Abstract
The goal of this study was to investigate whether increased nitrogen use efficiency found in Populus nigra L. (Jean Pourtet) under elevated CO2 would correlate with changes in the production of carbon-based secondary compounds (CBSCs). Using Free-Air CO2 Enrichment (FACE) technology, a poplar plantation was exposed to either ambient (about 370 µmol mol−1 CO2) or elevated (about 550 µmol mol−1 CO2) [CO2] for 5 years. After three growing seasons, the plantation was coppiced and half of the experimental plots were fertilized with nitrogen. CBSCs, total nitrogen and lignin-bound nitrogen were quantified in secondary sprouts in seasons of active growth and dormancy during 2 years after coppicing. Neither elevated CO2 nor nitrogen fertilisation alone or in combination influenced lignin concentrations in wood. Soluble phenolics and soluble proteins in wood decreased slightly in response to elevated CO2. Higher nitrogen supply stimulated formation of CBSCs and increased protein concentrations. Lignin-bound nitrogen in wood ranged from 0.37–1.01 mg N g−1 dry mass accounting for 17–26% of total nitrogen in wood, thus, forming a sizeable nitrogen fraction resistant to chemical degradation. The concentration of this nitrogen fraction was significantly decreased by elevated CO2, increased in response to nitrogen fertilisation and showed a significant CO2 × fertilisation interaction. Seasonal changes markedly affected the internal nitrogen pools. Soluble proteins in wood were 52–143% higher in the dormant than in the growth season. Positive correlations existed between the biosynthesis of proteins and CBSCs. The limited responses to elevated CO2 and nitrogen fertilisation indicate that growth and defence are well orchestrated in P. nigra and that changes in the balance of both resources—nitrogen and C—have only marginal effects on wood chemistry.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Plants produce a multitude of carbon-based secondary compounds (CBSCs), which have important functions, such as wound sealing, defence against herbivores and pathogens (Hartley and Jones 1997), control of the rates of plant decomposition (Cotrufo et al. 2005) and mediation of interactions between plants and soil biota (Harborne 1997; Northrup et al. 1998). Among these CBSCs, polyphenols derived from the phenylpropanoid pathway such as soluble phenolics, condensed tannins and lignin, are quantitatively most important accounting for about 30% of the organic carbon cycling in the terrestrial biosphere (Boudet et al. 2003).
Currently, human activities are increasing atmospheric CO2 concentrations. Forest trees respond to these changes with growth stimulation and increased nutrient uptake (Norby et al. 2005; Finzi et al. 2007). Although CBSCs constitute a significant sink for assimilated carbon, to date, there is no clear picture of how these compounds respond to elevated CO2 or nitrogen fertilisation. Studies conducted under greenhouse or growth chamber have shown that elevated CO2 increased the levels of proanthocyanidins and flavonoids (Lavola and Julkunen-Tiitto 1994) and condensed tannin (Roth and Lindroth 1994) in birch, but had no effects on condensed tannin in needles of pine (Roth and Lindroth 1994) and decreased concentrations of lignin in wood of silver birch (Kostiainen et al. 2006). Field experiments employing free air CO2 enrichment have shown that CO2 enrichment increased the levels of condensed tannin in leaves of birch but not in aspen (Lindroth et al. 2001), and had no effects on concentrations of lignin in litter of three poplar species (Cotrufo et al. 2005). In contrast to elevated CO2, nitrogen fertilisation generally decreased the levels of soluble phenolics and condensed tannins in plant tissues (e.g. Lavola and Julkunen-Tiitto 1994; Lindroth et al. 2002).
The complexity of the responses of CBSCs to elevated CO2 or nitrogen fertilisation is unexpected, based on a hypothesis concerning the carbon nutrient balance (CNB) in plants (Bryant et al. 1983). This hypothesis predicts that increased carbon supply increases the concentrations of CBSCs, while enhanced nitrogen availability will lead to increased growth and, therefore, divert carbon from the production of phenolics. The contrasting observations in tree responses to elevated CO2 and nitrogen reported in the literature demonstrate that currently our understanding of resource allocation in plants is limited.
In trees the internal balance between C and nitrogen is complex because growth depends not only on nitrogen resources directly available from soil or internal resources, but also on nitrogen resources from seasonal storage pools in bark, wood, and roots. These storage pools are filled in fall by nitrogen resorption from leaves and they are remobilised in spring to fuel formation of new leaves and xylem in deciduous tree species (Cooke and Weih 2005). Nitrogen storage may uncouple growth from nitrogen uptake and, therefore, biomass production may occur, even when external nitrogen supply is limited (Chapin et al. 1990). The C/N balance is not only affected by nitrogen but also by atmospheric CO2 concentrations (Ainsworth and Long 2005). It has been shown that growth of trees under elevated CO2 concentrations leads to increased photosynthesis and net productivity (Norby et al. 2005) and decreased nitrogen concentrations relative to biomass (Ainsworth and Long 2005; Körner et al. 2005; Norby and Iversen 2006; Finzi et al. 2007). Such shifts in C/N may also affect leaf tissue quality (Zak et al. 2000; Lindroth et al. 2002; Zak et al. 2003; Norby and Iversen 2006). Norby et al. (2001) conducted a meta-analysis of published data and found that mean reductions in nitrogen by 7.5% correlated with mean increases in lignin by 6.5% in leaf litter of plants grown under elevated compared with those grown under ambient CO2 concentrations. The depletion in nitrogen relative to tissue carbon concentrations, which was frequently found under elevated CO2 even when the plants were fertilised, has been termed ‘increased nitrogen use efficiency’ (Ainsworth and Long 2005; Finzi et al. 2007). In a previous study, we showed that a significant fraction of nitrogen in trees is bound to lignin forming so-called lignoprotein (Brinkmann et al. 2002). However, to date we have no information concerning how this fraction, which is probably of ecological significance because of its chemical stability, would respond to changes in environmental conditions.
