Abstract
Background and aims
Cytokinins are known to negatively regulate plant root development and phosphorus (P) starvation responses. In this study, two Brassica napus root mutants insensitive to exogenous cytokinin, prl1 (a mutant with an elongated primary root) and lrn1 (a mutant with more lateral roots), were identified and used to evaluate P efficiency, alongside wild type (WT) plants.
Methods
Solution and pot culture, RNA isolation, RT-PCR were used.
Results
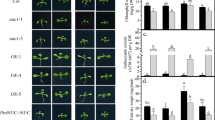
lrn1 produced the highest shoot dry weight (SDW) and root dry weight (RDW) among the three genotypes at both low P (LP) and high P (HP) conditions. prl1 had higher SDW at both P conditions, and higher RDW at LP, in contrast to WT plants. lrn1 not only accumulated more P from the culture medium but utilized it effectively in shoot growth, whilst prl1 just showed higher P use efficiency compared to WT at LP. Trans-zeatin riboside (tZR) and isopentenyl adenosine (iPA) concentration in lrn1 roots were both significantly lower than that in WT roots at both P conditions. Root iPA concentration in prl1 was lower than that in WT under both P conditions, however, root tZR concentration was greater in prl1 than WT under LP condition. Transcription of the P starvation induced genes BnSPX3;1 and BnSPX3;2 were up-regulated in the roots of mutants under both P conditions.
Conclusions
These results suggested that an improved root system might be associated with the reduced cytokinin concentration and lead to a significant increase in acquisition and utilization of P nutrient for lrn1.





Similar content being viewed by others
References
Ai PH, Sun SB, Zhao JN, Fan XR, Xin WJ, Guo Q, Yu L, Shen Q, Wu P, Miller AJ, Xu GH (2009) Two rice phosphate transporters, OsPht1;2 and OsPht1;6, have different functions and kinetic properties in uptake and translocation. Plant J 57:798–809. doi:10.1111/j.1365-313X.2008.03726.x
Bari R, Datt PB, Stitt M, Scheible WR (2006) PHO2, microRNA399, and PHR1 define a phosphate-signaling pathway in plants. Plant Physiol 141:988–999. doi:10.1104/pp.106.079707
Bates TR, Lynch JP (2000) The efficiency of Arabidopsis thaliana (Brassicaceae) root hairs in phosphorus acquisition. Am J Bot 87:964–970. doi:10.2307/2656995
Davies PJ (1995) Plant hormones: physiology, biochemistry, and molecular biology. Kluwer, Dordrecht, pp 1–38
Dello IR, Linhares FS, Scacchi E, Casamitjana-Martinez E, Heidstra R, Costantino P, Sabatini S (2007) Cytokinins determine Arabidopsis root-meristem size by controlling cell differentiation. Curr Biol 17:678–682. doi:10.1016/j.cub.2007.02.047
Duan HY, Shi L, Ye XS, Wang YH, Xu FS (2009) Identification of phosphorous efficient germplasm in oilseed rape. J Plant Nut 32:1148–1163. doi:10.1080/01904160902943171
Duan K, Yi K, Dang L, Huang H, Wu W, Wu P (2008) Characterization of a sub-family of Arabidopsis genes with the SPX domain reveals their diverse functions in plant tolerance to phosphorus starvation. Plant J 54:965–975. doi:10.1111/j.1365-313X.2008.03460.x
Duff SMG, Sarath G, Plaxton WC (1994) The role of acid-phosphatases in plant phosphorus metabolism. Physiol Plantarum 90:791–800. doi:10.1111/j.1399-3054.1994.tb02539.x
Fang ZY, Shao C, Meng YJ, Wu P, Chen M (2009) Phosphate signaling in Arabidopsis and Oryza sativa. Plant Sci 176:170–180. doi:10.1016/j.plantsci.2008.09.007
Franco-Zorrilla JM, Martin AC, Solano R, Rubio V, Leyva A, Paz-Ares J (2002) Mutations at CRE1 impair cytokinin-induced repression of phosphate starvation responses in Arabidopsis. Plant J 32:353–360. doi:10.1046/j.1365-313X.2002.01431.x
