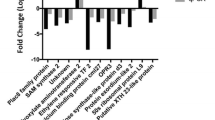
Abstract
Key message
The transcriptome comparison of two oak species reveals possible candidates accounting for the exceptionally thick and pure cork oak phellem, such as those involved in secondary metabolism and phellogen activity.
Abstract
Cork oak, Quercus suber, differs from other Mediterranean oaks such as holm oak (Quercus ilex) by the thickness and organization of the external bark. While holm oak outer bark contains sequential periderms interspersed with dead secondary phloem (rhytidome), the cork oak outer bark only contains thick layers of phellem (cork rings) that accumulate until reaching a thickness that allows industrial uses. Here we compare the cork oak outer bark transcriptome with that of holm oak. Both transcriptomes present similitudes in their complexity, but whereas cork oak external bark is enriched with upregulated genes related to suberin, which is the main polymer responsible for the protective function of periderm, the upregulated categories of holm oak are enriched in abiotic stress and chromatin assembly. Concomitantly with the upregulation of suberin-related genes, there is also induction of regulatory and meristematic genes, whose predicted activities agree with the increased number of phellem layers found in the cork oak sample. Further transcript profiling among different cork oak tissues and conditions suggests that cork and wood share many regulatory mechanisms, probably reflecting similar ontogeny. Moreover, the analysis of transcripts accumulation during the cork growth season showed that most regulatory genes are upregulated early in the season when the cork cambium becomes active. Altogether our work provides the first transcriptome comparison between cork oak and holm oak outer bark, which unveils new regulatory candidate genes of phellem development.






Similar content being viewed by others
References
Andersson-Gunnerås S, Mellerowicz EJ, Love J, Segerman B, Ohmiya Y, Coutinho PM et al (2006) Biosynthesis of cellulose-enriched tension wood in Populus: global analysis of transcripts and metabolites identifies biochemical and developmental regulators in secondary wall biosynthesis. Plant J 45:144–165. https://doi.org/10.1111/j.1365-313X.2005.02584.x
Andre C, Froehlich JE, Moll MR, Benning C (2007) A heteromeric plastidic pyruvate kinase complex involved in seed oil biosynthesis in Arabidopsis. Plant Cell 19:2006–2022
Andrés F, Coupland G (2012) The genetic basis of flowering responses to seasonal cues. Nat Rev Genet 13:627–639. https://doi.org/10.1038/nrg3291
Azeez A, Miskolczi P, Tylewicz S, Bhalerao RP (2014) A tree ortholog of APETALA1 mediates photoperiodic control of seasonal growth. Curr Biol 24:717–724. https://doi.org/10.1016/j.cub.2014.02.037
Barel G, Ginzberg I (2008) Potato skin proteome is enriched with plant defence components. J Exp Bot 59:3347–3357. https://doi.org/10.1093/jxb/ern184
Baud S, Wuillème S, Dubreucq B, De Almeida A, Vuagnat C, Lepiniec L et al (2007) Function of plastidial pyruvate kinases in seeds of Arabidopsis thaliana. Plant J 52:405–419. https://doi.org/10.1111/j.1365-313X.2007.03232.x
Beisson F, Li Y, Bonaventure G, Pollard M, Ohlrogge JB (2007) The acyltransferase GPAT5 is required for the synthesis of suberin in seed coat and root of Arabidopsis. Plant Cell 19:351–368. https://doi.org/10.1105/tpc.106.048033
Benjamini Y, Hochberg Y (1995) Controlling the false discovery rate: a practical and powerful approach to multiple testing. J R Stat Soc B 57:289–300. https://doi.org/10.2307/2346101