In this study, we used 1- and 2-year-old shoots of P. nigra growing during the second rotation cycle of a poplar plantation exposed to elevated CO2 (POPFACE/EUROFACE) for a total of 5 years. Since wood accounts for 61–74% of total plant C pools (Gielen et al. 2005; Liberloo et al. 2006; Luo et al. 2006), this experiment focused on wood but included leaf and bark tissues as necessary. In a previous study, we demonstrated that mobile carbon fractions such as storage carbohydrates showed significant seasonal fluctuations but elevated CO2 or nitrogen fertilisation caused only minor changes (Luo et al. 2006). The aim of the present study was to test the following hypotheses: (1) elevated CO2 and nitrogen fertilisation have opposite effects on CBSCs with elevated CO2 increasing but nitrogen fertilisation decreasing the levels of CBSCs; (2) elevated CO2 and nitrogen fertilisation have contrasting effects on nitrogen and soluble proteins with elevated CO2 leading to decreases while nitrogen fertilisation causes increases in the levels of nitrogen and soluble proteins; and (3) increased nitrogen use efficiency under elevated CO2 correlates negatively with changes in the production of CBSCs. For this purpose, we investigated structural and defence compounds such as lignin, phenolics, and proanthocyanidins—and their seasonal dynamics in relation to the pools of nitrogen and soluble proteins in wood as well as in leaf and bark tissues. In addition, the plant internal allocation of nitrogen to proteins or structurally bound forms of nitrogen (lignoproteins) was studied in poplar grown under elevated CO2 and with or without additional nitrogen fertilisation.
Materials and methods
Site description and FACE facilities
The experimental site was located on a former agricultural land in Central Italy (Tuscania, 42°22′N, 11°48′E, altitude 150 m). In spring 1999, six 30 × 30 m experimental plots were selected after the detailed soil analysis. Three of the plots were installed with FACE facilities, whereas the other three plots were left under ambient [CO2] conditions (370 µmol mol−1 CO2). Pure CO2 (Messer Griesheim, Sulzbach) was released through laser-drilled holes in polyethylene tubes to reach the target [CO2] of 550 µmol mol−1 inside the FACE plots. CO2 enrichment was provided during daytime (6:00–18:00) from bud burst to leaf fall (from April 1st to November 15th). A meteorological station was used to control the release of CO2 at each FACE plot. The FACE system was monitored by a computer to reach the target [CO2]. A 3-year-old poplar plantation exposed to FACE with trees reaching heights of about 9 m was coppiced in winter of 2001 (Calfapietra et al. 2003). In subsequent years secondary sprouts developed from the stools and FACE was maintained. The elevated CO2 concentrations, measured at 1 min intervals, were within 20% deviation from the pre-set target concentration of 550 µmol mol−1 for 89 and 72% of the time in the years 2002 and 2003, respectively (Miglietta, CNR-IATA, Florence, Italy, unpublished data). A detailed description about the FACE facilities was given by Miglietta et al. (2001).
Plant material and nitrogen fertilisation
Each experimental plot was divided in halves by a physical resin-glass barrier (1 m deep in the soil) to provide nitrogen fertilised and non-nitrogen fertilised control subplots in the post-coppice phase from year 2002 onwards. Each subplot was further divided into three triangular sectors. Each sector was planted with one of the following three poplar species, i.e. P. alba L. (2AS-11), P. × euramericana (Dode) Guinier (I-214) and P. nigra L. (Jean Pourtet), at a planting density of 10 000 trees ha−1 (1 m × 1 m). A detailed description of the clone properties was given by Calfapietra et al. (2001). Fertilisers [Navarson 20-6-6 (N–P–K) and ammonium nitrate 34-0-0 (N–P–K) in 2002 and 2003, respectively] were applied weekly adding up to the total supply of 212 kg N ha−1 in 2002 and to 290 kg N ha−1 in 2003. The non-nitrogen fertilised control subplots received the same fertilisers as the nitrogen fertilised subplots except for nitrogen. Fertilizer solutions were applied by a hydraulic pump (Ferti-injector Amiad, IMAGO srl, Italy) connected to the irrigation system.
Sampling
This study focused on P. nigra L. (Jean Pourtet) since it produced the most aboveground woody biomass under elevated CO2 or nitrogen fertilisation among three poplar species investigated in a previous study (Liberloo et al. 2005). The plantation had been coppiced after 3 years, because the trees had reached heights of 9 m (Calfapietra et al. 2003). In the present study, we investigated secondary sprouts, which are the usual product in plantations utilised for biomass and bioenergy generation (Sims et al. 2006). Since bud break of P. nigra in Central Italy occurs at the end of March and bud set is at the beginning of October (Calfapietra et al. 2001), field samplings were conducted in the first week of September (active season) in 2002 and 2003, respectively, and in the first week of March (dormant season) in 2003 and 2004, respectively. In each field campaign, a total of 24 secondary shoots of P. nigra were harvested from the experimental plots (two CO2 levels, two nitrogen levels). During each harvest one secondary shoot was cut at stool level. To be able to develop allometric relationships between shoot biomass and total above-ground biomass (Liberloo et al. 2006), the diameters of all shoots (>0.5 cm) per stool were measured at 20 cm above stool level. In September 2002 the shoot with the second thickest diameter and in March 2003 the third thickest one were harvested and weighed. In September 2003, two different stools were chosen for the harvest of secondary shoots in September 2003 and March 2004. Stem segments (about 3 cm long) were taken at the height of 1.9 m from the sampled shoots, debarked and immediately frozen in liquid nitrogen. During the harvests in September, 3 cm2 leaf discs were sampled. All samples were wrapped in foil and plunged immediately into liquid nitrogen, transferred to the laboratory, and stored at −80°C until subsequent analyses.
Analysis of soluble- and cell wall bound-phenolics
Soluble phenolics were extracted after Pritchard et al. (1997) and determined spectrophotometrically at 765 nm (Spectrophotometer DU 640, Beckmann, München, Germany) by using the Folin-Ciocalteus method (Swain and Goldstein 1964).
To evaluate cell wall-bound phenolic compounds, the pellet left after extraction of soluble phenolics was washed twice (10 min, 4,500 g, 4°C) with 2 ml n-hexane, dried at 60°C for 2 days and subsequently weighed. This fraction represented ‘cell walls’. Cell walls were homogenised in 2 ml of 1 M NaOH. The suspension was incubated in the ultrasonic bath (60 min, 4°C) and centrifuged (10 min, 4,500 g, 4°C), and the supernatant was collected. The pellet was extracted again with 2 ml of 1 M NaOH for 10 min in dark at room temperature. The supernatants were combined and adjusted to 10 ml with distilled water and analysed by the Folin-Ciocalteus method.