George TS, Gregory PJ, Hocking P, Richardson AE (2008) Variation in root-associated phosphatase activities in wheat contributes to the utilization of organic P substrates in vitro, but does not explain differences in the P-nutrition of plants when grown in soils. Environ Exp Bot 64:239–249. doi:10.1016/j.envexpbot.2008.05.002
Gonzalez-Rizzo S, Crespi M, Frugier F (2006) The Medicago truncatula CRE1 cytokinin receptor regulates lateral root development and early symbiotic interaction with Sinorhizobium meliloti. Plant Cell 18:2680–2693. doi:10.1105/tpc.106.043778
Hammond JP, Broadley MR, White PJ, King GJ, Bowen HC, Hayden R, Meacham MC, Mead A, Overs T, Spracklen WP, Greenwood DJ (2009) Shoot yield drives phosphorus use efficiency in Brassica oleracea and correlates with root architecture traits. J Exp Bot 60:1953–1968. doi:10.1093/jxb/erp083
Hermans C, Hammond JP, White PJ, Verbruggen N (2006) How do plants respond to nutrient shortage by biomass allocation? Trends Plant Sci 11:610–617. doi:10.1016/j.tplants.2006.10.007
Holford ICR (1997) Soil phoshorus its measurement, and its uptake by plants. Aust J Soil Res 35:227–239. doi:10.1071/S96047
Horgan JM, Wareing PF (1980) Cytokinins and the growth responses of seedlings of Betula pendula Roth, and Acer pseudoplatanus L. to nitrogen and phosphorus deficiency. J Exp Bot 31:525–532. doi:10.1093/jxb/31.2.525
Huang CY, Shirley N, Genc Y, Shi BJ, Langridge P (2011) Phosphate utilization efficiency correlates with expression of low-affinity phosphate transporters and noncoding RNA, IPS1 in barley. Plant Physiol 156:1217–1229. doi:10.1104/pp.111.178459
Lambers H, Shane MW, Cramer MD, Pearse SJ, Veneklaas EJ (2006) Root structure and functioning for efficient acquisition of phosphorus: matching morphological and physiological traits. Ann Bot 98:693–713. doi:10.1093/aob/mcl114
Lan P, Li W, Fischer R (2006) Arabidopsis thaliana wild type, pho1, and pho2 mutant plants show different responses to exogenous cytokinins. Plant Physiol Biochem 44:343–350. doi:10.1016/j.plaphy.2006.06.016
Laplaze L, Benkova E, Casimiro I, Maes L, Vanneste S, Swarup R, Weijers D, Calvo V, Parizot B, Herrera-Rodriguez MB, Offringa R, Graham N, Doumas P, Friml J, Bogusz D, Beeckman T, Bennett M (2007) Cytokinins act directly on lateral root founder cells to inhibit root initiation. Plant Cell 19:3889–3900. doi:10.1105/tpc.107.055863
Li JZ, Xie Y, Dai AY, Liu LF, Li ZC (2009) Root and shoot traits responses to phosphorus deficiency and QTL analysis at seedling stage using introgression lines of rice. J Genet Genomics 36:173–183. doi:10.1016/S1673-8527(08)60104-6
Li X, Mo XR, Shou HX, Wu P (2006) Cytokinin-mediated cell cycling arrest of pericycle founder cells in lateral root initiation of Arabidopsis. Plant Cell Physiol 47:1112–1123. doi:10.1093/pcp/pcj082
Lohar DP, Schaff JE, Laskey JG, Kieber JJ, Bilyeu KD, Bird DM (2004) Cytokinins play opposite roles in lateral root formation, and nematode and Rhizobial symbioses. Plant J 38:203–214. doi:10.1111/j.1365-313X.2004.02038.x
Lopez-Bucio J, Hernandez-Abreu E, Sanchez-Calderon L, Nieto-Jacobo MF, Simpson J, Herrera-Estrella L (2002) Phosphate availability alters architecture and causes changes in hormone sensitivity in the Arabidopsis root system. Plant Physiol 129:244–256. doi:10.1104/pp.010934
