Abstract
The mixture of other broadleaf species into beech forests in Central Europe leads to an increase of tree species diversity, which may alter soil biochemical processes. This study was aimed at 1) assessing differences in gross rates of soil N cycling among deciduous stands of different beech (Fagus sylvatica L.) abundance in a limestone area, 2) analyzing the relationships between gross rates of soil N cycling and forest stand N cycling, and 3) quantifying N2O emission and determining its relationship with gross rates of soil N cycling. We used 15N pool dilution techniques for soil N transformation measurement and chamber method for N2O flux measurement. Gross rates of mineral N production in the 0–5 cm mineral soil increased across stands of decreasing beech abundance and increasing soil clay content. These rates were correlated with microbial biomass which, in turn, was influenced by substrate quantity, quality and soil fertility. Leaf litter-N, C:N ratio and base saturation in the mineral soil increased with decreasing beech abundance. Soil mineral N production and assimilation by microbes were tightly coupled, resulting in low N2O emissions. Annual N2O emissions were largely contributed by the freeze-thaw event emissions, which were correlated with the amount of soil microbial biomass. Our results suggest that soil N availability may increase through the mixture of broadleaf species into beech forests.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Natural forest vegetation in Central Europe is unique due to the widespread occurrence of near-monospecific beech forests in which this single species occupies 80–100% of the canopy area. In limestone areas of Central Europe, the use and management of beech forests often resulted in an mixture of different proportions of other broadleaf species and an increase of tree species diversity. Changes in tree species or even the mixture of species can have a pronounced influence on various chemical, physical, and biological soil properties due to species-specific differences in nutrient uptake, litter chemistry, root activity, canopy interception and growth (Alriksson and Eriksson 1998; Binkley and Giardina 1998). The composition of the forest canopy was shown to influence the understory vegetation (Mölder et al. 2008), soil nutrient status (Dijkstra 2003; Guckland et al. 2009), mineralization processes (Raulund-Rasmussen and Vejre 1995; Son and Lee 1997), soil acidity (Binkley and Valentine 1991; Reich et al. 2005), composition and activity of soil fauna and microflora (Saetre et al. 1999; Neirynck et al. 2000; Cesarz et al. 2007), and soil structure (Graham et al. 1995).
Soil nitrogen dynamics are directly affected by litter quality (e.g. Taylor et al. 1991; Prescott 2002; Inagaki et al. 2004; Miyamoto and Hiura 2008) and soil conditions (Bengtsson et al. 2003; Booth et al. 2005; Kooijman et al. 2008). Distinctive differences were found between conifers and hardwood species with regard to their effects on stocks, distribution and mineralization rates of soil N (Jerabkova et al. 2006; Joshi et al. 2006; Inagaki et al. 2004). In addition, N2O emissions were significantly higher in deciduous than in coniferous forests (Ambus et al. 2006; Butterbach-Bahl et al. 2002). Even within deciduous forests, differences in broadleaf species affect soil N dynamics through their differences in litter chemistry and their effects on soil chemistry. The dominance of broadleaf species in hemlock-hardwood stands was shown to increase the rate of net mineralization and nitrification as a result of improved litter quality (Ferrari 1999). Species-related differences in litterfall fluxes of elements (Ca, Mg, K, P and N) and in soil acidification were observed under canopies of different deciduous species (Nordén 1994a, b). Soil biology, especially earthworm biomass, was also influenced by deciduous tree species (Neirynck et al. 2000; Cesarz et al. 2007). In general, beech litter was found to decompose more slowly than the litter of other Central European deciduous tree species (Wise and Schäfer 1994).
From our previous study in a broadleaf mixed forest with beech, ash (Fraxinus excelsior L.), lime (Tilia cordata Mill. and/or T. platyphyllos Scop.), hornbeam (Carpinus betulus L.), and maple (Acer pseudoplatanus L. and/or A. platanoides L.), we found that surface soil acidity, C and N accumulation in the humus layer, and leaf litter C:N ratio increased with increasing abundance of beech (Guckland et al. 2009). Differences in the redistribution of nutrients and alkalinity via leaf litter were identified as important factors that contributed to this beech effect. These results suggest that the abundance of beech might also affect processes of soil N transformation in deciduous mixed forests and thus in N availability to plants and microbes. In the present study, we report on the soil N dynamics in this broadleaf mixed forest. Three stands with different beech abundance were studied. We hypothesized that soil N transformation rates and thus N2O losses increase with decreasing beech abundance. Our objectives were i) to assess differences in gross rates of soil N cycling among stands of different beech abundance, ii) to analyze the relationships between gross rates of soil N cycling and forest stand N cycling, and iii) to quantify N2O emission and assess its relationship with gross rates of soil N cycling. To our knowledge, this study is the first to investigate differences in soil N cycling across a range of beech abundance in a European mixed deciduous forest.