Analysis of condensed tannins
Condensed tannins (proanthocyanidin) were determined after Booker et al. (1996). Fine ground materials were extracted twice with 50% methanol, the extracted solution was combined by sample and final volume was adjusted to 10 ml. Nine hundred µl of soluble extracts were mixed with 0.1 ml of 100% methanol and 6 ml of acid butanol [5% concentrated HCl in butanol (v/v)] and 0.2 ml of 2% FeNH4(SO4)2·12H2O in 2 M HCl [FeNH4(SO4)2·12H2O/HCl = w/v]. The solutions were incubated in sealed polypropylene tubes in the water bath at 95°C for 50 min and then cooled in dark. The extinction of the supernatant was determined spectrophotometrically at 550 nm (Porter et al. 1986; Booker et al. 1996). Since proanthocyanidins are commercially unavailable, the standard curve was constructed using condensed tannins purified from wood of P. nigra grown under natural conditions according to the procedures proposed by Warren et al. (1999), Booker et al. (1996) and Czochanska et al. (1980).
Analysis of lignin
Acid-insoluble (Klason) lignin was determined according to the method of Dence (1992). This analysis was carried out using wood samples of all harvest dates and leaf samples harvested in September 2003. The lignin concentrations were expressed on a dry mass basis.
Analyses of soluble protein and nitrogen pools
To analyse soluble protein concentrations, the tissues were ground to a fine powder with a ball mill cooled in liquid nitrogen (Retsch, Haan, Germany). Soluble proteins were extracted from 100 mg fine powder mixed with 2 ml 0.1 M KH2PO4/K2HPO4 buffer (pH 7.8) containing 100 mg polyvinylpolypyrrolidone (Sigma, St. Louis, USA) for 15 min on ice. The mixture was centrifuged (48400 g, 30 min, 4°C) and the supernatant was saved and subsequently gel-filtered over NAP 10 columns (Sephadex G-25 DNA Grade, Amersham Biosciences, Uppsala, Sweden) using 1 ml 0.1 M KH2PO4/K2HPO4 buffer (pH 7.0). Soluble proteins were determined in the eluate according to Bradford (1976), using bovine serum albumin (BSA, protein standard, Interchim, Montlucom, France) as the standard.
To determine nitrogen concentrations in leaves, wood and lignin, aliquots of 0.6–1.0 mg dry powder were weighed into 5 × 9 mm tin cartouches (Hekatech, Wegberg, Germany) and analysed by an Elemental Analyzer EA1108 (Carlo Erba Strumentazione, Rodano, Italy). Acetanilide (71.09% C, 10.36% N; Carlo Erba Strumentazione, Rodano, Italy) was used as the standard.
To calculate total above-ground nitrogen and lignin-bound nitrogen pools in sprouts of P. nigra, concentrations of nitrogen and lignin-bound nitrogen in wood were multiplied with aboveground woody biomass (Liberloo et al. 2006). The aboveground woody biomass in March is assumed to be equal to the one in September of previous year since the harvest in September is close to the end of the growing season and significant increases in aboveground woody biomass were unlikely (Calfapietra et al. 2001).
Statistical analysis
To determine the main effects of CO2 treatment (CO2), nitrogen fertilisation (N) and seasonal changes (Season) on all analysed parameters (except parameters in leaf tissues where only former two factors were applicable) with ANOVA, a randomised-complete-block design, with CO2, N, Season and their interactions as fixed factors and block as a random factor, was applied. All statistical tests were performed with Statgraphics (STN, St. Louis, Mo, USA) using the mixed procedure and plot as a replicate. When interactions were significant, a posteriori comparison of means was done. To reduce the chance on type I errors, all P values of these multi-comparisons were corrected by Tukey-HSD method. Data were tested for normality with the Shapiro-Wilk’s test. Differences between parameter means were considered significant when the P value of the ANOVA F test was less than 0.05.
Results
Influence of elevated CO2 and nitrogen fertilisation on carbon-based secondary compounds
Our data demonstrate that increasing age was the most important factor affecting the overall concentrations of lignin, soluble phenolics, cell wall-bound phenolics, and condensed tannins in poplar wood since the sum of CBSCs increased by about 30% from fall 2002 until March 2004 (Fig. 1; Table 1). This increase was mainly caused by increases in lignin, which constituted about 85 to 87% of total CBSCs (Fig. 1) and whose concentrations were affected by neither elevated CO2 nor nitrogen fertilisation (Table 1). However, there was a significant interaction between season and elevated CO2, which was due to the fact that elevated CO2 tended to increase in lignin in the active but to decrease it in the dormant season (Fig. 1; Table 1).
Carbon based secondary compounds in wood of P. nigra grown under either elevated (E) or ambient (A) [CO2], with (H) or without nitrogen fertilization (L). Black bar, Lignin; crossed bar, cell wall bound phenolics; white bar, soluble phenolics; hatched bar, soluble condensed tannins. The data shown indicate means ± SE (n = 6) based on dry weight. The results of the statistical analysis are shown in Table 1
In contrast to lignin, the other fractions of CBSC in woody tissue showed small but significant responses to elevated CO2 or nitrogen fertilisation (Table 1). Nitrogen fertilisation caused decreases in cell wall-bound phenolics (Fig. 1) but stimulated the concentrations of condensed tannins. This trend was more pronounced under ambient CO2 than under elevated CO2 (interaction of CO2 × N). Elevated CO2 caused a slight but significant reduction in the concentrations of soluble phenolics in wood of P. nigra (Fig. 1; Table 1).
Influence of elevated CO2 and fertilisation on soluble protein and internal nitrogen pools
We determined the concentrations of soluble proteins and nitrogen in wood and leaf tissues since both are important in this species for synthesis and seasonal storage of nitrogen-containing compounds (Fig. 2; Tables 2, 3). Elevated CO2 decreased soluble proteins and total nitrogen concentrations in leaves in September 2003, whereas nitrogen fertilisation led to increases in these compounds (Table 2).
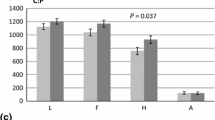
Soluble protein (a) and total nitrogen and lignin-bound nitrogen (b) in wood of P. nigra grown under elevated (E) or ambient (A) [CO2] and in the presence (H) or absence of nitrogen fertilisation (L). Bars indicate means ± SE (n = 6). Black bar, soluble protein; hatched bar, nitrogen bound to lignin; hatched + white bar, total nitrogen. The results of the statistical analysis are shown in Table 3
Our data show that the concentrations of soluble proteins in wood ranged from 7.8 to 21.6 mg g−1 depending on treatment and season (Fig. 2a). The concentrations of soluble proteins in wood showed significant seasonal variations and were markedly higher (52–143%) in the dormant season than in the growing season (Fig. 2a; Table 3). A higher nitrogen supply significantly enhanced concentrations of soluble proteins in wood of P. nigra (Fig. 2a; Table 3). Elevated CO2 resulted in decreased concentrations of soluble proteins in P. nigra wood (Fig. 2a; Table 3) and this effect was more pronounced in winter than in the growing season (Fig. 2a; Table 3).
