Abstract
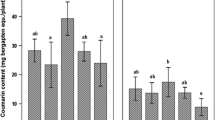
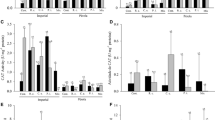
The effects of two arbuscular mycorrhizal fungi (AMF) (Glomus mosseae and G. claroideum) and a pathogenic fungus (Pythium ultimum) on the production of eight flavonoids in roots of two white clover (Trifolium repens L.) cultivars were evaluated. Quantification of AM and pathogenic fungi in the roots showed that the AM symbiosis significantly reduced P. ultimum biomass and in some cases prevented infection. The flavonoid productions in clover roots varied depending on the presence of beneficial and/or pathogenic fungi, fungal isolate or plant cultivar. Only plants colonized with G. claroideum showed detectable concentrations of either coumestrol or kaempferol (cultivar-dependant). In addition, inoculation with G. claroideum resulted in significantly higher concentrations of coumestrol in cv. Sonja and medicarpin in cv. Milo. A low production of coumestrol and kaempferol in mycorrhizal plants may be G. mosseae-specific. Only the concentrations of formononetin and daidzein increased in clover roots in response to infection with P. ultimum. These flavonoids are supposedly stress metabolites, synthesized or produced from glycosides in response to pathogen infection. However, the presence of one or both AMF significantly lowered the formononetin and daidzein concentrations, and overruled the inductive effect of P. ultimum. Therefore the antagonistic action of AM against the pathogen must take place through another mechanism.


Similar content being viewed by others
References
Al-Karaki GN, Hammad R (2001) Mycorrhizal influence on fruit yield and mineral content of tomato grown under salt stress. J Plant Nutr 24:1311–1323
Bennet D, Morley FHW, Axelsen A (1967) Bioassay responses of ewes to legume swards II. Uterine weight results from swards. Aust J Agric Res 18:504
Bickoff EM, Booth AN, Lyman RL, Livingston AL, Thompson CR, Kohler GO (1958) Isolation of a new estrogen from Ladino clover. J Agric Food Chem 6:536–539
Cantrell IC, Linderman RG (2001) Preinoculation of lettuce and onion with VA mycorrhizal fungi reduces deleterious effects of soil salinity. Plant Soil 233:269–281
Caradus JR, Woodfield DR (1997) World checklist of white clover varieties II. NZ J Agric Res 40:115–206
Catford JG, Stehelin C, Larose G, Piché Y, Vierheilig H (2006) Systemically suppressed isoflavonoids and their stimulating effects on nodulation and mycorrhization in alfalfa split-root systems. Plant Soil 285:257–266
Cruickshank IAM, Veeraraghavan J, Perrin DR (1974) Some physical factors affecting the formation and/or net accumulation of medicarpin in infection droplets on white clover leaflets. Aust J Plant Physiol 1:149–156
Davies FT Jr, Olalde-Portugal V, Aguilera-Gomez L, Alvarado MJ, Ferrera-Cerrato RC, Boutton TW (2002) Alleviation of drought stress of Chile ancho pepper (Capsicum annuum L. cv. San Luis) with arbuscular mycorrhiza indigenous to Mexico. Sci Hortic 92:347–359
Foo LY, Lu Y, Molan AL, Woodfield DR, McNabb WC (2000) The phenols and prodelphinidins of white clover flowers. Phytochemistry 54:539–548
Grant EA, Sallans WG (1964) Influence of plant extracts on germination and growth of eight forage species. J Brit Grassland Soc 19:191–197
Guenoune D, Galili S, Phillips DA, Volpin H, Chet I, Okon Y, Kapulnik Y (2001) The defense response elicited by the pathogen Rhizoctonia solani is suppressed by colonization of the AM-fungus Glomus intraradices. Plant Sci 160:925–932
Hendrix FF, Campbell WA (1973) Pythiums as plant pathogens. A Rev Phytopath 11:77–98
Hofmann RW, Swinny EE, Bloor SJ, Markham KR, Ryan KG, Campbell BD, Jordan BR, Fountain DW (2000) Responses of nine Trifolium repens L. populations to ultraviolet-B radiation: differential flavonol glycoside accumulation and biomass production. Ann Bot 86:527–537
Ingham JL (1978) Isoflavonoid and stilbene phytoalexins of the genus Trifolium. Biochem Syst Ecol 6:217–223
Ingham JL (1982) Phytoalexins from the Leguminosae. In: Bailey JA, Mansfield JW (eds) Phytoalexins. Blackie, London, UK, pp 21–80