Materials and methods
Site description
The study was conducted in mature stands of deciduous forest in the Hainich National Park, Thuringia, Germany. The site has an elevation of 350 m a.s.l.. The mean annual temperature is 7.5°C and the mean annual precipitation is 670 mm. The geological substrate is Triassic limestone covered with 60–70 cm loess. The soil type is a Stagnic Luvisol (FAO (1998) classification) with some of the characteristics given in Table 1. The soil exhibits stagnic properties during winter and spring but is quite dry during summer. The forest has a history of at least 200 years. This has not been managed since 1990; before that time it had been used for military training since the 1960s. In 1997, it became a national park. Different forest ownerships have generated a small-scale stand mosaic of species-poor, beech-dominated forest patches and stands with up to 14 deciduous tree species per hectare that are all growing under similar climatic conditions on the same geological substrate (limestone covered by loess). Clay content in the upper 30 cm of the loess layer varied between 14 and 45%. Our study is a part of a long-term project on biogeochemical cycles and biotic interactions in stands with decreasing abundance of beech and conversely with increasing tree species diversity. Three stands were selected with decreasing beech abundance (expressed as percentage of total tree basal area, Table 1): stand A is dominated by beech (89% of the tree basal area), stand B is a three-species stand with beech (59%), ash (13%), and lime (19%) as predominant species, and stand C is covered with beech (41%), ash (31%), lime (15%), hornbeam (4%) and maple (9%) as dominant species. Soil clay content was higher in the mixed stands of deciduous tree species than in the beech-dominated stand (Table 1). All stands grow on nearly flat terrain (slope ≤ 3%).
Soil and litter sampling
In each stand, three transects were randomly selected and on each transect two plots (5 × 5 m each) were established, totalling to 6 plots per stand. Measurements of soil N cycling, N2O emissions and soil properties were conducted on these plots. The distance between plots in one stand ranged from 10 to 50 m. Gross and net rates of N transformation were determined in November 2006 and April 2007 using intact soil cores from the upper 5 cm of the mineral soil. Five soil cores (diameter of 8 cm, height of 5 cm) were taken with stainless-steel cylinders on each plot, with approximately 1 cm distance between cores. The cores were transported to the laboratory and stored at 4°C overnight prior to measurements the following day. Additional soil cores from 0 to 5 cm and 5 to 10 cm were taken at each plot for general physical and chemical characteristics. Leaf litter was collected starting from September 2005 to December 2005 at monthly intervals, described in detail by Guckland et al. (2009). Five litter collectors (surface area of 0.29 m2) were randomly distributed along each of the three transects per plot. In addition, we sampled 12 points within a 12 × 12 m grid per stand using a circular frame (sampled surface of 300 cm2) for measurements of total N and organic C in the organic layer with a frame.
Analysis of leaf litter, organic layer and mineral soil
Leaf litter dry matter was determined gravimetrically after drying to constant mass at 60°C. Leaf litter from all litter collectors of the same transect was combined resulting in three pooled samples per stand. These pooled samples were ground and analyzed for total C and N using CNS Elemental Analyzer (Elementar Vario EL, Hanau, Germany) and base cations (Ca and Mg) by pressure digestion in concentrated HNO3 (König and Fortmann 1996) followed by analysis of the digests using Inductively Coupled Plasma-Atomic Emission Spectrometer (ICP-AES; Spectro Analytical Instruments, Kleve, Germany).
For the organic layer and mineral soil, total organic C and N from ground samples were determined as mentioned above. Soil pH was measured in a suspension with distilled H2O (5 g of soil to 15 mL of H2O). Cation exchange capacity (CEC) of the mineral soil was determined by percolating 2-mm sieved soil samples with 100 ml of 1 M NH4Cl for 4 h (König and Fortmann 1996) and measuring cations in percolates using ICP-AES. Base saturation was calculated as percentage base cations (Na, K, Ca and Mg) of the CEC. Soil texture was determined using the sieving and pipette method (Schlichting et al. 1995). Soil bulk density was determined by soil core method.
Gross N transformation rates, microbial biomass, and net N transformation rates
We used the 15N pool dilution technique and calculation procedures as described in details by Davidson et al. (1991) and Hart et al. (1994). Two intact cores of each plot were injected with (15NH4)2SO4 solution (for gross N mineralization and NH +4 consumption rates) and another two cores were injected with K15NO3 solution (for gross nitrification and NO −3 consumption rates). Each soil core received five 1-mL injections, containing 25 µg N mL−1 with 95% 15N enrichment. One core of each labeled pair was broken up immediately, mixed in a plastic bag, and a subsample was extracted with 0.5 mol L−1 K2SO4 solution (1:5 dry soil mass to solution ratio) ten minutes after 15N injection (T0). Mineral N extraction was done by shaking the samples for 1 hour and filtering them through K2SO4-prewashed filter papers. The T0 cores were used to correct for the reactions that occur immediately after addition of 15NH +4 and 15NO −3 . The other core of the 15NH +4 -labeled pair was incubated for 1 day at 10°C (T1). For the 15NO −3 -labelled cores, a test conducted prior to measurement showed no detectable change in the NO −3 concentration and 15NO −3 after 1 day of incubation, and hence we incubated these cores for 2 days at 10°C (T1). The T1 cores were then extracted with 0.5 mol L−1 K2SO4 as described above. Microbial assimilation of NH +4 was calculated as the difference between gross NH +4 consumption and gross nitrification rates (Davidson et al. 1991). Microbial assimilation of NO −3 was determined by the appearance of 15N in the CHCl3-labile microbial biomass, using the nonlinear model described by Davidson et al. (1991). About 25 g soil of the T1 15NO −3 -labeled samples were fumigated with CHCl3 for 5 days and then extracted with 0.5 mol L−1 K2SO4 as described above.