We confirmed that exposure of P. nigra to elevated CO2 during the first 2 years after coppicing (Fig. 2b; Table 3) increased nitrogen use efficiency as reported for several poplar genotypes by Calfapietra et al. (2007). In addition to total nitrogen, we also quantified a fraction of nitrogen which has so far gained little attention but which may be especially relevant for nitrogen recycling. This nitrogen fraction is co-isolated with lignin (referred to as lignin-bound nitrogen) and, thus, is extremely resistant to chemical degradation (Brinkmann et al. 2002; Dyckmans et al. 2002). Further analysis of this recalcitrant nitrogen fraction showed that it was due to amino acids, which constituted a sizeable fraction of the lignin (Brinkmann et al. 2002). In the present study, the concentrations of lignin-bound N in wood of P. nigra ranged from 0.37 to 1.01 mg N g−1 dry mass (Fig. 2b). Elevated CO2 significantly decreased concentrations of lignin-bound nitrogen in woody stem of P. nigra, whereas nitrogen fertilisation enhanced this fraction (Fig. 2b; Table 3). These effects were more pronounced in the dormant season than in the active season (interactions CO2 × season and N × season). Similar to soluble proteins, total nitrogen and lignin-bound nitrogen concentrations showed significant seasonal variations with higher concentrations in the dormant than in the growing season (Fig. 2b; Table 3).
The amounts of total nitrogen and of lignin-bound nitrogen in wood of P. nigra in this coppice system were estimated by using total nitrogen and lignin-bound nitrogen concentrations in wood (Fig. 2b) and the aboveground standing woody biomass at the different dates (Liberloo et al. 2006), (Fig. 3; Table 4). Although the nitrogen concentrations in wood grown under elevated CO2 were lower compared to those under ambient CO2 (Fig. 2b), total nitrogen and lignin-bound nitrogen pools in aboveground woody biomass increased 8–88% under elevated CO2 (Fig. 3). This was due to the stimulation of aboveground woody biomass by elevated CO2. In the dormant season, the pool of nitrogen in aboveground woody biomass was significantly higher than in the phase of active growth because of nitrogen storage during the dormant season (Fig. 3; Table 4).
Total nitrogen and lignin-bound nitrogen pools in wood per stool of P. nigra grown under either elevated (E) or ambient (A) [CO2] and in the presence (H) or absence of nitrogen fertilization. Pool sizes were calculated by multiplying the concentrations of nitrogen and lignin-bound nitrogen (Fig. 2b) in wood with aboveground woody biomass at corresponding dates (Liberloo et al. 2006). Since the trees were grown at 1 × 1 m density, the data indicate also nitrogen content in wood per stool. The data are means ± SE (n = 6). Black bar, lignin-bound nitrogen pools; black bar + white bar, total nitrogen pools. The results of the statistical analysis are shown in Table 4
Balance between carbon-based secondary compounds and soluble proteins
To check whether there was a negative relationship between soluble proteins and CBSCs, correlation analyses were performed for soluble phenolics, cell-wall bound phenolics, soluble condensed tannins, and the sum of these carbon-based secondary compounds with soluble proteins (Fig. 4). Reference data for lignin and other phenolic compounds are included as a supplement (supplementary Table 1). Our data clearly demonstrate that positive, statistically significant (P < 0.0001) correlations were always observed between soluble proteins and soluble phenolics, cell-wall bound phenolics, condensed tannins, and the sum of these CBSCs in leaf, bark and wood tissues of P. nigra grown under either elevated or ambient [CO2] and either in the presence or absence of fertilisation.
Correlation of soluble proteins with soluble phenolics (squares), cell-wall bound phenolics (closed circles), condensed tannins (open circles) and the sum of these CBSCs (triangles) in tested tissues. Data were from leaf, bark and wood of P. nigra grown under either elevated or ambient [CO2] and either in the presence or absence of nitrogen fertilisation. All correlations were statistically significant at P < 0.0001
Discussion
CBSCs and nitrogen in plant tissues are crucial components which affect the multitrophic interactions among plants, insects, pathogens and soil biota (Hartley and Jones 1997; Mattson et al. 2004; Mattson et al. 2005). Nitrogen-enriched plant tissues are preferred by insects, pathogens and soil microbes as a high quality source of energy, while CBSCs such as phenolics, condensed tannins and lignin provide a low quality source of energy composed of recalcitrant, slowly degrading forms of C (Chapman et al. 2005). Thus, changes in foliar and wood chemistry under elevated CO2 or nitrogen fertilisation likely alter the interactions among trees, insects, pathogens and soil biota by affecting microbial substrate availability, microbial communities, rates of decomposition and C/N cycling (Gielen and Ceulemans 2001).
Since net leaf photosynthesis of P. nigra at this site was stimulated under elevated CO2 (Bernacchi et al. 2003; Tricker et al. 2005), one might have expected increases in CBSCs such as phenolic compounds and tannins, as found in birch (Mattson et al. 2005). However, this was not observed in the present study (Fig. 1; Table 1). Poplar and birch are both fast-growing, short-lived and pioneering tree species. Thus, it is expected that their responses to elevated CO2 might be similar. Whether the observed differences between birch and poplar were species-related or reflect differences in CO2-responsiveness caused by different growth conditions (soil, climate) remains currently unknown. Phenolics and condensed tannins contribute to plant defences against herbivores (Lindroth et al. 2002; Mattson et al. 2005). Whether the observed decreases in soluble phenolics caused by elevated CO2 and in condensed tannins caused by nitrogen fertilisation (Fig. 1; Table 1) point to decreases in plant resistance is not known. It is also possible that decreased tissue concentrations were due to increased fluxes through their respective biosynthetic pathways caused by growth stimulation, which, however, needs further testing. In the Swiss crane experiment, in which the crowns of mature tree are exposed to elevated CO2, lignin concentrations were decreased in leaf litter suggesting a shift of carbon from recalcitrant fractions to more labile fractions (Körner et al. 2005). While this effect was found in some species such as Carpinus and Fagus, it was not present in others such as Quercus (Körner et al. 2005). This strongly suggests that CO2 responses are species-specific.