Bernal AJ, Yoo C-M, Mutwil M, Jensen JK, Hou G, Blaukopf C, Sørensen I, Blancaflor EB, Scheller HV, Willats WG (2008) Functional analysis of the cellulose synthase-like genes CSLD1, CSLD2, and CSLD4 in tip-growing Arabidopsis cells. Plant Physiol 148:1238–1253. https://doi.org/10.1104/pp.108.121939
Bernards MA (2002) Demystifying suberin. Can J Bot 80:227–240. https://doi.org/10.1139/B02-017
Bourgis F, Kilaru A, Cao X, Ngando-Ebongue GF, Drira N, Ohlrogge JB et al (2011) Comparative transcriptome and metabolite analysis of oil palm and date palm mesocarp that differ dramatically in carbon partitioning. Proc Natl Acad Sci USA 108:12527–12532. https://doi.org/10.1073/pnas.1106502108
Caño-Delgado A, Yin Y, Yu C, Vafeados D, Mora-García S, Cheng JC et al (2004) BRL1 and BRL3 are novel brassinosteroid receptors that function in vascular differentiation in Arabidopsis. Development 131:5341–5351. https://doi.org/10.1242/dev.01403
Caritat A, Gutierrez E, Molinas M (2000) Influence of weather on cork-ring width. Tree Physiol 20:893–900. https://doi.org/10.1093/treephys/20.13.893
Castola V, Marongiu B, Bighelli A, Floris C, Laï A, Casanova J (2005) Extractives of cork (Quercus suber L.): chemical composition of dichloromethane and supercritical CO2 extracts. Ind Crops Prod 21:65–69. https://doi.org/10.1016/j.indcrop.2003.12.007
Cavallini E, Matus JT, Finezzo L, Zenoni S, Loyola R, Guzzo F et al (2015) The phenylpropanoid pathway is controlled at different branches by a set of R2R3-MYB C2 repressors in grapevine. Plant Physiol 167:1448–1470. https://doi.org/10.1104/pp.114.256172
Choe S, Dilkes BP, Gregory BD, Ross AS, Yuan H, Noguchi T et al (1999) The Arabidopsis dwarf1 mutant is defective in the conversion of 24-methylenecholesterol to campesterol in brassinosteroid biosynthesis. Plant Physiol 119:897–907. https://doi.org/10.1104/pp.119.3.897
Compagnon V, Diehl P, Benveniste I, Meyer D, Schaller H, Schreiber L et al (2009) CYP86B1 is required for very long chain omega-hydroxyacid and alpha, omega-dicarboxylic acid synthesis in root and seed suberin polyester. Plant Physiol 150:1831–1843. https://doi.org/10.1104/pp.109.141408
Crevillén P, Yang H, Cui X, Greeff C, Trick M, Qiu Q et al (2014) Epigenetic reprogramming that prevents transgenerational inheritance of the vernalized state. Nature 515:587–590. https://doi.org/10.1038/nature13722
Enjuto M, Balcells L, Campos N, Caelles C, Arró M, Boronat A (1994) Arabidopsis thaliana contains two differentially expressed 3-hydroxy-3-methylglutaryl-CoA reductase genes, which encode microsomal forms of the enzyme. Proc Natl Acad Sci USA 91:927–931. https://doi.org/10.1073/pnas.91.3.927
Evert RF, Eichhorn SE (2006). Periderm. In Wiley-Blackwell (ed) Esau’s plant anatomy: meristems, cells, and tissues of the plant body: their structure, function, and development, 3rd edn. Wiley, New York
Fahn A (1967) Plant anatomy. Pergamon Press, Oxford
Fukuda H (1997) Tracheary element differentiation. Plant Cell 9:1147–1156. https://doi.org/10.1105/tpc.9.7.1147
Gil L (2014) Cork: a strategic material. Front Chem 2:1–2. https://doi.org/10.3389/fchem.2014.00016
Girard AL, Mounet F, Lemaire-Chamley M, Gaillard C, Elmorjani K, Vivancos J et al (2012) Tomato GDSL1 is required for cutin deposition in the fruit cuticle. Plant Cell 24:3119–3134. https://doi.org/10.1105/tpc.112.101055
Götz S, García-Gómez JM, Terol J, Williams TD, Nagaraj SH, Nueda MJ et al (2008) High-throughput functional annotation and data mining with the Blast2GO suite. Nucleic Acids Res 36:3420–3435. https://doi.org/10.1093/nar/gkn176
Gou JY, Yu XH, Liu CJ (2009) A hydroxycinnamoyltransferase responsible for synthesizing suberin aromatics in Arabidopsis. Proc Natl Acad Sci USA 106:18855–18860. doi:https://doi.org/10.1073/pnas.0905555106\r0905555106