Part of the extracts was used for analysis of NH +4 and NO −3 concentrations using continuous flow injection colorimetry (Cenco/Skalar Instruments, Breda, Netherlands). NH +4 was determined by Berthelot reaction method (Skalar Method 155–000) and NO −3 by copper-cadmium reduction method (NH4Cl buffer but without ethylenediamine tetraacetic acid; Skalar Method 461–000). The rest of the extracts was use for 15N analysis by diffusion of NH +4 (from the 15NH +4 -labeled cores) and of NO −3 (from the 15NO −3 -labeled cores). For the fumigated T1 15NO −3 -labeled samples, part of the extract was used for persulfate digestion for determination of 15N enrichment in extractable organic N pool (Corre et al. 2007), needed for the calculation of microbial assimilation of NO −3 . The same diffusion procedure and blank correction were followed as described in our earlier works (Corre et al. 2003; Corre and Lamersdorf 2004; Corre et al. 2007). 15N was analyzed using isotope ratio mass spectrometry (Finigan MAT, Bremen, Germany).
Microbial biomass C and N were determined from the T1 CHCl3-fumigated and the corresponding unfumigated samples. Organic C content of the K2SO4 extracts was analyzed by UV-enhanced persulfate oxidation using a Dohrmann DC-80 carbon analyzer with an infrared detector (Rosemount Analytical Division, Santa Clara, California, USA). The organic N content of the extracts was determined by persulfate digestion (Corre et al. 2007), followed by colorimetric analysis of NO −3 (as above). Microbial biomass C and N were calculated as the difference in extractable organic C and persulfate-N between the fumigated and unfumigated soils divided by k C = 0.45 and k N = 0.68 for 5-day fumigated samples (Brookes et al. 1985).
The remaining one soil core was used to estimate net N mineralization and net nitrification rates. The soil in the core was cut vertically into two parts. One part was removed from the core (T0) and the half that remained in the core was incubated for 14 days at 10°C (T1). The T0 and T1 soil samples were extracted with 0.5 mol L−1 K2SO4 solution and the extracts were analyzed for NH +4 and NO −3 contents as described above. Net N mineralization and nitrification rates were calculated as the difference between T1-and T0-NH +4 and NO −3 concentrations, respectively.
Calculation of mean residence time
The mean residence time (MRT) specifies the average length of time an N atom resides in a given pool; a low MRT indicates a rapid turnover of the N pool. The calculation of MRT (N pool ÷ flux rate) assumes that the N pool is at steady state and that the flux is equal to the rate of input to that pool. MRT was calculated for the following N pools: a) total N in the organic layer using leaf litter-N as input flux rate, b) NH +4 and NO −3 pools in the 0-5 cm mineral soil using gross N mineralization and gross nitrification as input flux rates, respectively, and c) microbial biomass N using NH +4 + NO −3 assimilation as input flux rate.
N2O fluxes, soil temperature, moisture content, and extractable N
N2O fluxes were measured biweekly from September 2005 to September 2007. On each plot, one permanent chamber base (0.04 m2 area, 0.15 m height, and inserted into the soil to 0.10 m depth) was installed three weeks before the start of measurements. Soil N2O fluxes were measured using vented static chambers (total volume 9.25 L). The cover was kept on the chamber base for one hour during which four gas samples were taken (0, 20, 40 and 60 minutes after closure) and stored in pre-evacuated glass containers with teflon-coated stopcock. Gas samples were analyzed using a gas chromatograph (GC 6000, Carlo Erba Instruments/Thermo Fisher Scientific, Milan, Italy) equipped with an electron capture detector and an autosampler system (Loftfield et al. 1997). Gas fluxes were calculated from linear regression of concentrations versus time for each chamber, corrected with measured air temperature and air pressure (Ruser et al. 1998). Zero fluxes (no change in concentration) were included. During periods with snow cover (thickness of the snow cover was ≤10 cm) chambers were carefully installed on the base frames without removing the snow. The annual N2O losses were approximated by applying the trapezoid rule on time intervals between measured flux rates, assuming constant flux rates per day.
Simultaneous to N2O flux measurements, soil temperature (at 5 cm depth) was measured and soil samples were taken from 0–5 cm mineral soil (except from January to March 2006 when the ground was frozen) for measurements of soil moisture content and extractable N. Soil moisture content was determined gravimetrically by oven-drying at 105°C for one day and expressed as water-filled pore space (WFPS) using the measured bulk density and assuming a particle density of 2.65 g cm−3 for mineral soil. Extractable N was determined from the soils extracted with 0.5 mol L−1 K2SO4 and the extracts analyzed for NH +4 , NO −3 (as above) and total N by continuous flow injection colorimetry (UV-persulfate oxidation followed by hydrazine sulfate reduction; Skalar Method 473-000).
Statistical analyses
For soil N transformation rates, differences between the two sampling dates (November 2006 and April 2007) for each stand were tested using Paired-samples T test. If there were no significant differences between sampling dates, we used the means of both sampling dates for further analyses; if otherwise, analysis was conducted separately for each sampling date. We used Kruskal-Wallis H test with multiple comparison extension to assess differences among stands (A, B, and C) when assumptions of normality and homogeneity of variance were not met. For parameters that showed normal distribution and homogenous variance, differences among stands were evaluated using one-way analysis of variance with Tukey HSD test. Spearman rank correlation was used to test relationships among soil N cycling parameters and controlling factors. Linear regression analysis was conducted to relate gross rates of mineral N production in the soil to annual leaf litter N input and C:N ratio using the mean values per transect. All tests were considered significant at p ≤ 0.05 and were conducted with STATISTICA 7.1.