Lignin constitutes an important fraction of woody biomass. In the present study we showed that during 2 years in the post-coppice phase, neither elevated CO2 nor nitrogen fertilisation significantly affected lignin concentrations in stem wood of P. nigra (Fig. 1; Table 1). In studies conducted mainly on leaves from greenhouse-grown plants, contrasting effects of elevated CO2 on lignin ranged from increases (Fordham et al. 1997; Penuelas and Estiarte 1998), decreases (Blaschke et al. 2002), to no changes in lignin (Norby et al. 2001; Schlimme et al. 2002; Blaschke et al. 2004). It is difficult to assess the significance of these previous studies in the context of the present work, since we do not know whether annual plants or perennials with determinate growth like beech or conifers would respond to elevated CO2 in the same manner as indeterminate growing poplar species and whether experimental enclosures or pot limitations might have had additional effects. To date only a few studies have addressed the influence of elevated CO2 on lignin under field conditions (Anttonen et al. 2001; Kaakinen et al. 2004; Kostiainen et al. 2004; Chapman et al. 2005; Kostiainen et al. 2006). Elevated CO2 had no effects on both gravimetric and acid-soluble lignin in stem wood of aspen (Kaakinen et al. 2004). Elevated CO2 also had no effects on gravimetric lignin but decreased the concentration of acid-soluble lignin in stem wood of 41-year-old Norway spruce trees (Kostiainen et al. 2004). Mattson et al. (2005) found moderate increases in lignin concentrations in stem (+4%) but strong increases in leaves (+14%) of potted birch exposed for one season to elevated CO2 in the ASPEN FACE experiment. In field-grown poplar of our study including analyses in the pre-coppicing phase (Cotrufo et al. 2005) such increases in leaf lignin were not found (Table 1). Lignification was generally higher in poplar than in birch, pointing to higher sink strength of this metabolic pathway in poplar than in birch. Our data suggest that homeostasis was maintained between carbon allocation to growth and lignin formation in P. nigra under nitrogen fertilization and elevated CO2.
It is notable that the lignin concentration of poplar stem wood increased during the whole two-year investigation period, indicating that after the first season, in which the majority of lignin was formed (Sept 2002 in Fig. 1) significant lignification still took place (+30%). Lignification continuing for several years has also been reported for wood of poplar species (Klasnja et al. 2003) and for wood of spruce (Polle et al. 1997). Since poplar wood of 3–4 years of age contains about 22% lignin (Klasnja et al. 2003; Kaakinen et al. 2004) and mature poplar wood (8–9 years old) contains 16.6–26.4% lignin (Goyal et al. 1999), it is possible that the lignin will further slightly increase in stem wood of poplar under the current experimental conditions (Fig. 1). The lignin fraction isolated with the Klason method from wood contained 60% carbon (data not shown). Using the aboveground standing woody biomass pools at the different dates (Liberloo et al. 2006), we estimated that during 2 years after coppicing the following amounts of carbon (C) were sequestered in lignin: on non-fertilised plots 766 g C m−2 under ambient CO2 and 990 g C under elevated CO2 and on fertilised plots 618 g C m−2 under ambient CO2 and 1034 g C m−2 under elevated CO2. The increases under elevated CO2 were caused by growth stimulation. In addition to carbon, nitrogen was also sequestered in this fraction (Fig. 3; Table 4) since about 17–26% of total wood-nitrogen was bound to ‘lignin’ (Fig. 2b).
True lignin of angiosperms is a heteropolymer of syringyl and coniferyl alcohol that does not contain any nitrogen. We suspect that cell wall proteins, which may have structural or enzymatic functions in wood such as extensin and peroxidases are covalently bound and embedded into the lignin matrix while the lignin polymer is being formed. In a previous study, Brinkmann et al. (2002) showed that ‘lignin’ isolated by the Klason procedure or similar protocols contained amino acids and, therefore, referred to this nitrogen-fraction as ‘ligno-protein’. Since ligno-proteins are resistant to degradation by harsh chemical treatments, they are probably also more resistant to microbial degradation than soluble proteins. Although we found that the concentrations of lignin-bound nitrogen in wood decreased under elevated CO2, the total pool of lignin-bound nitrogen in wood increased under elevated CO2 due to the stimulation of aboveground woody biomass (Fig. 3; Table 4). Based on this observation we may anticipate that nitrogen cycling will be affected under elevated CO2 or nitrogen fertilisation because a greater fraction might be retained in a resilient pool. This requires further testing.
Similar to the lignin-bound nitrogen fraction, soluble protein and total nitrogen concentrations in leaves and woody stem decreased under elevated CO2 (Fig. 2; Tables 2, 3). In the literature, decreases in nitrogen concentrations have frequently been reported under elevated CO2 (Poorter et al. 1997; Cotrufo et al. 1998; Long et al. 2004). It was assumed that nitrogen pools in tissues would be diluted if plants accumulated more soluble sugars or starch in tissues under elevated CO2 (Stitt and Krapp 1999). However, this was not the case in the present study because elevated CO2 did not induce increases in total non-structural carbohydrate concentrations (Luo et al. 2006). However, nitrogen use efficiency of the trees increased as also found in other studies (Long et al. 2004; Calfapietra et al. 2007). Lower levels of nitrogen in storage tissues such as stem wood may limit the re-growth at the beginning of the growing season when growth mainly depends on internal nitrogen resources (Millard 1996; Geßler et al. 2004; Cooke and Weih 2005). However, it is worthwhile to note that, in contrast to the nitrogen concentrations in wood tissue, the absolute amount of nitrogen pools in wood per stool in this coppice system was stimulated by elevated CO2 (Fig. 3; Table 4). This observation supports recent findings that in temperate tree species grown under field conditions increased productivity with elevated CO2 relative to ambient CO2 was sustained by higher nitrogen assimilation (Finzi et al. 2007).