Gou M, Hou G, Yang H, Zhang X, Cai Y, Kai G, Liu CJ (2017) The MYB107 transcription factor positively regulates suberin biosynthesis. Plant Physiol 173(2):1045–1058. https://doi.org/10.1104/pp.16.01614
Graça J (2015) Suberin the biopolyester at the frontier of plants. Front Chem. https://doi.org/10.3389/fchem.2015.00062
Graça J, Santos S (2007) Suberin: a biopolyester of plants’ skin. Macromol Biosci 7:128–135. https://doi.org/10.1002/mabi.200600218
Gray-Mitsumune M, Mellerowicz EJ, Abe H, Schrader J, Winzéll A, Sterky F, Blomqvist K, McQueen-Mason S, Teeri TT, Sundberg B (2004) Expansins abundant in secondary xylem belong to subgroup A of the alpha-expansin gene family. Plant Physiol 135:1552–1564. https://doi.org/10.1104/pp.104.039321
Groover AT, Mansfield SD, DiFazio SP, Dupper G, Fontana JR, Millar R et al (2006) The Populus homeobox gene ARBORKNOX1 reveals overlapping mechanisms regulating the shoot apical meristem and the vascular cambium. Plant Mol Biol 61:917–932. https://doi.org/10.1007/s11103-006-0059-y
Hirakawa Y, Kondo Y, Fukuda H (2010) TDIF peptide signaling regulates vascular stem cell proliferation via the WOX4 homeobox gene in Arabidopsis. Plant Cell 22:2618–2629. https://doi.org/10.1105/tpc.110.076083
Höfer R, Briesen I, Beck M, Pinot F, Schreiber L, Franke R (2008) The Arabidopsis cytochrome P450 CYP86A1 encodes a fatty acid -hydroxylase involved in suberin monomer biosynthesis. J Exp Bot 59:2347–2360. https://doi.org/10.1093/jxb/ern101
Holloway PJ (1983) Some variations in the composition of suberin from the cork layers of higher plants. Phytochemistry 22:495–502. https://doi.org/10.1016/0031-9422(83)83033-7
Howard ET (1977) Bark structure of southern upland oaks. Wood Fiber 9:172–183
Irish VF, Sussex IM (1990) Function of the apetala-1 gene during Arabidopsis floral development. Plant Cell 2:741–753. https://doi.org/10.1105/tpc.2.8.741
Ji J, Shimizu R, Sinha N, Scanlon MJ (2010) Analyses of WOX4 transgenics provide further evidence for the evolution of the WOX gene family during the regulation of diverse stem cell functions. Plant Signal Behav 5:916–920. https://doi.org/10.1104/pp.109.149641
Jin H, Cominelli E, Bailey P, Parr A, Mehrtens F, Jones J et al (2000) Transcriptional repression by AtMYB4 controls production of UV-protecting sunscreens in Arabidopsis. EMBO J 19:6150–6161. https://doi.org/10.1093/emboj/19.22.6150
Jung JH, Park JH, Lee S, To TK, Kim JM, Seki M et al (2013) The cold signaling attenuator HIGH EXPRESSION OF OSMOTICALLY RESPONSIVE GENE1 activates FLOWERING LOCUS C transcription via chromatin remodeling under short-term cold stress in Arabidopsis. Plant Cell 25:4378–4390. https://doi.org/10.1105/tpc.113.118364
Klahre U, Noguchi T, Fujioka S, Takatsuto S, Yokota T, Nomura T et al (1998) The Arabidopsis DIMINUTO/DWARF1 gene encodes a protein involved in steroid synthesis. Plant Cell 10:1677–1690. https://doi.org/10.1105/tpc.10.10.1677
Kosma DK, Murmu J, Razeq FM, Santos P, Bourgault R, Molina I et al (2014) AtMYB41 activates ectopic suberin synthesis and assembly in multiple plant species and cell types. The Plant J 80:216–229. https://doi.org/10.1111/tpj.12624
Lashbrooke J, Cohen H, Levy-Samocha D, Tzfadia O, Panizel I, Zeisler V et al (2016) MYB107 and MYB9 homologs regulate suberin deposition in angiosperms. Plant Cell 28:2097–2116. https://doi.org/10.1105/tpc.16.00490
Legay S, Sivadon P, Blervacq AS, Pavy N, Baghdady A, Tremblay L et al (2010) EgMYB1, an R2R3 MYB transcription factor from eucalyptus negatively regulates secondary cell wall formation in Arabidopsis and poplar. New Phytol 188:774–786. https://doi.org/10.1111/j.1469-8137.2010.03432.x