Results
Leaf litter, organic layer and mineral soil
The annual leaf litter input, concentrations and stocks of Ca, Mg and N, and leaf litter quality (C:N ratio) increased with decreasing beech abundance and increasing soil clay content. This was paralleled by decreasing mass, total N stocks and turnover time of total N pool in the organic layer (Table 2). Similarly, soil pH, base saturation (Table 1), concentrations and stocks of Ca, Mg and total N increased while C:N ratio decreased (Table 2) in the upper mineral soil with decreasing beech abundance. Clay content correlated with soil pH (r = 0.82, p < 0.01, n = 18), base saturation (r = 0.85, p < 0.01, n = 18) and total N (r = 0.87, p < 0.01, n = 18) in 0–5 cm mineral soil.
Gross rates of N transformation, N pools and mean residence time of N pools
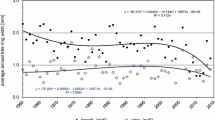
Gross rates of soil N transformation and N pools (except for NH +4 ) did not differ between sampling dates in all stands. Gross N mineralization rates in the 0–5 cm mineral soil increased with decreasing beech abundance (Fig. 1). These values result in annual gross N mineralization rates of 450, 700 and 1030 kg N ha−1 yr−1 for stands A, B and C, respectively, assuming constant rates throughout the year. Gross NH +4 assimilation rates were comparable and correlated with gross N mineralization rates (Fig. 1, Table 3). Gross nitrification rates were 5–14% of gross N mineralization rates, and also increased with decreasing beech abundance (Fig. 1). Gross nitrification rates were correlated with gross N mineralization rates, while microbial assimilation rates of NO −3 were correlated with and as high as gross nitrification rates (Fig. 1, Table 3). All N transformation processes were positively correlated with microbial N which, in turn, was positively correlated with total N, total C, pH and base saturation across stands (Table 3). Annual gross rates of N mineralization and nitrification were correlated with N input via leaf litter and leaf litter C:N ratio (Fig. 2).
Gross rates of N transformation (mg N kg−1 d−1), N pools (mg N kg−1, upper numbers in boxes) and mean residence time (d, lower numbers in boxes). For each parameter, means (±1 SE, n = 6) followed by a different letter indicate significant differences among stands (Kruskal-Wallis H-test with multiple comparison extension at p ≤ 0.05). NH +4 pool is given separately for November 2006 (first values) and April 2007 (second values) since for each stand these sampling periods differed (Paired-samples T-test at p ≤ 0.05)
Regression analysis between annual gross N mineralization and nitrification rates using the mean values for each transect (n = 9; three transects per stand) a annual leaf litter-N input (gross N mineralization = 0.03x–2.6, r2 = 0.61, p = 0.007; gross nitrification = 0.005x–0.06, r2 = 0.51, p = 0.018) and b leaf litter C:N ratio (gross N mineralization = −0.03x + 2.7, r2 = 0.56, p = 0.012; gross nitrification = −0.01x + 0.4, r2 = 0.46, p = 0.026)
NH +4 concentrations were lower in November 2006 than in April 2007, and in April 2007 NH +4 levels increased with decreasing beech abundance. NO −3 concentrations and microbial biomass N also increased with decreasing beech abundance. The NH +4 pool MRT was about 1 day, NO −3 pool MRT was 2–5 days, and microbial N MRT was 2–3 weeks; these did not differ among stands (Fig. 1).
Net rates of N transformation
The net N release differed between the two sampling dates (Fig. 3). In November 2006, negative values of net N mineralization and nitrification rates (except for stand B, although this was not different from zero; One-sample T test at p = 0.35) were observed in the 0-5 cm mineral soil, implying that microbial N assimilation exceeded mineral N production. In April 2007, we observed positive values of net rates of N transformation in all stands (Fig. 3). Net N mineralization and nitrification rates were negatively correlated with microbial C:N ratio (r = −0.47, p < 0.05, n = 36, and r = −0.42, p < 0.05, n = 36, respectively). The mean microbial C:N ratio across stands was significantly higher in November (9.2 ± 0.8) than in April (7.8 ± 0.4).
N2O flux rates and soil factors
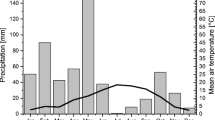
The emission rates of N2O ranged from −31.4 to 167.8 µg N2O-N m−2 h−1 but most (75%) of the measured fluxes did not differ from zero (Fig. 4a). The highest N2O emissions occurred between February and March 2006 when there was intermittent freezing and thawing of the soil. These emissions accounted 90%, 94% and 46% of the total emissions during the first year in stands A, B and C, respectively. Peak emissions during this freeze-thaw period were correlated with mean microbial biomass N (r = 0.61, p < 0.01, n = 18). Annual N2O emissions increased with decreasing beech abundance (0.11 ± 0.11, 0.19 ± 0.16, and 0.40 ± 0.23 kg N2O-N ha−1 yr-1 in stands A, B, and C respectively; p = 0.02). Soil extractable N was dominated by organic N (ranging from 8–14 mg N kg−1 across stands) and constituted less NH +4 (3.4–4.0 mg N kg−1) and NO −3 (0.9–2.0 mg N kg−1); these did not differ among stands and exhibited no seasonal variability (Fig. 4b). We found no correlations between N2O flux rates and extractable N, WFPS (Fig. 4c) or soil temperature (Fig. 4d).
