, Cornelissen JHC, van der Putten WH (2012) Reciprocal effects of litter from exotic and congeneric native plant species via soil nutrients. PlosOne e31596 doi:10.1371/journal.pone.0031596
Mendiburu F (2012) Agricolae: Statistical Procedures for Agricultural Research. R package version 1.1-1. Published at http://R-project.org
Merilä P, Lämsa MM, Stark S, Spetz P, Vierikko K, Derome J, Fritze H (2010) Soil organic matter quality as a link between microbial community structure and vegetation composition along a successional gradient in a boreal forest. Appl Soil Ecol 46:259–267. doi:10.1016/j.apsoil.2010.08.003
Mitchell RJ, Campbell CD, Chapman SJ, Cameron CM (2010a) The ecological engineering impact of single tree species on the soil microbial community. J Ecol 98:50–61. doi:10.1111/j.1365-2745.2009.01601.x
Mitchell RJ, Hester AJ, Campbell CD, Chapman SJ, Cameron CM, Hewison RL, Potts JM (2010b) Is vegetation composition or soil chemistry the best predictor of soil microbial community? Plant Soil 333:417–430. doi:10.1007/s11104-010-0357-7
Mitchell RJ, Hester AJ, Campbell CD, Chapman SJ, Cameron CM, Hewison RL, Potts JM (2012) Explaining the variation in the soil microbial community: do vegetation composition and soil chemistry explain the same or different parts of the microbial variation? Plant Soil 351:355–362. doi:10.1007/s11104-011-0968-7
Mukhopadhyay S, Joy VC (2010) Influence of leaf litter on microbial functions and nutrient status of soil: ecological suitability of Forest trees for afforestation in tropical laterite wastelands. Soil Biol Biochem 42:2306–2315. doi:10.1016/j.soilbio.2010.09.007
Muller-Dombois D, Ellemberg H (1974) Aims and methods of vegetation ecology. Wiley, New York
Oksanen J, Kindt R, Legendre P, O’Hara RB (2007) Vegan: community ecology package version 1.17-11. Available: http://cran-r-project.org
Orwin KH, Buckland SM, Johnson D, Turner BL, Smart S, Oakley S, Bardgett RD (2010) Linkages of plants traits to soil properties and the functioning of temperate grassland. J Ecol 98:1074–1083. doi:10.1111/j.1365-2745.2010.01679.x
Peres-Neto PR, Jackson DA (2001) How well do multivariate data sets match? The advantages of a Procrustean superimposition approach over the Mantel test. Oecologia 129:169–178. doi:10.1007/s004420100720
Powers JS, Peréz-Aviles D (2012) Edaphic factors are a more important control on surface fine roots than stand age in secondary tropical dry forests. Biotropica 0:1–9. doi:10.1111/j.1744-7429.2012.00881.x
Purin S, Rillig M (2007) The arbuscular mycorrhizal fungal protein glomalin: limitations, progress, and a new hypothesis for its function. Pedobiologia 51:123–130. doi:10.1016/j.pedobi.2007.03.002
Rousk J, Brookes PC, Bääth E (2009) Contrasting soil pH effects on fungal and bacterial growth suggest functional redundancy in carbon mineralization. Appl Environ Microbiol 75:1589–1596. doi:10.1128/AEM.02775-08
Scherer-Lorenzen M (2008) Functional diversity affects decomposition processes in experimental grassland. F Ecol 22:547–555. doi:10.1111/j.1365-2435.2008.01389.x
Scnhürer J, Rosswall T (1982) Fluorescein diacetate hydrolysis as a measurement of total microbial activity in soil and litter. Appl Environ Microb 43:1256–1261
Silva DJ (1990) Análise de Alimentos. Métodos Químicos e Biológicos. Viçosa Federal University Press, Viçosa
Silva EE, Azevedo PHS, De-Polli H (2007). Determinação da respiração basal (RBS) e quociente metabólico do solo (qCO2). Comunicado Técnico. Seropédica: Embrapa
Singh BK, Nunan N, Ridgway KP, Mcnicol J, Young JPW, Daniell TJ, Prosser JI, Millard P (2008) Relationship between assemblages of mycorrhizal fungi and bacteria on grass roots. Environ Microbiol 10:534–542. doi:10.1111/j.1462-2920.2007.01474.x