Martin AC, del Pozo JC, Iglesias J, Rubio V, Solano R, de La Pena A, Leyva A, Paz-Ares J (2000) Influence of cytokinins on the expression of phosphate starvation responsive genes in Arabidopsis. Plant J 24:559–567. doi:10.1046/j.1365-313X.2002.01431.x
Pant BD, Musialak-Lange M, Nuc P, May P, Buhtz A, Kehr J, Walther D, Scheible WR (2009) Identification of nutrient-responsive Arabidopsis and rapeseed microRNAs by comprehensive real-time polymerase chain reaction profiling and small RNA sequencing. Plant Physiol 150:1541–1555. doi:10.1104/pp.109.139139
Riefler M, Novak O, Strnad M, Schmulling T (2006) Arabidopsis cytokinin receptor mutants reveal functions in shoot growth, leaf senescence, seed size, germination, root development, and cytokinin metabolism. Plant Cell 18:40–54. doi:10.1105/tpc.105.037796
Rubio V, Bustos R, Irigoyen ML, Cardona-Lopez X, Rojas-Triana M, Paz-Ares J (2009) Plant hormones and nutrient signaling. Plant Mol Biol 69:361–373. doi:10.1007/s11103-008-9380-y
Sakakibara H (2006) Cytokinins: activity, biosynthesis, and translocation. Annu Rev Plant Biol 57:431–449. doi:10.1146/annurev.arplant.57.032905.105231
Salama AM, Wareing PF (1979) Effects of mineral nutrition on endogenous cytokinins in plants of sunflower (Helianthus annuus L.). J Exp Bot 30:971–981. doi:10.1093/jxb/30.5.971
Schmitz RY, Skoog F (1972) Cytokinins: synthesis and biological activity of geometric and position isomers of zeatin. Plant Physiol 50:702–705. doi:10.1104/pp.50.6.702
Shin H, Shin HS, Dewbre GR, Harrison MJ (2004) Phosphate transport in Arabidopsis: Pht1;1 and Pht1;4 play a major role in phosphate acquisition from both low- and high-phosphate environments. Plant J 39:629–642. doi:10.1111/j.1365-313X.2004.02161.x
Shin H, Shin HS, Chen RJ, Harrison MJ (2006) Loss of At4 function impacts phosphate distribution between the roots and the shoots during phosphate starvation. Plant J 45:712–726. doi:10.1111/j.1365-313X.2005.02629.x
Spiess LD (1975) Comparative activity of isomers of zeatin and ribosyl-zeatin on Funaria hygrometrica. Plant Physiol 55:583–585. doi:10.1104/pp.55.3.583
Strnad M (1997) The aromatic cytokinins. Physiol Plant 101:674–688. doi:10.1111/j.1399-3054.1997.tb01052.x
Su JY, Xiao YM, Li M, Liu QY, Li B, Tong YP, Jia JZ, Li ZS (2006) Mapping QTLs for phosphorus-deficiency tolerance at wheat seedling stage. Plant Soil 281:25–36. doi:10.1007/s11104-005-3771-5
Su JY, Qi Z, Li HW, Li B, Jing RL, Tong YP, Li ZS (2009) Detection of QTLs for phosphorus use efficiency in relation to agronomic performance of wheat grown under phosphorus sufficient and limited conditions. Plant Sci 176:824–836. doi:10.1016/j.plantsci.2009.03.006
Wang N, Wang YJ, Tian F, King GJ, Zhang CY, Long Y, Shi L, Meng JL (2008) A functional genomics resource for Brassica napus: development of an EMS mutagenized population and discovery of FAE1 point mutations by TILLING. New Phytol 180:751–765. doi:10.1111/j.1469-8137.2008.02619.x
Wang XM, Yi KK, Tao Y, Wang F, Wu ZC, Jiang DA, Chen X, Zhu LH, Wu P (2006) Cytokinin represses phosphate-starvation response through increasing of intracellular phosphate level. Plant Cell Environ 29:1924–1935. doi:10.1111/j.1365-3040.2006.01568.x
Wang XR, Shen JB, Liao H (2010) Acquisition or utilization, which is more critical for enhancing phosphorus efficiency in modern crops? Plant Sci 179:302–306. doi:10.1016/j.plantsci.2010.06.007
Wang ZY, Hu H, Huang HJ, Duan K, Wu ZC, Wu P (2009) Regulation of OsSPX1 and OsSPX3 on expression of OsSPX domain genes and Pi-starvation signaling in rice. J Integr Plant Biol 51:663–674. doi:10.1111/j.1744-7909.2009.00834.x
