Abstract
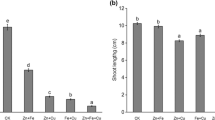
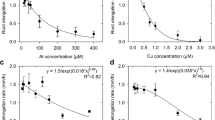
This work investigated how copper (Cu) phytotoxicity affected iron (Fe) nutrition and root elongation in hydroponically grown durum wheat (Triticum turgidum durum L., cv Acalou) in order to establish the critical level of Cu concentration in roots above which significant Cu phytotoxicity occurs. This was assessed at two levels of Fe supply (2 and 100 μM). Severe symptoms of Cu phytotoxicity were observed at Cu2+ concentration above 1 μM, i.e. interveinal chlorosis symptoms and global root growth alteration. Total root Cu concentration of about 100, 150 and 250–300 mg kg−1 corresponded to 10%, 25% and 50% reduction in root elongation, respectively. Copper and Fe concentrations as well as amounts of Cu and Fe accumulated in shoots varied inversely which suggested an antagonism between Cu and Fe leading to Fe deficiency. In addition, the root-induced release of complexing compounds increased significantly with increasing Cu concentration in nutrient solution and was positively correlated with Cu uptake without significant difference between the two Fe treatments (high and low Fe supply). This work suggests that total root Cu concentration might be a simple, sensitive indicator of Cu rhizotoxicity. It also indicated that Cu phytotoxicity which may have resulted in Fe deficiency and significant increase in root-induced release of complexing compounds (presumably phytosiderophores) was independent of the level of Fe supply provided that the threshold values of phytotoxicity were based on the free Cu-ion concentration.





Similar content being viewed by others
References
Ali NA, Bernal MP, Ater M (2002) Tolerance and bioaccumulation of copper in Phragmites australis and Zea mays. Plant Soil 239:103–111
Blamey FP (2001) The role of the root cell wall in aluminium toxicity. In: Ae, N, Okada, K, Ancha, S, Arihara, J (eds) Plant nutrition acquisition: new perspectives, workshop on new concepts of plant nutrient acquisition. Springer, New York, pp 201–226
Borkert CM, Cox FR, Tucker MR (1998) Zinc and copper toxicity in peanut, soybean, rice, and corn in soil mixtures. Comm Soil Sci Plant Anal 29:2991–3005
Chaignon V, Hinsinger P (2003) A biotest for evaluating copper bioavailability to plants in a contaminated soil. J Environ Qual 32:824–833
Chaignon V, Di Malta D, Hinsinger P (2002) Fe-deficiency increases Cu acquisition by wheat cropped in a Cu-contaminated vineyard soil. New Phytol 154:121–130
Chaignon V, Sanchez-Neira I, Herrmann P, Jaillard B, Hinsinger P (2003) Copper bioavailability and extractability as related to chemical properties of contaminated soils from a vine-growing area. Environ Pollut 123:229–238
Chaney RL (1980) Health risks associated with toxic metals in municipal sludge. In: Bitton G, Damro DL, Davidson GT, Davisdson JM (eds) Sludge: health risks of land application. Ann Arbor Science, Ann Arbor, MI, pp 59–83
Davis RD, Beckett PHT (1978) Upper critical levels of toxic elements in plants. II. Critical levels of copper in young barley, wheat, rape, lettuce and ryegrass, and of nickel and zinc in young barley and ryegrass. New Phytol 80:23–32
Degryse F, Smolders E, Parker DR (2006) Metal complexes increase uptake of Zn and Cu by plants: implications for uptake and deficiency studies in chelator-buffered solutions. Plant Soil 289:171–185
Delas J (1963) La toxicité du cuivre accumulé dans les sols. Agrochimica 7:258–288
Iwasaki K, Sakurai K, Takahashi E (1990) Copper binding by the root cell walls of Italian ryegrass and red clover. Soil Sci Plant Nutr (Tokyo) 36:431–439
Kinraide TB, Yermiyahu U (2007) A scale of metal ion binding strengths correlating with ionic charge, Pauling electronegativity, toxicity, and other physiological effects. J Inorg Chem 101:1201–1213
Kinraide TB, Pedler JF, Parker DR (2004) Relative effectiveness of calcium and magnesium in the alleviation of rhizotoxicity in wheat induced by copper, zinc, aluminum, sodium, and low pH. Plant Soil 259:201–208
Kopittke PM, Menzies NW (2006) Effect of Cu toxicity on growth of cowpea (Vigna unguiculata). Plant Soil 279:287–296
Kraemer SM, Crowley DE, Kretzschmar R (2006) Geochemical aspects of phytosiderophore-promoted iron acquisition by plants. Adv Agron 91:1–46
Lexmond TM, Van der Vorm PDJ (1981) The effect of pH on copper toxicity to hydroponically grown maize. Neth J Agric Sci 29:217–238
Llugany M, Lombini A, Poshenrieder C, Dinelli E, Barceló J (2003) Different mechanisms account for enhanced copper resistance in Silene armeria ecotypes from mine spoil and serpentine sites. Plant Soil 251:55–63
Ma JF (2005) Plant root responses to three abundant soil minerals: silicon, aluminium and iron. Crit Rev Plant Sci 24:267–281
Ma JF, Nomoto K (1993) Inhibition of mugineic acid-ferric complex uptake in barley by copper, zinc and cobalt. Physiol Plant 89:331–334
Ma JF, Ryan PR, Delhaize E (2001) Aluminium tolerance in plants and the complexing role of organic acids. Trends Plant Sci 6:273–278
MacNicol RD, Beckett PHT (1985) Critical tissue concentrations of potentially toxic elements. Plant Soil 85:107–129