Legay S, Guerriero G, Deleruelle A, Lateur M, Evers D, André CM, Hausman J-F (2015) Apple russeting as seen through the RNA-seq lens: strong alterations in the exocarp cell wall. Plant Mol Biol 88:21–40. https://doi.org/10.1007/s11103-015-0303-4
Legay S, Guerriero G, André C, Guignard C, Cocco E, Charton S et al (2016) MdMyb93 is a regulator of suberin deposition in russeted apple fruit skins. New Phytol 212:977–991. https://doi.org/10.1111/nph.14170
Lendzian KJ (2006) Survival strategies of plants during secondary growth: barrier properties of phellems and lenticels towards water, oxygen, and carbon dioxide. J Exp Bot 57:2535–2546. https://doi.org/10.1093/jxb/erl014
Li H, Durbin R (2010) Fast and accurate long-read alignment with Burrows-Wheeler transform. Bioinformatics 26:589–595. https://doi.org/10.1093/bioinformatics/btp698
Li W, Godzik A (2006) Cd-hit: a fast program for clustering and comparing large sets of protein or nucleotide sequences. Bioinformatics 22:1658–1659. https://doi.org/10.1093/bioinformatics/btl158
Li Y, Beisson F, Koo AJK, Molina I, Pollard M, Ohlrogge JB (2007) Identification of acyltransferases required for cutin biosynthesis and production of cutin with suberin-like monomers. Proc Natl Acad Sci USA 104:18339–18344. https://doi.org/10.1073/pnas.0706984104
Li-Beisson Y, Shorrosh B, Beisson F, Andersson MX, Arondel V, Bates PD et al (2013) Acyl-lipid metabolism. Arab B 11:e0161. https://doi.org/10.1199/tab.0161
Mardis ER (2008) Next-generation DNA sequencing methods. Annu Rev Genomics Hum Genet 9:387–402. https://doi.org/10.1146/annurev.genom.9.081307.164359
McQueen-Mason S, Cosgrove DJ (1994) Disruption of hydrogen bonding between plant cell wall polymers by proteins that induce wall extension. Proc Natl Acad Sci USA 91:6574–6578. https://doi.org/10.1073/pnas.91.14.6574
Miguel A, Milhinhos A, Novák O, Jones B, Miguel CM (2015) The SHORT-ROOT-like gene PtSHR2B is involved in Populus phellogen activity. J Exp Bot 67:1545–1555. https://doi.org/10.1093/jxb/erv547
Molina I, Kosma D (2015) Role of HXXXD-motif/BAHD acyltransferases in the biosynthesis of extracellular lipids. Plant Cell Rep 34:587–601. https://doi.org/10.1007/s00299-014-1721-5
Molina I, Li-Beisson Y, Beisson F, Ohlrogge JB, Pollard M (2009) Identification of an Arabidopsis feruloyl-coenzyme A transferase required for suberin synthesis. Plant Physiol 151:1317–1328. https://doi.org/10.1104/pp.109.144907\rpp.109.144907
Nagata N, Asami T, Yoshida S (2001) Brassinazole, an inhibitor of brassinosteroid biosynthesis, inhibits development of secondary xylem in cress plants (Lepidium sativum). Plant Cell Physiol 42:1006–1011. https://doi.org/10.1093/pcp/pce122
Nilsson J, Karlberg A, Antti H, Lopez-Vernaza M, Mellerowicz E, Perrot-Rechenmann C et al (2008) Dissecting the molecular basis of the regulation of wood formation by auxin in hybrid aspen. Plant Cell 20:843–855. https://doi.org/10.1105/tpc.107.055798
Pereira-Leal JB, Abreu IA, Alabaça CS, Almeida MH, Almeida P, Almeida T et al (2014) A comprehensive assessment of the transcriptome of cork oak (Quercus suber) through EST sequencing. BMC Genom 15:371. https://doi.org/10.1186/1471-2164-15-371
Pfaffl MW (2001) A new mathematical model for relative quantification in real-time RT-PCR. Nucleic Acids Res 29:e45
Pollard M, Beisson F, Li Y, Ohlrogge JB (2008) Building lipid barriers: biosynthesis of cutin and suberin. Trends Plant Sci 13:236–246. https://doi.org/10.1016/j.tplants.2008.03.003
Porto DD, Bruneau M, Perini P, Anzanello R, Renou JP, dos Santos HP et al (2015) Transcription profiling of the chilling requirement for bud break in apples: a putative role for FLC-like genes. J Exp Bot 66:2659–2672. https://doi.org/10.1093/jxb/erv061