Means of a N2O fluxes (±1 SE, n = 6), b extractable N, c water-filled pore space (WFPS), and d soil temperature at 0–5 cm mineral soil measured in stands with different abundance of beech during the 2-yr study period. Extractable N, WFPS and soil temperature are presented as means across the three stands (n = 18) because these parameters did not differ among stands in all sampling dates. The hatched period represents the time with a continuous snow cover (thickness of snow cover was ≤10 cm)
Discussion
Leaf litter quality and soil fertility
The increasing leaf litter N input and decreasing organic layer mass and N turnover time in the organic layer suggest faster litter decay across stands with decreasing beech abundance. The turnover time of litter N in the organic layer does not necessarily reflect litter mineralization since it does not provide information on the form and fate of C and N losses from the forest floor. The faster turnover of litter N in the organic layer of the mixed stands (stands B and C) than in the beech stand (stand A) can be explained by differences in litter quality. The higher recalcitrance of beech litter is probably due to the lower nutrient contents, higher C:N ratio (Table 2) and higher lignin:N ratio (Jacob et al. 2009; Jacob 2010) compared to litter of the other deciduous tree species. Lignin:N ratio is known to be a key factor determining dynamics of litter decomposition (Taylor et al. 1991; Scott and Binkley 1997; Goh and Totua 2004). Our findings supported other studies that showed beech litter decomposition to be slower than litter of other broadleaf tree species with the exception of oak (Wise and Schäfer 1994; Finzi et al. 1998; Neirynck et al. 2000). The increased leaf litter quality with decreasing beech abundance was also paralleled by increased soil fertility (i.e., low acidity, high base saturation, large stocks of Ca, Mg and N, and low C:N ratio) in the upper mineral soil. In a related study, we have shown that the abundance of beech and tree species-related differences in magnitude of soil-tree nutrient cycling have contributed to the differences in surface soil acidification and base cation stocks (Guckland et al. 2009). In their study, the influence of soil texture on the soil-tree nutrient cycling feedback could not be separated. The correlation of clay content with measures of soil fertility suggests that the higher clay contents in the mixed stands with high leaf litter quality may also have augmented the retention of nutrients released from litter decomposition. In view of these beech-related feedbacks on litter quality and soil biochemical conditions, we investigated how the microbially-mediated soil N transformation rates differ across sites of different beech abundance. We also like to point out that the analyzed stands differed in soil clay content and that it was not possible to clearly separate effects of clay content and beech abundance.
Gross rates of N transformation
The gross N mineralization rates measured in this study were comparable to the rates measured in other beech forests on Lithic Dystrochrept soils (Verchot et al. 2001; Geßler et al. 2005) and beech-oak forests on Dystric Cambisol soils (Bengtsson et al. 2003). Corre et al. (2003) reported lower gross NH +4 transformation rates in a Dystric Cambisol mineral soil under beech in Central Germany that has a more acidic soil and lower base cation stocks than our study site. Median gross rates of N mineralization in a mixed beech-oak stand on a Dystric Cambisol soil in Sweden were 2–6 folds higher than ours (Bengtson et al. 2006). Their large spatial variability was explained by tree species impact and variations in soil moisture and temperature.
Our study showed that the increasing gross N transformation rates with decreasing beech abundance were correlated with microbial N (Fig. 1, Table 3). In turn, microbial N was correlated with measures of soil fertility (Table 3), suggesting the indirect influence of soil biochemical conditions on gross N transformation rates. The high leaf litter quality and improved soil fertility supported larger microbial biomass in the mixed stands than in the beech stand (Fig. 1). The link between microbial biomass and gross N transformation rates is attributed to the role of microbial biomass size in driving the cycling of nutrients in the soil (Knops et al. 2002). In addition, the correlations among annual gross N mineralization and nitrification rates with annual leaf litter N input and leaf litter C:N ratio across stands (Fig. 2) also suggest the influence of substrate quantity and quality on gross rates of mineral N production. Thus, the increasing N availability (measured by gross N transformation rates) with decreasing beech abundance were influenced both by the increases in microbial biomass size and substrate availability.
Microbial assimilation of NH +4 was a larger fate of produced NH +4 than nitrification (Fig. 1). A similar NH +4 -dominated soil N cycle was also reported by Bengtsson et al. (2003) and Corre et al. (2007) for different deciduous and spruce forests. Our results showed that nitrifiers were poor competitors for NH +4 and the produced NO −3 was largely assimilated by microbial biomass. Despite longer MRT of NO −3 than of NH +4 pool (Fig. 1), NO −3 concentrations were lower than NH +4 concentrations (Figs. 1 and 4). The closely-coupled NH +4 cycling, low rates and closely-coupled NO −3 cycling, and fast turn over of microbial biomass indicated efficient retention of mineral N in the soil.
Net rates of soil N cycling did not reflect soil N availability
Net N transformation rates are the net result of the production and consumption of NH +4 and NO −3 . The net assimilation of mineral N by microbial biomass (i.e., negative rates of net N mineralization and nitrification) observed in November 2006 was possibly due to a flash of C inputs from litterfall, setting high demand for microbial assimilation of N. Support for this comes from the high microbial C:N ratio in autumn, indicating high assimilation of C, and from the negative correlations between microbial C:N ratios and net N mineralization and nitrification rates. Low or absence of net release of mineral N in forest soils following litterfall in autumn was also observed in other beech stands (Gasche et al. 2002; Geßler et al. 2005). The net production (positive rates) of mineral N in April 2007 suggests low microbial consumption relative to production of mineral N. This was supported by the higher NH +4 concentrations in spring than in autumn (Fig. 1) and the high proportion of net nitrification to net N mineralization rates in the mixed stands with high NH +4 levels (Fig. 3). Studies have shown that microbial production and consumption of mineral N do not vary commensurately across seasons, which may result in unrelated net and gross rates of N transformation processes (Davidson et al. 1992; Corre et al. 2002). Our results suggest that the net N cycling rates were influenced by microbial consumption as driven by flashes of C input and did not reflect the patterns of soil N availability across stands.
