Abstract
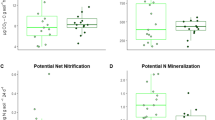
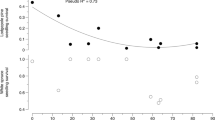
Plant–soil feedbacks have been observed in many forest communities, but the role of the mycorrhizal community in perpetuating feedback loops is still poorly understood. Mycorrhizal community composition is closely linked to soil properties and host plant composition, which highlights their potential importance in plant–soil–fungus loops. Eastern hemlock (hemlock; Tsuga canadensis) seedlings were grown in soil bioassays in growth chambers and transplanted under closed forest canopy to examine the effect of hardwood and hemlock forest soil on seedling growth, survival, and ectomycorrhizal fungi (EMF) colonization. Seedlings propagated in hemlock forest soil had greater height growth compared with sterile control soil and achieved greater mycorrhizal colonization than seedlings grown in hardwood forest soils after 9 months in a growth chamber. Outplanted seedlings grown in hemlock communities achieved significantly greater increment growth than those seedlings grown in hardwood communities (mean height difference (95% CI) = 0.39 cm (0.14–0.63 cm)), although final survival and EMF colonization was similar between forest types. EMF diversity (Shannon-Wiener index (SE) = 1.88 (0.28) and 1.23 (0.44) for hardwood and hemlock, respectively) and community assemblage (Jaccard index (SE) = 19.0% (4%)) differed between the two forest communities. EMF community assemblage was associated with both the forest type (i.e. plant community/microsite effects) and initial soil type (i.e. soil characteristics/resistant inoculum). The results support previously observed positive feedbacks between conspecifics under hemlock forest communities and provides evidence for the role of the EMF community within this feedback loop. Alternatively, the reduced growth of hemlocks under hardwoods may be attributed to the different EMF community associated with that forest.




Similar content being viewed by others
References
Ashkannejhad S, Horton TR (2006) Ectomycorrhizal ecology under primary succession on coastal sand dunes: interactions involving Pinus contorta, suilloid fungi and deer. New Phytol 169:345–354
Baayen RH (2010) languageR: Data sets and functions with “Analyzing linguistic data: a practical introduction to statistics”. R package version 1.0
Bates D (2005) Fitting linear mixed models in R. R News 5:27
Bigelow SW, Canham CD (2002) Community organization of tree species along soil gradients in a northeastern USA forest. J Ecol 90:188–200
Booth MG (2004) Mycorrhizal networks mediate overstory-understorey competition in a temperate forest. Ecol Lett 7:538–546
Catovsky S, Bazzaz FA (2000) The role of resource interaction and seedling regeneration in maintaining a positive feedback in hemlock stands. J Ecol 88:100–112
Chao A (1987) Estimating the population size for capture-recapture data with unequal catchability. Biometrics 43:783–791
Colwell RK, Coddington JA (1994) Estimating terrestrial biodiversity through extrapolation. Philos T Roy Soc B 345:101–118
Davis MB, Calcote RR, Sugita S, Takahara H (1998) Patchy invasion and the origin of a hemlock-hardwoods forest mosaic. Ecology 79:2641–2659
R Development Core Team (2010) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria, ISBN 3-900051-07-0. Available from. http://www.R-project.org
Dickie IA, Koide RT, Steiner KC (2002) Influences of established trees on mycorrhizas, nutrition, and growth of Quercus rubra seedlings. Ecol Mono 72:505–521
Dickie IA, Schnitzer SA, Reich PB, Hobbie SE (2005) Spatially disjunct effects of co-occurring competition and facilitation. Ecol Lett 8:1191–1200
Ehrenfeld JG, Ravit B, Elgersma K (2005) Feedback in the plant–soil system. Ann Rev Env Res 30:75–115
Finzi AC, Canham CD, Van Breemen N (1998a) Canopy tree–soil interactions within temperate forests: species effects on soil carbon and nitrogen. Ecol Appl 8:440–446
Finzi AC, Canham CD, Van Breemen N (1998b) Canopy tree–soil interactions within temperate forests: species effects on pH and cations. Ecol Appl 8:447–454
Frazer GW, Canham CD, Lertzman CD (2000) Gap light analyzer (GLA), Version 2.0 Technological Tools Bulletin of the Ecological Society of America, pp 191–197
Frelich LE, Calcote RR, Davis MB, Pastor J (1993) Patch formation and maintenance in an old-growth hemlock-hardwood forest. Ecology 74:513–527
Gardes M, Bruns TD (1996) ITS-RFLP matching for the identification of fungi. In: Walker J (ed) Methods in molecular biology, vol. 50: species diagnostic protocols: PCR and other nucleic acid methods, vol 50. Humana, Totowa, pp 177–186
Godman RM, Lancaster K (1990) Tsuga canadensis (L.) Carr. Eastern hemlock. In: Bruns RM, Honkala BH (eds) Silvics of North America: 1. Conifers. U.S.D.A., Forest Service Agricultural Handbook 654:604−612
Haskins KE, Gehring CA (2005) Evidence for mutualist limitation: the impacts of conspecific density on the mycorrhizal inoculum potential of woodland soils. Oecologia 145:123–131
Horton TR, Bruns TD, Parker VT (1999) Ectomycorrhizal fungi associated with Arctostaphylos contribute to Pseudotsuga menziesii establishment. Can J Bot 77:93–102
Horton TR, Molina R, Hood K (2005) Douglas-fir ectomycorrhizae in 40- and 400-year-old stands: mycobiont availability to late successional western hemlock. Mycorrhiza 15:393–403
