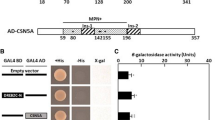
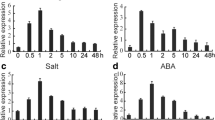
Abstract
Key message
A stress induced calcium-binding protein, RD20/CLO3 interacts with the alpha subunit of the heterotrimeric G-protein complex in Arabidopsis and affects etiolation and leaf morphology.
Abstract
Heterotrimeric G proteins and calcium signaling have both been shown to play a role in the response to environmental abiotic stress in plants; however, the interaction between calcium-binding proteins and G-protein signaling molecules remains elusive. We investigated the interaction between the alpha subunit of the heterotrimeric G-protein complex, GPA1, of Arabidopsis thaliana with the calcium-binding protein, the caleosin RD20/CLO3, a gene strongly induced by drought, salt and abscisic acid. The proteins were found to interact in vivo by bimolecular fluorescent complementation (BiFC); the interaction was localized to the endoplasmic reticulum and to oil bodies within the cell. The constitutively GTP-bound GPA1 (GPA1QL) also interacts with RD20/CLO3 as well as its EF-hand mutant variations and these interactions are localized to the plasma membrane. The N-terminal portion of RD20/CLO3 was found to be responsible for the interaction with GPA1 and GPA1QL using both BiFC and yeast two-hybrid assays. RD20/CLO3 contains a single calcium-binding EF-hand in the N-terminal portion of the protein; disruption of the calcium-binding capacity of the protein obliterates interaction with GPA1 in in vivo assays and decreases the interaction between the caleosin and the constitutively active GPA1QL. Analysis of rd20/clo3 mutants shows that RD20/CLO3 plays a key role in the signaling pathway controlling hypocotyl length in dark grown seedlings and in leaf morphology. Our findings indicate a novel role for RD20/CLO3 as a negative regulator of GPA1.







Similar content being viewed by others
References
Abell BM, Holbrook LA, Abenes M, Murphy DJ, Hills MJ, Moloney MM (1997) Role of the proline knot motif in oleosin endoplasmic reticulum topology and oil body targeting. Plant Cell 9(8):1481–1493. https://doi.org/10.1105/tpc.9.8.1481
Aharon GS, Gelli A, Snedden WA, Blumwald E (1998) Activation of a plant plasma membrane Ca2+ channel by TG alpha1, a heterotrimeric G protein alpha-subunit homologue. FEBS Lett 424:17–21
Aubert Y, Vile D, Pervent M, Aldon D, Ranty B, Simonneau T, Vavasseur A, Galaud JP (2010) RD20, a stress-inducible caleosin, participates in stomatal control, transpiration and drought tolerance in Arabidopsis thaliana. Plant Cell Physiol 51:1975–1987
Auslender EL, Dorion S, Dumont S, Rivoal J (2015) Expression, purification and characterization of Solanum recombinant cytosolic pyruvate kinase. Protein Expr Purif 110:7–13
Blée E, Boachon B, Burcklen M, Le Guédard M, Hanano A, Heintz D, Ehlting J, Herrfurth C, Feussner I, Bessoule J-J (2014) The reductase activity of the Arabidopsis caleosin responsive to dessication 20 mediates gibberellin-dependent flowering time, abscisic acid sensitivity, and tolerance to oxidative stress. Plant Physiol 166:109–124
Chakravorty D, Gookin TE, Milner MJ, Yu Y, Assmann SM (2015) Extra-large G proteins (XLGs) expand the repertoire of subunits in Arabidopsis heterotrimeric G protein signaling. Plant Physiol 169(1):512–529
Chapman KD, Dyer JM, Mullen RT (2012) Biogenesis and functions of lipid droplets in plants: thematic review series: lipid droplet synthesis and metabolism: from yeast to Man. J Lipid Res 53(2):215–226. https://doi.org/10.1194/jlr.R021436
Chater CC, Oliver J, Casson S, Gray JE (2014) Putting the brakes on: abscisic acid as a central environmental regulator of stomatal development. New Phytol 202:376–391