Kormanik PP, McGraw AC (1982) Quantification of vesicular–arbuscular mycorrhizae in plant roots. In: Schenck NC (ed) Methods and principles of mycorrhizal research. The American Phytopathological Society, St. Paul, MN, pp 37–45
Larose G, Chênevert R, Moutoglis P, Gagné S, Piché Y, Vierheilig H (2002) Flavonoid levels in roots of Medicago sativa are modulated by the developmental stage of the symbiosis and the root colonizing arbuscular mycorrhizal fungus. J Plant Physiol 159:1329–1339
Larsen J, Ravnskov S, Jakobsen I (2003) Combined effect of an arbuscular mycorrhizal fungus and a biocontrol bacterium against Pythium ultimum in soil. Folia Geobot 38:145–154
Linderman RG (2000) Effects of mycorrhizas on plant tolerance to diseases, Mycorrhiza-disease interactions. In: Kapulnik Y, Douds DD Jr (eds) Arbuscular mycorrhizas: physiology and function. Kluwer, The Netherlands, pp 345–365
Masterson CL (1965) Studies of the toxicity of legume seeds towards Rhizobium. Ann Inst Pasteur (Paris) 109(Suppl):216–217
Morandi D, Bailey JA, Gianinazzi-Pearson V (1984) Isoflavonoid accumulation in soybean roots infected with vesicular–arbuscular mycorrhizal fungi. Physiol Plant Pathol 24:357–364
Nair MG, Safir GR, Siqueira JO (1991) Isolation and identification of vesicular–arbucular mycorrhiza-stimulatory compounds from clover (Trifolium repens) roots. Appl Environ Microbiol 57(2):434–439
Nakatani M, Tajiri M, Kaga T, Fukami R, Takada Y, Hase T (1989) A dihydroxycyclopentadienone and other constituents from the seeds of Trifolium repens. Phytochemistry 28:2499–2501
Newman EI (1966) A method of estimating the total length of roots in a sample. J Appl Ecol 3:139–145
Newman EI, Rovira AD (1975) Allelopathy among some British grassland species. J Ecol 63:727–737
Oleszek W, Stochmal A (2002) Triterpene saponins and flavonoids in the seeds of Trifolium species. Phytochemistry 61:165–170
Pemberton IJ, Smith GR, Philey GL, Rouquette FM (1998) First report of Pythium ultimum, P. irregulare, Rhizoctonia solani AG4, and Fusarium proliferatum from arrowleaf clover (Trifolium vesiculosum): a disease complex. Plant Dis 82:128
Ponce MA, Scervino JM, Erra-Balsells R, Ocampo JA, Godeas AM (2004) Flavonoids form shoots and roots of Trifolium repens (white clover) grown in presence or absence of the arbuscular mycorrhizal fungus Glomus intraradices. Phytochemistry 65:1925–1930
Ratti N, Alam M, Sharma S, Janardhanan KK (1998) Effects of Glomus aggregatum on lethal yellowing disease of java citronella caused by Pythium aphanidermatum. Symbiosis 24:115–126
Ravnskov S, Jakobsen I (1995) Functional compatibility in arbuscular mycorrhizas measured as hypal P transport to the plant. New Phytol 129:611–618
Rosendahl CN, Rosendahl S (1990) The role of vesicular–arbuscular mycorrhiza in controlling damping-off and growth reduction in cucumber caused by Pythium ultimum. Symbiosis 9:363–366
Ruan Y, Kotraish V, Straney DC (1995) Flavonoids stimulate spore germination in Fusarium solani pathogenic on legumes in a manner sensitive to inhibitors of cAMP-dependent protein kinase. MPMI 8(6):929–938
Saba N, Drane HM, Herbert CN, Holdsworth RJ (1974) Seasonal variation in oestrogenic activity, coumestrol and formononetin content of white clover. J Agric Sci 83:505–510
Sachse J (1974) Die Bestimmung östrogener Isoflavone und Cumöstrol in Klee (Trifolium pratense L. und Trifolium repens L.). J Chromatogr A 96(1):123–136
Saloniemi H, Kallela K, Saastamoinen I (1993) Study of the phytoestrogen content of goat’s rue (Galega orientalis), alfalfa (Medicago sativa) and white clover (Trifolium repens). Agric Sci Finland 2:517–524
Scervino JM, Ponce MA, Erra-Bassells R, Vierheilig H, Ocampo JA, Godeas A (2005a) Flavonoids exclusively present in mycorrhizal roots of white clover exhibit a different effect on arbuscular mycorrhizal fungi than flavonoids exclusively present in non-mycorrhizal roots of white clover. J Plant Interact 1(1):15–22