Wasaki J, Rothe A, Kania A, Neumann G, Romheld V, Shinano T, Osaki M, Kandeler E (2005) Root exudation, phosphorus acquisition, and microbial diversity in the rhizosphere of white lupine as affected by phosphorus supply and atmospheric carbon dioxide concentration. J Environ Qual 34:2157–2166. doi:10.2134/jeq2004.0423
Werner T, Schmulling T (2009) Cytokinin action in plant development. Curr Opin Plant Biol 12:527–538. doi:10.1016/j.pbi.2009.07.002
Werner T, Motyka V, Laucou V, Smets R, Van Onckelen H, Schmulling T (2003) Cytokinin-deficient transgenic Arabidopsis plants show multiple developmental alterations indicating opposite functions of cytokinins in the regulation of shoot and root meristem activity. Plant Cell 15:2532–2550. doi:10.1105/tpc.014928
Werner T, Nehnevajova E, Kollmer I, Novak O, Strnad M, Kramer U, Schmulling T (2010) Root-specific reduction of cytokinin causes enhanced root growth, drought tolerance, and leaf mineral enrichment in Arabidopsis and tobacco. Plant Cell 22:3905–3920. doi:10.1105/tpc.109.072694
Westerman RL (ed) (1990) Soil testing and plant analysis, 3rd edn. Soil Science Society of America, Madison
White PJ, Hammond JP (2008) Phosphorus nutrition of terrestrial plants. In: The Ecophysiology of Plant-Phosphorus Interactions, vol 7. plant ecophysiology. pp 51–81
Williamson LC, Ribrioux SP, Fitter AH, Leyser HM (2001) Phosphate availability regulates root system architecture in Arabidopsis. Plant Physiol 126:875–882. doi:10.1104/pp.126.2.875
Wissuwa M, Yano M, Ae N (1998) Mapping of QTLs for phosphorus-deficiency tolerance in rice. Theor Appl Genet 97:777–783. doi:10.1007/s001220050955
Wu SR, Cheng WF, Zhou X (1988) Enzyme linked immunosorbent assay for endogenous plant hormones. Plant Physiol Commun 5:53–57. doi:CNKI:SUN:ZWSL.0.1988-05-020
Yang GZ, Ding GD, Shi L, Cai HM, Xu FS (2011) Characterization of phosphorus starvation-induced gene BnSPX3 in Brassica napus. Plant Soil 350:339–351. doi:10.1007/s11104-011-0913-9
Yang M, Ding GD, Shi L, Feng J, Xu FS, Meng JL (2010) Quantitative trait loci for root morphology in response to low phosphorus stress in Brassica napus. Theor Appl Genet 121:181–193. doi:10.1007/s00122-010-1301-1
Yang XJ, Finnegan PM (2010) Regulation of phosphate starvation responses in higher plants. Ann Bot 105:513–526. doi:10.1093/aob/mcq015
Zhang HW, Huang Y, Ye XS, Xu FS (2010) Analysis of the contribution of acid phosphatase to P efficiency in Brassica napus under low phosphorus conditions. Sci China Life Sci 53:709–717. doi:10.1007/s11427-010-4008-2
Acknowledgments
We greatly acknowledge the efforts of Mayank Gautam (Huazhong Agricultural University), Annaliese Mason (The University of Western Australia) and Philip J. White (Scottish Crop Research Institute) for critical reading and editing the English version of the manuscript. We sincerely thank funding agencies, National Basic Research and Development Program of China (2011CB100301), National Nature Science Foundation of China (30600373) and Natural Science Funds for Distinguished Young Scholar in Hubei Province of China (2011CDA090) for their financial support.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible Editor: Hans Lambers.
Rights and permissions
About this article
Cite this article
Shi, T., Zhao, D., Li, D. et al. Brassica napus root mutants insensitive to exogenous cytokinin show phosphorus efficiency. Plant Soil 358, 61–74 (2012). https://doi.org/10.1007/s11104-012-1219-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-012-1219-2