Chen JG, Gao Y, Jones AM (2006) Differential roles of Arabidopsis heterotrimeric G-protein subunits in modulating cell division in roots. Plant Physiol 141:887–897
Chen JC, Tsai CC, Tzen JT (1999) Cloning and secondary structure analysis of caleosin, a unique calcium-binding protein in oil bodies of plant seeds. Plant Cell Physiol 40:1079–1086
Chen JG, Willard FS, Huang J, Liang J, Chasse SA, Jones AM, Siderovski DP (2003) A seven-transmembrane RGS protein that modulates plant cell proliferation. Science 301:1728–1731
Clough SJ, Bent AF (1998) Floral dip: a simplified method for Agrobacterium-mediated transformation of Arabidopsis thaliana. Plant J 16:736–743
De Vendittis E, Adinolfi BS, Amatruda MR, Raimo G, Masullo M, Bocchini V (1999) The A26G replacement in the consensus sequence A-X-X-X-X-G-K-[T, S] of the guanine nucleotide binding site activates the intrinsic GTPase of the elongation factor 2 from the archaeon Sulfolobus solfataricus. Eur J Biochem 262:600–605
Dorion S, Rivoal J (2003) Quantification of uridine 5’-diphosphate (UDP)-glucose by high-performance liquid chromatography and its application to a nonradioactive assay for nucleoside diphosphate kinase using UDP-glucose pyrophosphorylase as a coupling enzyme. Anal Biochem 323:188–196
Edwards K, Johnstone C, Thompson C (1991) A simple and rapid method for the preparation of plant genomic DNA for PCR analysis. Nucleic Acids Res 19:1349
Frandsen GI, Mundy J, Tzen JT (2001) Oil bodies and their associated proteins, oleosin and caleosin. Physiol Plant 112(3):301–307. https://doi.org/10.1034/j.1399-3054.2001.1120301.x
Gendreau E, Traas J, Demos’ T, Crandjean O, Caboche M, Hofte H (1997) Cellular basis of hypocotyl growth in Arabidopsis thaliana. Plant Physiol 114:295
Gookin TE, Assmann SM (2014) Significant reduction of BiFC non-specific assembly facilitates in planta assessment of heterotrimeric G-protein interactors. Plant J 80:553–567
Hanano A, Bessoule J-J, Heitz T, Blée E (2015) Involvement of the caleosin/peroxygenase RD20 in the control of cell death during Arabidopsis responses to pathogens. Plant Signal Behav 10(3):e991574
Huang CY, Chung CI, Lin YC, Hsing YI, Huang AH (2009) Oil bodies and oleosins in Physcomitrella possess characteristics representative of early trends in evolution. Plant Physiol 150:1192–1203
Ito H, Fukuda Y, Murata K, Kimura A (1983) Transformation of intact yeast cells treated with alkali cations. J Bacteriol 153:163–168
Johnston CA, Taylor JP, Gao Y, Kimple AJ, Grigston JC, Chen JG, Siderovski DP, Jones AM, Willard FS (2007) GTPase acceleration as the rate-limiting step in Arabidopsis G protein-coupled sugar signaling. Proc Natl Acad Sci USA 104:17317–17322
Jones AM (2002) G-protein-coupled signaling in Arabidopsis. Curr Opin Plant Biol 5:402–407
Jones AM, Ecker JR, Chen JG (2003) A reevaluation of the role of the heterotrimeric G protein in coupling light responses in Arabidopsis. Plant Physiol 131:1623–1627
Kapila J, De Rycke R, Angenon G (1997) An Agrobacterium-mediated transient gene expression system for intact leaves. Plant Sci 122:101–108
Karimi M, Inze D, Depicker A (2002) GATEWAYTM vectors for Agrobacterium-mediated plant transformation. Trends Plant Sci 7:193–195
Kato C, Mizutani T, Tamaki H, Kumagai H, Kamiya T, Hirobe A, Fujisawa Y, Yato H, Iwasaki Y (2004) Characterization of heterotrimeric G protein complexes in rice plasma membrane. Plant J 38:320–331
Khalil HB, Wang Z, Wright JA, Ralevski A, Donayo AO, Gulick PJ (2011) Heterotrimeric Gα subunit from wheat (Triticum aestivum), GA3, interacts with the calcium-binding protein, Clo3, and the phosphoinositide-specific phospholipase C, PI-PLC1. Plant Mol Biol 77:145–158