Scervino JM, Ponce MA, Erra-Bassells R, Vierheilig H, Ocampo JA, Godeas A (2005b) Flavonoids exhibit fungal species and genus specific effects on the presymbiotic growth of Gigaspora and Glomus. Mycol Res 109(7):789–794
Scott D (1975) Allelopathic interactions of resident tussock grassland species on germination of oversown seed. J Exp Agric 3:135–141
Siqueira JO, Safir GR, Nair MG (1991) Stimulation of VAM formation and growth of white clover by flavonoid compounds. New Phytol 118:87–93
St-Arnaud M, Hamel C, Caron M, Fortin JA (1994) Inhibition of Pythium ultimum in roots and growth substrate of mycorrhizal Tagetes patula colonized with Glomus interadices. Can J Plant Pathol 16:187–194
Steinkellner S, Lendzemo V, Langer I, Schweiger P, Khaosaad T, Toussaint J-P, Vierheilig H (2007) Flavonoids and strigolactones in root exudates as signals in symbiotic and pathogenic plant–fungus interactions. Molecules 12:1290–1306
Sundaresan P, Ubalthoose Raja N, Gunasekaran P (1993) Induction and accumulation of phytoalexins in cowpea roots infected with a mycorrhizal fungus Glomus fasciculatum and their resistance to Fusarium wilt disease. J Biosci 18:291–301
van den Bosch J, Mercer CF (1996) Variation in white clover for root-knot nematode resistance. NZ J Agric Res 39:137–148
VanEtten HD (1976) Antifungal activity of pterocarpans and other selected isoflavonoids. Phytochemistry 15:655–659
Vetter J (1995) Isoflavones in different parts of common Trifolium species. J Agric Food Chem 43:106–108
Vierheilig H (2004a) Further root colonization by arbuscular mycorrhizal fungi in already mycorrhizal plants is supressed after a critical level of root colonization. J Plant Physiol 161:339–341
Vierheilig H (2004b) Regulatory mechanisms during the plant-arbuscular mycorrhizal fungus interaction. Can J Bot 82:1166–1176
Virtanen AI, Hietala PK (1958) Isolation of an anti-sclerotinia factor, 7-hydroxy-4′-methoxyisoflavone from red clover. Acta Chem Scand 12(3):579–580
Volpin H, Elkind Y, Okon Y, Kapulnik Y (1994) A vesicular arbuscular mycorrhizal fungus (Glomus intraradix) induces a defence response in alfalfa roots. Plant Physiol 104:683–689
Volpin H, Phillips DA, Okon Y, Kapulnik Y (1995) Suppression of an isoflavonoid phytoalexin defence response in mycorrhizal alfalfa roots. Plant Physiol 108:1449–1454
Weidenbörner M, Hindorf H, Jha HC, Tsotsonos P, Egge H (1990) Antifungal activity of isoflavonoids in different reduced stages on Rhizoctonia solani and Sclerotium rolfsii. Phytochemistry 29:801–803
Whipps J (2004) Prospects and limitations of mycorrhizas in biocontrol of root pathogens. Can J Bot 82:1198–1227
Wong E, Latch GCM (1971a) Leguminosae. Coumestans in diseased white clover. Phytochemistry 10:466–468
Wong E, Latch GCM (1971b) Effect of fungal diseases on phenolic contents of white clover. NZJ Agric Res 14:633–638
Wu Q, Wang M, Simon JE (2003) Determination of isoflavones in red clover and related species by high-performance liquid chromatography combined with ultraviolet and mass spectrometric detection. J Chromatogr 1016:195–209
Wyss P, Boller T, Wiemken A (1991) Phytoalexin response is elicited by a pathogen (Rhizoctonia solani) but not by a mycorrhizal fungus (Glomus mosseae) in soybean roots. Experientia 47:395–399
Zahid MI, Gurr GM, Nikandrow A, Hodda M, Fulkerson WJ, Nicol HI (2001) Survey of fungi and nematodes associated with root and stolon diseases of white clover in the subtropical dairy region of Australia. Austr J Exp Agric 41:1133–1142
Acknowledgment
We thank Dr. Paul M. Dewick, University of Nottingham, England, for providing a sample of medicarpin.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Petra Marschner
Rights and permissions
About this article
Cite this article
Carlsen, S.C.K., Understrup, A., Fomsgaard, I.S. et al. Flavonoids in roots of white clover: interaction of arbuscular mycorrhizal fungi and a pathogenic fungus. Plant Soil 302, 33–43 (2008). https://doi.org/10.1007/s11104-007-9452-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-007-9452-9