Sinsabaugh RL, Klug MJ, Collins HP, Yeager PE, Peterson SO (1999) Characterizing soil microbial communities. In: Robertson GP, Coleman DC, Bledsoe CS, Sollins P (eds) Standard soil methods for long-term ecological research. Oxford University Press, New York, pp 318–348
Tabatabai MA (1994) Soil enzymes. In: Chair RWW, Angle S, Bottomley P, Benzdicek D, Smith S, Tabatabai A, Wollum A (eds) Methods of soil analysis: microbiological and biochemical properties. Soil Science Society of America, Madison, pp 775–834
Vance ED, Brookes PC, Jenkinson DS (1987) An extraction method for measuring soil microbial biomass C. Soil Biol Biochem 19:703–707. doi:10.1016/0038-0717(87)90052-6
Van der Heijden MGA, Bardgett RD, van Straalen NM (2008) The unseen majority: soil microbes as drivers of plant diversity and productivity in terrestrial ecosystems. Ecol Lett 11:296–310. doi:10.1111/j.1461-0248.2007.01139.x
van der Heijden MGA, Klironomos J, Ursic M, Moutoglis P, Streitwolf-Engel R, Boller T, Wiemken A, Snaders IR (1998) Mycorrhizal fungi determines plant biodiversity, ecosystem variability and productivity. Nature 396:69–72. doi:10.1038/23932
Van Soest PJ (1967) Development of a comprehensive system of feed analyses and its application to forage. J Anim Sci 26:119–128
Wardle DA (1992) A comparative assessment of the factors which influence microbial biomass carbon and nitrogen levels in soil. Biol Rev Camb Philos Soc 67:321–358. doi:10.1111/j.1469-185X.1992.tb00728.x
Williamson WM, Wardle DA, Yeates GW (2005) Changes in soil microbial and nematode communities during ecosystem decline across a long-term chronosequence. Soil Biol Biochem 37:1289–1301. doi:10.1016/j.soilbio.2004.11.025
Wright SF, Upadhyaya A (1998) A survey of soils for aggregate stability and glomalina, a glycoprotein produced by hyphae of arbuscular mycorrhizal fungi. Plant Soil 198:97–107. doi:10.1023/A:1004347701584
Zak DR, Holmes WE, White DC, Peacok AD, Tilman D (2003) Plant diversity, soil microbial communities, and ecosystem function: are there any links? Ecology 84:2042–2052. doi:10.1890/02-0433
Zhang FP, Li CF, Tong LG, Yue LX, Lid P, Ciren YJ, Cao CG (2010) Response of microbial characteristics to heave metal pollution in mining soil in central Tibet, China. Appl Soil Ecol 45:144–151. doi:10.1016/j.apsoil.2010.03.006
Acknowledgments
We thank the Graduate Program in Agronomy and Soil Science of Federal Rural University of Rio de Janeiro, the Brazilian Federal Agency for Graduate Education (CAPES), the Inter-American Institute for Global Change Research (IAI)—US National Science Foundation (Grant GEO-04523250) and Vale S/A for providing structural support and sponsoring the study. We are also thankful to Dr. Luiz Eduardo Dias and Igor Assis of Federal University of Viçosa for helping with the chemical analyses, and to the staff of the Environmental Management Department of Vale S/A (GABAM), Tarcísio Majevski, Paulo Rocha, and João Carlos Henriques, for providing information on the study area. The authors also thank the valuable suggestions of the anonymous reviewers of this manuscript.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible Editor: Gerlinde De Deyn.
Rights and permissions
About this article
Cite this article
Lisboa, F.J.G., Chaer, G.M., Jesus, E.C. et al. The influence of litter quality on the relationship between vegetation and below-ground compartments: a Procrustean approach. Plant Soil 367, 551–562 (2013). https://doi.org/10.1007/s11104-012-1491-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-012-1491-1