Kim G-T, Tsukaya H, Uchimiya H (1998) The ROTUNDIFOLIA3gene of Arabidopsis thaliana encodes a new member of the cytochrome P-450 family that is required for the regulated polar elongation of leaf cells. Genes Dev 12:2381–3239
Klopffleisch K, Phan N, Augustin K, Robert BS, Booker KS, Botella JR, Carpita NC, Carr T, Chen J, Cooke TR, Frick-Cheng A, Friedman EJ, Fulk B, Hahn MG, Jiang K, Jorda L, Kruppe L, Liu C, Lorek J, McCann MC, Molina A, Moriyama EN, Mukhtar MS, Mudgil Y, Pattathil S, Schwarz J, Seta S, Tan M, Temp U, Trusov Y, Urano D, Welter B, Yang J, Panstruga R, Uhrig JF, Jones AM (2011) Arabidopsis G-protein interactome reveals connections to cell wall carbohydrates and morphogenesis. Mol Syst Biol 7:532
Kreps JA, Wu Y, Chang H-S, Zhu T, Wang X, Harper JF (2002) Transcriptome changes for Arabidopsis in response to salt, osmotic and cold stress. Plant Physiol 130:2129–2141
Lu Q, Tang X, Tian G, Wang F, Liu K, Nguyen V, Kohalmi SE, Keller WA, Tsang EW, Harada JJ, Rothstein SJ, Cui Y (2010) Arabidopsis homolog of the yeast TREX-2 mRNA export complex: components and anchoring nucleoporin. Plant J 61(2):259–270
Marrari Y, Crouthamel M, Irannejad R, Wedegaertner PB (2007) Assembly and trafficking of heterotrimeric G proteins. Biochemistry 46:7665–7677
Misra S, Wu Y, Venkataraman G, Sopory SK, Tuteja N (2007) Heterotrimeric G-protein complex and G-protein-coupled receptor from a legume (Pisum sativum): role in salinity and heat stress and cross-talk with phospholipase C. Plant J 51:656–669
Naested H, Frandsen GI, Jauh GY, Hernandez-Pinzon I, Nielsen HB, Murphy DJ, Rogers JC, Mundy J (2000) Caleosins: Ca2+-binding proteins associated with lipid bodies. Plant Mol Biol 44:463–476
Nilson SE, Assmann SM (2010) The alpha-subunit of the Arabidopsis heterotrimeric G protein, GPA1 is a regulator of transpiration efficiency. Plant Physiol 152:2067–2077
Okamoto M, Tanaka Y, Abrams SR, Kamiya Y, Seki M, Nambara E (2009) High humidity induces abscisic acid 8′-hydroxylase in stomata and vasculature to regulate local and systemic abscisic acid responses in Arabidopsis. Plant Physiol 149:825–834
Oono Y, Seki M, Nanjo T, Narusaka M, Fujita M, Satoh R, Satou M, Sakurai T, Ishida J, Akiyama K, Lida K, Maruyama K, Satoh S, Yamaguchi-Shinozaki K, Shhinozaki K (2003) Monitoring expression profiles of Arabidopsis gene expression during rehydration process after dehydration using ca. 7000 full-length cDNA microarray. Plant J 34:868–887
Pandey S (2019) Heterotrimeric G-protein signaling in plants: conserved and novel mechanisms. Annu Rev Plant Biol 70:213–238. https://doi.org/10.1146/annurev-arplant-050718-100231
Pandey S, Chen JG, Jones AM, Assmann SM (2006) G-protein complex mutants are hypersensitive to abscisic acid regulation of germination and postgermination development. Plant Physiol 141:243–256
Pandey S, Nelson DC, Assmann SM (2009) Two novel GPCR-type G proteins are abscisic acid receptors in Arabidopsis. Cell 136:136–148
Perfus-Barbeoch L, Jones AM, Assmann SM (2004) Plant heterotrimeric G protein function: insights from Arabidopsis and rice mutants. Curr Op in Plant Biology 7:719–731
Peng Y, Chen L, Li S, Zhang Y, Xu R, Liu Z, Liu W, Kong J, Huang X, Wang Y, Cheng B, Zheng L, Li Y (2018) BRI1 and BAK1 interact with G proteins and regulate sugar-responsive growth and development in Arabidopsis. Nat Commun 9:1522. https://doi.org/10.1038/s41467-018-03884-8
Phobius A combined transmembrane topology and signal peptide predictor. Stockholm Bioinformatic Center. http://phobius.sbc.su.se/.
Piazza M, Taiakina V, Dieckmann T, Guillemette JG (2017) Structural consequences of calmodulin EF hand mutations. Biochemistry 56(7):944–956. https://doi.org/10.1021/acs.biochem.6b01296
Rasband WS (1997–2014) ImageJ, U. S. National Institutes of Health, Bethesda, Maryland, USA, http://imagej.nih.gov/ij/.