N2O emission
Annual N2O emissions from the different stands were generally low, which reflect the efficient retention of N through the closely-coupled soil N cycling in our sites. These values were comparable to the estimated N2O emission from deciduous forest soils in Germany with an average of 0.37 kg N2O-N ha−1 yr−1 (Schulte-Bisping et al. 2003). Studies have shown that emissions were smaller in coniferous stands than in broadleaf stands (Butterbach-Bahl et al. 1998; Butterbach-Bahl et al. 2002; Borken and Beese 2006), which were suggested to be caused by smaller gross rates of N mineralization and nitrification in coniferous than broadleaf stands (Ambus et al. 2006). There is little information on the influence of different deciduous tree species on N2O emission. Our results show that increasing annual N2O emissions with decreasing beech abundance were related to the winter emissions during a freeze-thaw period. These emissions increased with the amount of soil microbial biomass.
It should be stressed that the observed differences in N2O emissions among stands originated from a short pulse of activity during intermittent freezing and thawing. The contribution of freeze-thaw N2O emissions to annual N2O loss supported previous findings in agricultural and forest ecosystems that frost periods are of crucial importance in estimating annual N2O emissions from temperate terrestrial ecosystems (Flessa et al. 1995; Papen and Butterbach-Bahl 1999; Teepe et al. 2000). Our results suggest that the magnitude of freeze-thaw N2O emissions might be influenced by the microbial biomass size, which increased with decreasing abundance of beech and increasing soil fertility. This claim is supported by the results of Papen and Butterbach-Bahl (1999) who showed that increased N2O emissions from forest soils during frost periods were fuelled by easily degradable substrate derived from dead microbial biomass. Sterilization experiments showed that N2O emissions during freeze-thaw cycles originate from microbial N transformation (Röver et al. 1998), and Teepe et al. (2001) pointed out that N2O production in frozen soil layers may originate from denitrification in thin liquid water films surrounding the soil matrix.
Conclusions
This study has shown that abundance of beech in mixed deciduous forests can have a pronounced effect on the stand and soil N cycling. These effects of beech abundance have probably also been augmented by the coincidentally high clay contents in stands with low beech abundance. Across stands of decreasing beech abundance, leaf litter-N input, leaf litter quality, turnover time of total N pool in the organic layer, soil fertility and microbial biomass increased, which the latter in turn resulted to a positive feedback of N cycling in the mineral soil. Gross N transformation rates in the mineral soil increased with decreasing beech abundance. The produced NH +4 was largely assimilated by the microbial biomass. Net N cycling rates did not reflect the trends of gross N cycling rates because microbial production of mineral N did not vary commensurately with microbial consumption of mineral N across stands and seasons. Both NH +4 and NO −3 cycling were closely-coupled and resulted to an efficient retention of mineral N in the soil. This efficient N retention was reflected by the generally low N2O emissions. Annual N2O emissions were mainly contributed by the freeze-thaw event emissions, which were correlated with the amount of microbial biomass. Our results suggest that increasing the tree species diversity in beech stands growing on limestone areas by the mixture of valuable broadleaf tree species may enhance rates of N cycling in the stand and within the soil. Moreover, we like to point out that other factors, which were not addressed in this study, may influence net and gross rates of soil N turnover in stands with different deciduous tree species. Among those are for example tree species-dependent differences of rooting depth, light penetration through the canopy and timing of litterfall.
References
Alriksson A, Eriksson HM (1998) Variations in mineral nutrient and C distribution in the soil and vegetation compartments of five temperate tree species in NE Sweden. For Ecol Manage 108:261–273
Ambus P, Zechmeister-Boltenstern S, Butterbach-Bahl K (2006) Sources of nitrous oxide emitted from European forest soils. Biogeosciences 3:135–145
Bengtson P, Falkengren-Grerup U, Bengtsson G (2006) Spatial distributions of plants and gross N transformation rates in a forest soil. J Ecol 94:754–764
Bengtsson G, Bengtson P, Månsson KF (2003) Gross nitrogen mineralisation-, immobilisation-, and nitrification rates as a function of soil C/N ratio and microbial activity. Soil Biol Biochem 35:143–154
Binkley D, Valentine D (1991) Fifty-year biogeochemical effects of green ash, white pine and Norway spruce in a replicated experiment. For Ecol Manage 40:13–25
Binkley D, Giardina C (1998) Why do tree species affect soils? The warp and woof of tree-soil interactions. Biogeochemistry 42:89–106
Booth MS, Stark JM, Rastetter E (2005) Controls on nitrogen cycling in terrestrial ecosystems: a synthetic analysis of literature data. Ecol Monogr 75:139–157