Seasonal fluctuations in nitrogen-containing compounds in deciduous species are essential to support new growth at the beginning of the growing season (Sauter and van Cleve 1994; Geßler et al. 2004). Higher concentrations of soluble proteins, lignin-bound nitrogen and total nitrogen in wood in winter than in the growth season (Fig. 2, Table 3) are consistent with results of a more detailed study on poplar, where the authors showed that parenchyma cells in wood served as storage tissue for seasonally accumulated proteins (Sauter and van Cleve 1994). Increases in nitrogen in wood are caused by nitrogen translocation from leaves to the stem during leaf senescence (Norby et al. 2000; Hörtensteiner and Feller 2002). In the spring, these nitrogen reserves are transported to the growing sites to supply nitrogen for early season development of expanding buds (Bollmark et al. 1999). The seasonal nitrogen storage in wood indicates that harvests in winter remove significantly more nitrogen out of the system than in fall.
Our results suggest that poplars grown in a coppicing system maintain simultaneously high rates of biosynthesis of both CBSCs and proteins in addition to increased growth rates (Fig. 4). If increased nitrogen demand leads to increased production of phenylalanine, the joint precursor of both protein biosynthesis and the phenylpropanoid pathway, the products of both pathways are simultaneously formed at increased rates. Similar increases in CBSC concentrations have been found in nitrogen-fertilised birch trees under elevated CO2 (Mattson et al. 2004).
In summary, elevated CO2 did not increase the concentrations of phenol-based compounds; nitrogen fertilisation increased the levels of nitrogen and soluble proteins in wood. Our initial hypothesis that increased nitrogen use efficiency under elevated CO2 would negatively correlate with changes in CBSCs must be rejected. It is obvious that growth and production of defence compounds were well-balanced in the aboveground parts of P. nigra. We may still anticipate effects on biogechemical cycles if higher amounts of nitrogen are bound in a recalcitrant fraction due to general growth stimulation with elevated CO2.
References
Ainsworth EA, Long SP (2005) What have we learned from 15 years of free-air CO2 enrichment (FACE)? A meta-analytic review of the responses of photosynthesis, canopy properties and plant production to rising CO2. New Phytol 165:351–372
Anttonen S, Vapaavuori E, Kostiainen K, Isebrands JG, McDonald E, Sober J, Karnosky DF (2001) Effect of elevated CO2 and O3 on the chemical composition of wood in aspen clones: results after 3 years of exposure in the Aspen FACE project. In: Radoglou K (ed) Forest research: a challenge for an integrated European approach. International Conference Proceedings, Thessaloniki, Greece, pp 239–242
Bernacchi CJ, Calfapietra C, Davey PA, Wittig VE, Scarascia-Mugnozza G, Raines CA, Long SP (2003) Photosynthesis and stomatal conductance responses of poplars to free-air CO2 enrichment (PopFACE) during the first growth cycle and immediately following coppice. New Phytol 159:609–621
Blaschke L, Forstreuter M, Sheppard LJ, Leith IK, Murray MB, Polle A (2002) Lignification in beech (Fagus sylvatica) grown at elevated CO2 concentrations: interaction with nutrient availability and leaf maturation. Tree Physiol 22:469–477
Blaschke L, Legrand M, Mai C, Polle A (2004) Lignification and structural biomass production in tobacco with suppressed caffeic/5-hydroxy ferulic acid-O-methyl transferase activity under ambient and elevated CO2 concentrations. Physiol Plant 121:75–83
Bollmark L, Sennerby-Forsse L, Ericsson T (1999) Seasonal dynamics and effects of nitrogen supply rate on nitrogen and carbohydrate reserves in cutting-derived Salix viminalis plants. Can J Forest Res 29:85–94
Booker FL, Anttonen S, Heagle AS (1996) Catechin, proanthocyanidin and lignin concentrations of loblolly pine (Pinus taeda) needles after chronic exposure to ozone. New Phytol 132:483–492
Boudet AM, Kajita S, Grima-Pettenati J, Goffner D (2003) Lignins and lignocellulosics: a better control of synthesis for new and improved uses. Trends Plant Sci 8:576–581
Bradford MM (1976) A rapid and sensitive method for the quantification of microgram quantities of proteins utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Brinkmann K, Blaschke L, Polle A (2002) Comparison of different methods for lignin determination as a basis for calibration of near-infrared reflectance spectroscopy and implications of lignoproteins. J Chem Ecol 28:2483–2501
Bryant JP, Chapin FS III, Klein DR (1983) Carbon/nutrient balance of boreal plants in relation to vertebrate herbivory. Oikos 40:357–368
Calfapietra C, Gielen B, Sabatti M, Angelis PD, Scarascia-Mugnozza G, Ceulemans R (2001) Growth performance of Populus exposed to “Free Air Carbon Dioxide Enrichment” during the first growing season in the POPFACE experiment. Ann For Sci 58:819–828
Calfapietra C, Gielen B, Sabatti M, Angelis PD, Miglietta F, Mugnozza GS, Ceulemans R (2003) Do above-ground growth dynamics of poplar change with time under CO2 enrichment. New Phytol 160:305–318
Calfapietra C, De Angelis P, Gielen B, Lukac M, Moscatelli MC, Avino G, Lagomarsino A, Polle A, Ceulemans R, Scarascia Mugnozza G, Hoosbeek M, Cotrufo MF (2007) Nitrogen use efficiency of a short-rotation poplar plantation is increased under elevated CO2. Tree Physiol 27:1153–1163
Chapin FS, Schulze ED, Mooney HA (1990) The ecology and economics of storage in plants. Ann Rev Ecol Syst 21:423–447
Chapman JA, King JS, Pregitzer KS, Zak DR (2005) Effects of elevated CO2 and tropospheric O3 on tree fine root decomposition. Tree Physiol 25:1501–1510
Cooke JEK, Weih M (2005) Nitrogen storage and seasonal nitrogen cycling in Populus: bridging molecular physiology and ecophysiology. New Phytol 167:19–30