Ritchie S, Gilroy S (2000) Abscisic acid stimulation of phospholipase D in the barley aleurone is G-protein-mediated and localized to the plasma membrane. Plant Physiol 124:693–702
Shimada TL, Shimada T, Hara-Nishimura I (2010) A rapid and non-destructive screenable marker, FAST, for identifying transformed seeds of Arabidopsis thaliana. Plant J 61:519–528
Steffens B, Sauter S (2010) G proteins as regulators in ethylene-mediated hypoxia signaling. Plant Signal Behav 5:375–378
Studier FW (2005) Protein production by auto-induction in high density shaking cultures. Protein Expr Purif 41(1):207–234
Takahashi S, Katagiri T, Yamaguchi-Shinozaki K, Shinozaki K (2000) An Arabidopsis gene encoding a Ca2+-binding protein is induced by abscisic acid during dehydration. Plant Cell Physiol 41:898–903
Temple BRS, Jones AM (2007) The plant heterotrimeric G-protein complex. Annu Review Plant Biology 58:249–266
Tsuge, T., Tsukaya, H., and Uchimiya, H. (1996). Two independent and polarized processes of cell elongation regulate leaf blade expansion in Arabidopsis thaliana (L.) Heynh. Development. 122(5):1589–1600.
Tuteja N, Mahajan S (2007) Calcium signaling network in plants: an overview. Plant Signal Behav 2(2):79–85. https://doi.org/10.4161/psb.2.2.4176
Uhrig JF, Mutondo M, Zimmermann I, Deeks MJ, Machesky LM, Thomas P, Uhrig S, Rambke C, Hussey PJ, Hülskamp M (2007) The role of Arabidopsis SCAR genes in ARP2-ARP3-dependent cell morphogenesis. Development 134:967–977
Ullah H, Chen JG, Temple B, Alonso J, Ecker J, Boyes D, Davis K, Jones AM (2003) The β-subunit of the Arabidopsis g protein negatively regulates auxin-induced cell division and affects multiple developmental processes. Plant Cell 15:393–409
Ullah H, Chen JG, Young JC, Im K-H, Sussman MR, Jones AM (2001) Modulation of cell proliferation by heterotrimeric G protein in Arabidopsis. Science 292:2066
Wang S, Assmann SM, Fedoroff NV (2008) Characterization of the Arabidopsis heterotrimeric G protein. J Biol Chem 283:13913–13922
Wang X, Ullah H, Jones AJ, Assmann SM (2001) G protein regulation of ion channels and abscisic acid signaling in Arabidopsis guard cells. Science 292:2070–2072
Xiong LW, Kleerekoper QK, Wang X, Putkey JA (2010) Intra- and interdomain effects due to mutation of calcium-binding sites in calmodulin. J Biol Chem 285(11):8094–8103. https://doi.org/10.1074/jbc.M109.065243
Acknowledgements
We would like to thank Dr. Alan Jones, North Carolina State University for the gift of the gpa1 mutant line, Dr. Sarah Assmann (Pennsylvania State U) for the gift of the GPA1 promoter::GUS transgenic Arabidopsis reporter line and for the AGB1/AGG3(γ)-BD fusion for Y2H, and Dr. Elizabeth Blée (Université de Strasbourg) for the gift of the P35S:RD20/rd20 line. We would like to thank Dr. Deborah Maret for editorial assistance. The authors acknowledge the Centre for Microscopy and Cellular Imaging (CMCI) funded by Concordia University, Montreal, Canada and the Canada Foundation for Innovation.
Funding
This work is supported by a grant from the Natural Science and Engineering Research Council of Canada.
Author information
Authors and Affiliations
Contributions
PJG designed the project. SCB, ZW, JAW, and HK participated in the experimental design. SCB, MKMA, ZW, JAW, HK, GG, ME, JR and MJL performed the experiments and analyzed data. SCB, MKMA, JAW, and PJG contributed to writing and revision of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare there is no conflict of interest.
Consent for publication
All authors have given consent to publish.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Brunetti, S.C., Arseneault, M.K.M., Wright, J.A. et al. The stress induced caleosin, RD20/CLO3, acts as a negative regulator of GPA1 in Arabidopsis. Plant Mol Biol 107, 159–175 (2021). https://doi.org/10.1007/s11103-021-01189-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11103-021-01189-x