Borken W, Beese F (2006) Methane and nitrous oxide fluxes of soils in pure and mixed stands of European beech and Norway spruce. Eur J Soil Sci 57:617–625
Brookes PC, Landman A, Pruden G, Jenkinson DS (1985) Chloroform fumigation and the release of soil nitrogen: a rapid direct extraction method to measure microbial biomass nitrogen in soil. Soil Biol Biochem 17:837–842
Butterbach-Bahl K, Gasche R, Huber C, Kreutzer K, Papen H (1998) Impact of N-input by wet deposition on N-trace gas fluxes and CH4-Oxidation in spruce forest ecosystems of the temperate zone in Europe. Atmos Environ 32(3):559–564
Butterbach-Bahl K, Rothe A, Papen H (2002) Effect of tree distance on N2O and CH4-fluxes from soils in temperate forest ecosystems. Plant Soil 240:91–103
Cesarz S, Fahrenholz N, Migge-Kleian S, Platner C, Schaefer M (2007) Earthworm communities in relation to three diversity in a deciduous forest. Eur J Soil Biol 43:61–67
Corre MD, Lamersdorf NP (2004) Reversal of nitrogen saturation after long-term deposition reductions: impact on soil nitrogen cycling. Ecology 85:3090–3104
Corre MD, Schnabel RR, Stout WL (2002) Spatial and seasonal variation of gross nitrogen transformations and microbial biomass in unmanaged temperate grassland. Soil Biol Biochem 34:445–457
Corre MD, Beese FO, Brumme R (2003) Soil nitrogen cycle in high nitrogen deposition forest: changes under nitrogen saturation and liming. Ecol Appl 13:287–298
Corre MD, Brumme R, Veldkamp E, Beese FO (2007) Changes in nitrogen cycling and retention processes in soils under spruce forests along a nitrogen enrichment gradient in Germany. Glob Chang Biol 13:1509–1527
Davidson EA, Hart SC, Shanks CA, Firestone MK (1991) Measuring gross nitrogen mineralization, immobilisation, and nitrification by 15N isotopic pool dilution in intact soil cores. J Soil Sci 42:335–349
Davidson EA, Hart SC, Firestone MK (1992) Internal cycling of nitrate in soils of a mature coniferous forest. Ecology 73:1148–1156
Dijkstra FA (2003) Calcium mineralization in the forest floor and surface soil beneath different tree species in the northeastern US. For Ecol Manage 175:185–194
FAO (1998) FAO, ISRIC, ISSS World Reference Base for Soil Resources. World Soil Resources Reports 84. Rome, Italy
Ferrari JB (1999) Fine-scale patterns of leaf litterfall and nitrogen cycling in an old-growth forest. Can J For Res 29:291–302
Finzi AC, Canham CD, Van Breemen N (1998) Canopy tree-soil interactions within temperate forests: species effects on soil carbon and nitrogen. Ecol Appl 8:440–446
Flessa H, Dorsch P, Beese F (1995) Seasonal Variation of N2O and CH4 fluxes in differently managed arable soils in southern Germany. J Geophys Res-Atmospheres 100:23115–23124
Gasche R, Butterbach-Bahl K, Papen H (2002) Development and application of a method for determination of net nitrification rates. Plant Soil 240:57–65
Geßler A, Jung K, Gasche R, Papen H, Heidenfelder A, Börner E, Metzler B, Augustin S, Hildebrand E, Rennenberg H (2005) Climate and forest management influence nitrogen balance of European beech forests: microbial N transformations and inorganic N net uptake capacity of mycorrhizal roots. Eur J For Res 124:95–111
Goh KM, Totua SS (2004) Effect of organic and plant residue and orchard management practice on decomposition rates of residues. Commun Soil Sci Plant Anal 35:441–460
Graham RC, Ervin JO, Wood HB (1995) Aggregate stability under oak and pine after 4 four decades of soil development. Soil Sci Soc Am J 59:1740–1744
Guckland A, Brauns M, Flessa H, Thomas F, Leuschner C (2009) Acidity, nutrient socks and organic matter content in soils of a temperate deciduous forest with different abundance of European beech (Fagus sylvatica L.). J Plant Nutr Soil Sci 172:500–511
Hart SC, Nason GE, Myrold DD, Perry DA (1994) Dynamics of gross nitrogen transformations in an old-growth forest: the carbon connection. Ecology 75:880–891
Inagaki Y, Miura S, Kohzu A (2004) Effects of forest type and stand age on litterfall quality and soil N dynamics in Shikoku district, southern Japan. For Ecol Manag 202:107–117
Jacob M (2010) Productivity and nutrient relations of trees in deciduous forests differing in tree species diversity. Dissertation, Georg-August-Universität Göttingen
Jacob M, Weland N, Leuschner C, Schaefer M, Thomas FM (2009) Nutrient release from decomposing leaf litter of temperate deciduous forest trees along a gradient of increasing tree species diversity. Soil Biol Biochem 41:2122–2130
Jerabkova L, Prescott CE, Kishchuk BE (2006) Nitrogen availability in soil and forest floor of contrasting types of boreal mixedwoods forests. Can J For Res 36:112–122
Joshi AB, Vann DR, Johnson AH (2006) Litter quality and climate decouple nitrogen mineralization and productivity in Chilean temperate rainforests. Soil Sci Soc Am J 70:153–162
Knops JMH, Bradley KL, Wedin DA (2002) Mechanism of plant species impacts on ecosystem nitrogen cycling. Ecol Lett 5:454–466
König N, Fortmann H (1996) Probenvorbereitungs-, Untersuchungs- und Element-bestimmungsmethoden des Umweltanalytiklabors der Niedersächsischen Forstlichen Versuchsanstalt und des Zentrallabor 2 des Forschungszentrums Waldökosysteme. Berichte des Forschungszentrums Waldökosysteme, Reihe B, Band 49