Cotrufo MF, Ineson P, Scott AY (1998) Elevated CO2 reduces the nitrogen concentration of plant tissues. Global Change Biol 4:43–54
Cotrufo MF, Angelis PD, Polle A (2005) Leaf litter production and decomposition in a poplar short-rotation coppice exposed to free air CO2 enrichment (POPFACE). Global Change Biol 11:971–982
Czochanska Z, Foo LY, Newman RH, Porter LJ (1980) Polymeric proanthocyanidins. Stereochemistry, structural units, and molecular weight. J Chem Soc Perkin Trans I:2278–2286
Dence CW (1992) Lignin determination. In: Stephen YL (ed) Methods in lignin chemistry. Springer-Verlag, Berlin, pp 33–61
Dyckmans J, Flessa H, Brinkmann K, Mai C, Polle A (2002) Carbon and nitrogen dynamics in acid detergent fibre lignins of beech (Fagus sylvatica L.) during the growth phase. Plant Cell Environ 25:469–478
Finzi AC, Norby RJ, Calfapietra C, Gallet-Budynek A, Gielen B, Holmes WE, Hoosbeek MR, Iversen CM, Jackson RB, Kubiske ME, Ledford J, Liberloo M, Oren R, Polle A, Pritchard S, Zak DR, Schlesinger WH, Ceulemans R (2007) Increases in nitrogen uptake rather than nitrogen use efficiency support higher rates of temperate forest productivity under elevated CO2. Proc Natl Acad Sci USA 104:14014–14019
Fordham M, Barnes JD, Bettarini I, Polle A, Slee N, Raines C, Miglietta F, Raschi A (1997) The impact of elevated CO2 on growth and photosynthesis in Agrostis canina L. ssp. monteluccii adapted to contrasting atmospheric CO2 concentrations. Oecologia 110:169–178
Geßler A, Kopriva S, Rennenberg H (2004) Regulation of nitrate uptake at the whole-tree level: interaction between nitrogen compounds, cytokinins and carbon metabolism. Tree Physiol 24:1313–1321
Gielen B, Ceulemans R (2001) The likely impact of rising atmospheric CO2 on natural and managed Populus: a literature review. Environ Poll 115:335–358
Gielen B, Calfapietra C, Lukac M, Wittig VE, Angelis PD, Janssens IA, Moscatelli MC, Grego S, Cotrufo MF, Godbold DL, Hoosbeek MR, Long SP, Miglietta F, Polle A, Bernacchi CJ, Davey PA, Ceulemans R, Scarascia-Mugnozza GE (2005) Net carbon storage in a poplar plantation (POPFACE) after three years of free-air CO2 enrichment. Tree Physiol 25:1399–1408
Goyal GC, Fisher JJ, Krohn MJ, Packood RE, Olson JR (1999) Variability in pulping and fiber characteristics of hybrid poplar trees due to their genetic makeup, environmental factors, and tree age. TAPPI 82:141–147
Harborne JB (1997) Plant secondary metabolism. In: Crawley MJ (ed) Plant ecology. 2nd edn. Blackwell Science, Oxford, pp 132–155
Hartley SE, Jones CG (1997) Plant chemistry and herbivory: or why the world is green. In: Crawley MJ (ed) Plant ecology 2nd edn, Blackwell Science, Oxford, pp 284–324
Hörtensteiner S, Feller U (2002) Nitrogen metabolism and remobilization during senescence. J Exp Bot 53:927–937
Kaakinen S, Kostiainen K, Fredrik EK, Saranpää P, Kubiske ME, Sober J, Karnosky DF, Vapaavuori E (2004) Stem wood properties of Populus tremuloides, Betula papyrifera and Acer saccharum saplings after three years of treatments to elevated carbon dioxide and ozone. Global Change Biol 10:1513–1525
Klasnja B, Kopitovic S, Orlovic S (2003) Variability of some wood properties of eastern cottonwood (Populus deltoides Bartr.) clones. Wood Sci Technol 37:331–337
Körner C, Asshoff R, Bignucolo O, Hättenschwiler S, Keel SG, Peláez-Riedl S, Pepin S, Siegwolf RTW, Zotz G (2005) Carbon flux and growth in mature deciduous forest trees exposed to elevated CO2. Science 309:1360–1362
Kostiainen K, Kaakinen S, Saranpaa P, Sigurdsson B, Linder S, Vapaavuori E (2004) Effect of elevated [CO2] on stem wood properties of mature Norway spruce grown at different soil nutrient availability. Global Change Biol 10:1526–1538
Kostiainen K, Jalkanen H, Kaakinen S, Saranpää P, Vapaavuori E (2006) Wood properties of two silver birch clones exposed to elevated CO2 and O3. Global Change Biol 12:1230–1240
Lavola A, Julkunen-Tiitto R (1994) The effect of elevated carbon dioxide and fertilization on primary and secondary metabolites in birch, Betula pendula (Roth). Oecologia 99:315–321
Liberloo M, Dillen S, Calfapietra C, Marinari S, Luo ZB, Angelis PD, Ceulemans R (2005) Elevated CO2 concentration, fertilization and their interaction: growth stimulation in a short-rotation poplar coppice (EUROFACE). Tree Physiol 25:179–189
Liberloo M, Calfapietra C, Lukac M, Godbold D, Luo ZB, Polle A, Kull O, Marek M, Raines C, Taylor G, Scarascia-Mugnozza G, Ceulemans R (2006) Woody biomass production during second rotation of a bio-energy Populus plantation increases in a future high CO2 world. Global Change Biol 12:1094–1106
Lindroth RL, Kopper BJ, Parsons WFJ, Bockheim JG, Karnosky DF, Hendrey GR, Pregitzer KS, Isebrands JG, Sober J (2001) Consequences of elevated carbon dioxide and ozone for foliar chemical composition and dynamics in trembling aspen (Populus tremuloides) and paper birch (Betula papyrifera). Environ Pollut 115:395–404
Lindroth RL, Osier TL, Barnhill HRH, Wood SA (2002) Effects of genotype and nutrient availability on phytochemistry of trembling aspen (Populus tremuloides Michx.) during leaf senescence. Biochem Syst Ecol 30:297–307
Long SP, Ainsworth EA, Rogers A, Ort DR (2004) Rising atmospheric carbon dioxide: plants FACE the future. Ann Rev Plant Biol 55:591–628
Luo Z-B, Calfapietra C, Liberloo M, Scarascia-Mugnozza G, Polle A (2006) Carbon partitioning to mobile and structural fractions in poplar wood under elevated CO2 (EUROFACE) and N-fertilization. Global Change Biol 12:272–283
Mattson WJ, Kuokkanen K, Niemela P, Julkunen-Tiitto R, Kellomaki S, Tahvanainen J (2004) Elevated CO2 alters birch resistance to lagomorpha herbivores. Global Change Biol 10:1402–1413
Mattson WJ, Julkunen-Tiitto R, Herms DA (2005) CO2 enrichment and carbon partitioning to phenolics: do plant responses accord better with the protein competition or the growth differentiation balance models. Oikos 111:337–347