Marschner H (1995) Mineral nutrition of higher plants, 2nd edn. Academic, London, UK, p 889
Marschner H, Treeby M, Römheld V (1989) Role of root-induced changes in the rhizosphere for iron acquisition in higher plants. Z Pflanzenernahr Bodenkd 152:197–204
McBride MB (2001) Cupric ion activity in peat soil as a toxicity indicator for maize. J Environ Qual 30:78–84
Meda AR, Scheuermann EB, Prechsl UE, Erenoglu B, Schaaf G, Hayen H, Weber G, von Wirén N (2007) Iron acquisition by phytosiderophores contributes to cadmium tolerance. Plant Physiol 143:1761–1773
Michaud AM, Bravin MN, Galleguillos M, Hinsinger P (2007) Copper uptake and phytotoxicity as assessed in situ for durum wheat (Triticum turgidum durum L.) cultivated in Cu-contaminated, former vineyard soils. Plant Soil 298:99–111
Murakami T, Ise K, Hayakawa M, Kamei S, Takagi S (1989) Stabilities of metal complexes of mugineic acids and their specific affinities for iron (III). Chem Lett 1989:2137–2140
Nian H, Yang ZM, Ahn SJ, Cheng ZJ, Matsumoto HA (2002) Comparative study on the aluminium- and copper-induced organic acid exudation from wheat roots. Physiol Plant 116:328–335
Parker DR (1995) Root growth analysis: an underutilised approach to understanding aluminium rhizotoxicity. Plant Soil 171:151–157
Parker DR, Chaney RL, Norvell WA (1995) Chemical equilibrium models: applications to plant nutrition research. In: Loeppert RH, Schwab AP, Goldberg S (eds) Chemical equilibrium and reaction models. Special Publication no. 42. Soil Science Society of America, Madison, WI, pp 163–200
Quartacci MF, Pinzino C, Sgherri CLM, Dalla Vecchia F, Navari-Izzo F (2000) Growth in excess copper induces changes in the lipid composition and fluidity of PSII-enriched membranes in wheat. Physiol Plant 108:87–93
Reichman SM, Parker DR (2005) Metal complexation by phytosiderophores in the rhizosphere. In: Huang PM, Gobran GR (eds) Biogeochemistry of trace elements in the rhizosphere. Elsevier, Amsterdam, pp 129–156
Reichman SM, Parker DR (2007a) Probing the effects of light and temperature on diurnal rhythms of phytosiderophore release in wheat. New Phytol 174:101–108
Reichman SM, Parker DR (2007b) Critical evaluation of three indirect assays for quantifying phytosiderophores released by the roots of Poaceae. Eur J Soil Sci 58:844–853
Reuter DJ, Robinson JB (1997) Plant analysis: an interpretation manual. CSIRO, Collingwood, Australia, p 572
Robin A, Vansuyt G, Hinsinger P, Meyer JM, Briat JF, Lemanceau P (2008) Iron dynamics in the rhizosphere: consequences for plant health and nutrition. Adv Agron 99 (in press)
Rooney CP, Zhao FJ, McGrath SP (2007) Phytotoxicity of nickel in a range of European soils: Influence of soil properties, Ni solubility and speciation. Environ Pollut 145:596–605
Schaider LA, Parker DR, Sedlak DL (2006) Uptake of EDTA-complexed Pb, Cd and Fe by solution- and sand-cultured Brassica juncea. Plant Soil 286:377–391
Sheldon AR, Menzies NW (2005) The effect of copper toxicity on the growth and root morphology of Rhodes grass (Chloris gayana Knuth.) in resin buffered solution culture. Plant Soil 278:341–349
Taylor GJ, Foy CD (1985) Differential uptake and toxicity of ionic and chelated copper in Triticum aestivum. Can J Bot 63:1271–1275
Treeby M, Marschner H, Römheld V (1989) Mobilization of iron and other micronutrient cations from a calcareous soil by plant-borne, microbial, and synthetic metal chelators. Plant Soil 114:217–226
Vindimian E (2001) Regtox. Available at: http://eric.vindimian.9online.fr/ (last access on 08/08/2007)
Wheeler DM, Power IL (1995) Comparison of plant uptake and plant toxicity of various ions in wheat. Plant Soil 172:167–173
Xuan Y, Scheuermann EB, Meda AR, Hayen H, von Wirén N, Weber G (2006) Separation and identification of phytosiderophores and their metal complexes in plants by zwitterionic hydrophilic interaction liquid chromatography coupled to electrospray ionization mass spectrometry. J Chromatography 1136:73–81
Yruela I, Pueyo JJ, Alonso PJ, Picorel R (1996) Photoinhibition of photosystem II from higher plants – effect of copper inhibition. J Biol Chem 271:27408–27415
Zhang FS, Treeby M, Römheld V, Marschner H (1991) Mobilization of iron by phytosiderophores as affected by other micronutrients. Plant Soil 130:173–178
Zhao FJ, Rooney CP, Zhang H, McGrath SP (2006) (25) Comparison of soil solution speciation and diffusive gradients in thin-films measurement as an indicator of copper bioavailability to plants. Environ Toxicol Chem733–742
Acknowledgments
We thank Didier Arnal, Nicole Balsera and Lucien Roger for technical help. We also thank Jean-François Briat, Michel Mench and Erik Smolders for their constructive comments. Financial support for this work was provided by the French ECCO-ECODYN programme.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible Editor: Henk Schat.
Rights and permissions
About this article
Cite this article
Michaud, A.M., Chappellaz, C. & Hinsinger, P. Copper phytotoxicity affects root elongation and iron nutrition in durum wheat (Triticum turgidum durum L.). Plant Soil 310, 151–165 (2008). https://doi.org/10.1007/s11104-008-9642-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-008-9642-0