Ishida TA, Nara K, Hogetsu T (2007) Host effects on ectomycorrhizal fungal communities: insights from eight host species in mixed conifer-broadleaf forests. New Phytol 174:430–440
Johnson NC, Graham JH, Smith FA (1997) Functioning of mycorrhizal associations along the mutualism–parasitism continuum. New Phytol 135:575–585
Jonsson L, Dahlberg A, Nilsson MC, Karen O, Zackrisson O (1999) Continuity of ectomycorrhizal fungi in self-regenerating boreal Pinus sylvestris forests studied by comparing mycobiont diversity on seedlings and mature trees. New Phytol 142:151–162
Kennedy PG, Izzo AD, Bruns TD (2003) High potential for common mycorrhizal networks between understory and canopy trees in a mixed evergreen forest. J Ecol 91:1071–1080
Kobe RK, Pacala SW, Silander JA, Canham CD (1995) Juvenile tree survivorship as a component of shade tolerance. Ecol Appl 5:517–532
Lewin DC (1974) The vegetation of the ravines of the southern Finger Lakes, New York region. Am Midl Nat 91:315–342
Molina R, Massicotte H, Trappe JM (1992) Specificity phenomena in mycorrhizal symbioses: community–ecological consequences and practical implications. In: Allen MF (ed) Mycorrhizal functioning an integrative plant–fungal process. Chapman and Hall, New York, pp 357–423
Nara K (2005) Ectomycorrhizal networks and seedling establishment during early primary succession. New Phytol 169:169–178
Nara K, Hogetsu T (2004) Ectomycorrhizal fungi on established shrubs facilitate subsequent seedling establishment of successional plant species. Ecology 85:1700–1707
Nobis M, Hunziker U (2005) Automatic thresholding for hemispherical canopy-photographs based on edge detection. Agr For Meteorol 128:243–250
Oksanen J, Kindt R, Legendre P, O’Hara RB, Stevens MHH (2010) Vegan: community ecology package. R package version 1.17-2. http://r-forge.rproject.org/projects/vegan/
Perry DA, Amaranthus MP, Borcher JG, Borcher SL, Brainerd RE (1989) Bootstrapping in ecosystems. BioScience 39:230–237
Phillips RP, Fahey TJ (2006) Tree species and mycorrhizal associations influence the magnitude of rhizosphere effects. Ecology 87:1302–1313
Pinheiro JC, Bates DM (2000) Mixed-effects models in S and S-PLUS. Springer-Verlag, New York
Pinheiro J, Bates D, DebRoy S, Sarkar D, the R Core team (2008) nlme: linear and nonlinear mixed effects models. R package version 3:1–90
Reynolds HL, Packer A, Bever JD, Clay K (2003) Grassroots ecology plant–microbe–soil interactions as drivers of plant community structure and dynamics. Ecology 84:2281–2291
Rogers RS (1978) Forests dominated by hemlock (Tsuga canadensis): distribution as related to site and postsettlement history. Can J Bot 56:843–854
Schwarz PA, Fahey TJ, McCulloch CE (2003) Factors controlling spatial variation of tree species abundance in a forested landscape. Ecology 84:1862–1878
Templer PH, Dawson TE (2004) Nitrogen uptake by four tree species of the Catskill Mountains, New York: implications for forest dynamics. Plant Soil 262:251–261
Turner GD, Lewis JD, Mates-Muchin JT, Schuster WF, Watt L (2009) Light availability and soil source influence ectomycorrhizal fungal communities on oak seedlings grown in oak- and hemlock-associated soils. Can J For Res 39:1247–1258
Van Breemen N, Finzi AC, Canham CD (1997) Canopy tree-soil interactions within tem-perate forests: effects of soil texture and elemental composition on species distributions. Can J For Res 27:1110–1116
van der Heijden MGA, Klironomos JN, Ursic M, Moutogolis P, Strietwolf-Engel R, Boller T, Wiemken A, Sanders IR (1998) Mycorrhizal fungal diversity determines plant biodiversity, ecosystem variability and productivity. Nature 396:69–72
Weber A, Karst J, Gilbert B, Kimmins JP (2005) Thuja plicata exclusion in ectomycorrhizal-dominated forests: testing the role inoculums potential of arbuscular mycorrhizal fungi. Oecologia 143:148–156
Weckel M, Tirpak JM, Nagy C, Christie R (2006) Structural and compositional change in old-growth eastern hemlock Tsuga canadensis forest, 1965–2004. For Ecol Manag 231:114–118
Woods KD (1984) Patterns of tree replacement: canopy effects on understory patter in hemlock-northern hardwood forests. Vegetatio 56:87–107
Zhang Y, Chen JM, Miller JM (2005) Determining digital hemispherical photograph exposure for leaf area index estimation. Agr For Meteorol 133:166–181
Acknowledgements
This work was supported by the Mianus River Gorge Preserve, a TA fellowship from SUNY-ESF, Department of Environmental and Forest Biology, and the SUNY-ESF Zabel award. Thanks to all those who helped with field work and analyses, especially Joseph Vineis. We would also like to thank the Mianus River Gorge Preserve for providing a study site and assistance in the field especially Mark Weckel, Chris Nagy, and Rod Christie. We would also like to thank the anonymous reviewers and handling editor, Dr. Thom Kuyper, for their constructive feedback and revisions which have helped to clarify and improve the paper.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible Editor: Thom W. Kuyper.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplementary 1
(DOC 337 kb)
Supplementary 2
(DOC 40 kb)
Supplementary 3
(DOC 103 kb)
Rights and permissions
About this article
Cite this article
O’Brien, M.J., Gomola, C.E. & Horton, T.R. The effect of forest soil and community composition on ectomycorrhizal colonization and seedling growth. Plant Soil 341, 321–331 (2011). https://doi.org/10.1007/s11104-010-0646-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-010-0646-1