Miglietta F, Peressotti A, Vaccari FP, Zaldei A, Angelis PD, Scarascia-Mugnozza G (2001) Free-air CO2 enrichment (FACE) of a poplar plantation: the POPFACE fumigation system. New Phytol 150:465–476
Millard P (1996) Ecophysiology of the internal cycling of nitrogen for tree growth. Z Pflanz Bodenkunde 159:1–10
Norby RJ, Iversen CM (2006) Nitrogen uptake, distribution, turnover, and efficiency of use in a CO2-enriched sweetgum forest. Ecol 87:5–14
Norby RJ, Long TM, Hartz-Rubin JS, O’Neill EG (2000) Nitrogen resorption in senescing tree leaves in a warmer, CO2-enriched atmosphere. Plant Soil 224:15–29
Norby RJ, Cotrufo MF, Ineson P, O’-Neill EG, Canadell JG (2001) Elevated CO2, litter chemistry, and decomposition: a synthesis. Oecologia 127:153–165
Norby RJ, DeLucia EH, Gielen B, Calfapietra C, Giardina CP, King JS, Ledford J, McCarthy HR, Moore DJP, Ceulemans R, Angelis PD, Finzi AC, Karnosky DF, Kubiske ME, Lukac M, Pregitzer KS, Scarascia-Mugnozza GE, Schlesinger WH, Oren R (2005) Forest response to elevated CO2 is conserved across a broad range of productivity. Proc Natl Acad Sci USA 102:18052–18056
Northrup RR, Dahlgren RA, McColl JG (1998) Polyphenols as regulators of plant-litter-soil interactions: a positive feedback. Biogeochem 42:189–220
Penuelas J, Estiarte M (1998) Can elevated CO2 affect secondary metabolism and ecosystem function. Trends Ecol Evol 13:20–24
Polle A, Otter T, Sandermann JRH (1997) Biochemistry and physiology of lignin synthesis. In: Rennenberg H, Eschrich W, Ziegler H (eds) Trees - contributions to modern tree physiology. Backhuys Publishers, Leiden, pp 455–475
Poorter H, Van Berkel Y, Baxter R (1997) The effect of elevated CO2 on the chemical composition and construction costs of leaves of 27 C3 species. Plant Cell Environ 20:472–482
Porter LJ, Hrstich L, Chan BG (1986) The conversion of procyanidins and prodelphinidins to cyanidin and delphinidin. Phytochem 25:223–230
Pritchard S, Peterson C, Runion GB, Prior S, Rogers H (1997) Atmospheric CO2 concentration, N availability, and water status affect patterns of ergastic substance deposition in longleaf pine (Pinus palustris Mill.) foliage. Trees 11:494–503
Roth SK, Lindroth RL (1994) Effects of CO2-mediated changes in paper birch and white pine chemistry on gypsy moth performance. Oecologia 98:133–138
Sauter JJ, van Cleve B (1994) Storage, mobilization and interrelations of starch, sugars, protein and fat in the ray storage tissue of poplar trees. Trees 8:297–304
Schlimme M, Blaschke L, Lagrimini M, Polle A (2002) Growth performance and lignification in tobacco with suppressed apoplastic anionic peroxidase activity under ambient and elevated CO2 concentrations. Int J Plant Sci 163:749–754
Sims RH, Hastings A, Schlamadinger B, Taylor G, Smith P (2006) Energy crops: current status and future prospects. Global Change Biol 12:2054–2076
Stitt M, Krapp JA (1999) The molecular physiological basis for the interaction between elevated carbon dioxide and nutrients. Plant Cell Environ 22:583–622
Swain T, Goldstein JL (1964) The quantitative analysis of phenolic compounds. In: Pridham JB (ed) Methods in polyphenol chemistry. Pergamon Press, Oxford, pp 131–145
Tricker PJ, Trewin H, Kull O, Clarkson GJJ, Eensalu E, Tallis MJ, Colella A, Doncaster CP, Sabatti M, Taylor G (2005) Stomatal conductance and not stomatal density determines the long-term reduction in leaf transpiration of poplar in elevated CO2. Oecol 143:652–660
Warren JM, Allen HL, Booker FL (1999) Mineral nutrition, resin flow and phloem phytochemistry in loblolly pine. Tree Physiol 19:655–663
Zak DR, Pregitzer KS, Curtis PS, Vogel CS, Holmes WE, Lussenhop J (2000) Atmospheric CO2, soil-N availability, and allocation of biomass and nitrogen by Populus tremuloides. Ecol Appl 10:34–46
Zak DR, Holmes WE, Finzi AC, Norby RJ, Schlesinger WH (2003) Soil nitrogen cycling under elevated CO2: a synthesis of forest FACE experiments. Ecol Appl 13:1508–1514
Acknowledgements
We are grateful to the European Union (contract number: EVR1-CT-2002-40027) and the Programme ‘Nachwuchswissenschaftler aus außereuropäischen Ländern nach Niedersachsen’ for financial support. Christine Kettner, Gisbert Langer-Kettner, Michael Reichel, Rainer Schulz and Thomas Klein are acknowledged for their assistance with sample collection in the field. We thank Marina Horstmann and Sabine Elend for providing assistance with the analysis of nitrogen and soluble proteins, respectively.
Open Access
This article is distributed under the terms of the Creative Commons Attribution Noncommercial License which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible Editor: Erik A. Hobbie.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplementary Table 1
Concentrations of soluble phenolics, cell wall-bound phenolics, soluble condensed tannins (CT) and lignin in mature leaves of P. nigra grown under either FACE (E) or ambient [CO2] (A) and with (H) or without nitrogen fertilisation (L) during two growing seasons. The values shown indicate means (±SE, n = 6). P values of the ANOVAs of CO2, nitrogen (N) and their interactions are also shown (DOC 41.0 KB).
Rights and permissions
Open Access This is an open access article distributed under the terms of the Creative Commons Attribution Noncommercial License (https://creativecommons.org/licenses/by-nc/2.0), which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
About this article
Cite this article
Luo, ZB., Calfapietra, C., Scarascia-Mugnozza, G. et al. Carbon-based secondary metabolites and internal nitrogen pools in Populus nigra under Free Air CO2 Enrichment (FACE) and nitrogen fertilisation. Plant Soil 304, 45–57 (2008). https://doi.org/10.1007/s11104-007-9518-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-007-9518-8