Kooijman AM, Kooijman-Schouten MM, Martinez-Hernandez GB (2008) Alternative strategies to sustain N-fertility in acid and calcaric beech forests: low microbial N-demand versus high biological activity. Basic Appl Ecol 9:410–421
Loftfield N, Flessa H, Augustin J, Beese F (1997) Automated gas chromatographic system for rapid analysis of the atmospheric trace gases methane, carbon dioxide, and nitrous oxide. J Environ Qual 26:560–564
Miyamoto T, Hiura T (2008) Decomposition and nitrogen release from the foliage litter of fir (Abies sachalinensis) and oak (Quercus crispula) under different forest canopies in Hokkaido. Ecol Res 23:673–680
Mölder A, Bernhardt-Römermann M, Schmidt W (2008) Zur Beziehung zwischen Baumschichtvielfalt und Produktivität der Krautschicht in Laubwäldern. Naturschutz und Biologische Vielfalt 60:139–144
Neirynck J, Mirtcheva S, Sioen G, Lust N (2000) Impact of Tilia platyphyllos Scop., Fraxinus excelsior L., Acer pseudoplatanus, Quercus robur L. and Fagus sylvatica L. on earthworm biomass and physico-chemical properties of a loamy soil. For Ecol Manage 133:275–286
Nordén U (1994a) Influence of tree species on acidification and mineral pools in deciduous forest soils of South Sweden. Water Air Soil Pollut 76:363–381
Nordén U (1994b) Leaf litterfall concentrations and fluxes in deciduous tree species. Scand J For Res 9:9–16
Papen H, Butterbach-Bahl K (1999) A 3-year continuous record of nitrogen trace gas fluxes from untreated and limed soil of a N-saturated spruce and beech forest ecosystem in Germany—1. N2O emissions. J Geophys Res-Atmospheres 104:18487–18503
Prescott CE (2002) Do rates of litter decomposition tell us anything we really need to know? For Ecol Manage 220:66–74
Raulund-Rasmussen K, Vejre H (1995) Effect of tree species and soil properties on nutrient immobilization in the forest floor. Plant Soil 168:345–352
Reich PB, Oleksyn J, Modrzynski J, Mrozinski P, Hobbie SE, Eissenstat DM, Chorover J, Chadwick O, Hale CM, Tjoelker MG (2005) Linking litter calcium, earthworm and soil properties: a common garden test with 14 tree species. Ecol Lett 8:811–818
Röver M, Heinemeyer O, Kaiser E-A (1998) Microbial induced nitrous oxide emissions from an arable soil during winter. Soil Biol Biochem 30:1859–1865
Ruser R, Flessa H, Schilling R, Steindl H, Beese F (1998) Soil compaction and fertilization effects on nitrous oxide and methane fluxes in potato fields. Soil Sci Soc Am J 62:1587–1595
Saetre P, Brandtberg P-O, Lundkvist H, Bengtsson J (1999) Soil organisms and carbon, nitrogen and phosphorous mineralization in Norway spruce and mixed Norway spruce-birch stands. Biol Fertil Soils 28:382–388
Schlichting E, Blume H-P, Stahr K (1995) Bodenkundliches Praktikum. Pareys Studientexte 81. Blackwell Wissenschafts-Verlag, Wien, pp 112–116
Schulte-Bisping H, Brumme R, Priesack E (2003) Nitrous oxide emission inventory of German forest soils. Journal of Geophysical Research-Atmospheres 108: doi:10.1029/2002JD002292
Scott NA, Binkley D (1997) Foliage litter quality and annual net N mineralization: comparison across North American forest sites. Oecologia 111:151–159
Son Y, Lee IK (1997) Soil nitrogen mineralization in adjacent stands of larch, pine and oak in central Korea. Annales des Sciences Forestieres 54:1–8
Taylor BR, Prescott CE, Parson WJF, Parkinson D (1991) Substrate control of litter decomposition in 4 Rocky-Mountain coniferous forests. Can J Bot 69:2242–2250
Teepe R, Brumme R, Beese F (2000) Nitrous oxide emission from frozen soils under agricultural, fallow and forest land. Soil Biol Biochem 32:1807–1810
Teepe R, Brumme R, Beese F (2001) Nitrous oxide emission from soil during freezing and thawing periods. Soil Biol Biochem 33:1269–1275
Verchot LV, Holmes Z, Milon L, Groffman PM, Lovett GM (2001) Gross vs net rates of N mineralization and nitrification as indicators of functional differences between forest types. Soil Biol Biochem 33:1889–1901
Wise DH, Schäfer M (1994) Decomposition of leaf-litter in a mull beech forest—comparison between canopy and herbaceous species. Pedobiologia 38:269–288
Acknowledgement
We thank the Deutsche Forschungsgemeinschaft for funding this study (DFG, Graduiertenkolleg 1086). During the supervision of this study, M. D. Corre was funded by the Robert Bosch Foundation as a junior research group leader.
Open Access
This article is distributed under the terms of the Creative Commons Attribution Noncommercial License which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Ute Skiba.
Rights and permissions
Open Access This is an open access article distributed under the terms of the Creative Commons Attribution Noncommercial License (https://creativecommons.org/licenses/by-nc/2.0), which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
About this article
Cite this article
Guckland, A., Corre, M.D. & Flessa, H. Variability of soil N cycling and N2O emission in a mixed deciduous forest with different abundance of beech. Plant Soil 336, 25–38 (2010). https://doi.org/10.1007/s11104-010-0437-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-010-0437-8