Rains MK, Gardiyehewa de Silva ND, Molina I (2017) Reconstructing the suberin pathway in poplar by chemical and transcriptomic analysis of bark tissues. Tree Physiol 1:1–22. https://doi.org/10.1093/treephys/tpx060
Reinbothe C, Springer A, Samol I, Reinbothe S (2009) Plant oxylipins: role of jasmonic acid during programmed cell death, defence and leaf senescence. FEBS J 276:4666–4681. https://doi.org/10.1111/j.1742-4658.2009.07193.x
Ricardo CPP, Martins I, Francisco R, Sergeant K, Pinheiro C, Campos A et al (2011) Proteins associated with cork formation in Quercus suber L. stem tissues. J Proteomics 74:1266–1278. https://doi.org/10.1016/j.jprot.2011.02.003
Schreiber L, Franke R, Hartmann K (2005) Wax and suberin development of native and wound periderm of potato (Solanum tuberosum L.) and its relation to peridermal transpiration. Planta 220:520–530. https://doi.org/10.1007/s00425-004-1364-9
Serra O, Soler M, Hohn C, Franke R, Schreiber L, Prat S et al (2009) Silencing of StKCS6 in potato periderm leads to reduced chain lengths of suberin and wax compounds and increased peridermal transpiration. J Exp Bot 60:697–707. https://doi.org/10.1093/jxb/ern314
Serra O, Hohn C, Franke R, Prat S, Molinas M, Figueras M (2010) A feruloyl transferase involved in the biosynthesis of suberin and suberin-associated wax is required for maturation and sealing properties of potato periderm. Plant J 62:277–290. https://doi.org/10.1111/j.1365-313X.2010.04144.x
Sheldon CC, Rouse DT, Finnegan EJ, Peacock WJ, Dennis ES (2000) The molecular basis of vernalization: the central role of FLOWERING LOCUS C (FLC). Proc Natl Acad Sci USA 97:3753–3758. https://doi.org/10.1073/pnas.97.7.3753
Shindo C, Lister C, Crevillen P, Nordborg M, Dean C (2006) Variation in the epigenetic silencing of FLC contributes to natural variation in Arabidopsis vernalization response. Genes Dev 20:3079–3083. https://doi.org/10.1101/gad.405306
Sibout R, Plantegenet S, Hardtke CS (2008) Flowering as a condition for xylem expansion in Arabidopsis hypocotyl and root. Curr Biol 18:458–463. https://doi.org/10.1016/j.cub.2008.02.070
Silva SP, Sabino MA, Fernandes EM, Correlo VM, Boesel LF, Reis RL (2005) Cork: properties, capabilities and applications. Int Mater Rev 50:345–365. https://doi.org/10.1179/174328005X41168
Soler M, Serra O, Molinas M, Huguet G, Fluch S, Figueras M (2007) A genomic approach to suberin biosynthesis and cork differentiation. Plant Physiol 144:419–431. https://doi.org/10.1104/pp.106.094227
Soler M, Serra O, Molinas M, García-Berthou E, Caritat A, Figueras M (2008) Seasonal variation in transcript abundance in cork tissue analyzed by real time RT-PCR. Tree Physiol 28:743–751. https://doi.org/10.1093/treephys/28.5.743
Soler M, Serra O, Fluch S, Molinas M, Figueras M (2011) A potato skin SSH library yields new candidate genes for suberin biosynthesis and periderm formation. Planta 233:933–945. https://doi.org/10.1007/s00425-011-1350-y
Soler M, Plasencia A, Larbat R, Pouzet C, Jauneau A, Rivas S et al (2017) The Eucalyptus linker histone variant EgH1.3 cooperates with the transcription factor EgMYB1 to control lignin biosynthesis during wood formation. New Phytol 213:287–299. https://doi.org/10.1111/nph.14129
Suer S, Agusti J, Sanchez P, Schwarz M, Greb T (2011) WOX4 imparts auxin responsiveness to cambium cells in Arabidopsis. Plant Cell 23:3247–3259. https://doi.org/10.1105/tpc.111.087874
Teixeira RT, Fortes AM, Pinheiro C, Pereira H (2014) Comparison of good- and bad-quality cork: application of high-throughput sequencing of phellogenic tissue. J Exp Bot 65:4887–4905. https://doi.org/10.1093/jxb/eru252
Teixeira RT, Fortes AM, Bai H, Pinheiro C, Pereira H (2017) Transcriptional profiling of cork oak phellogenic cells isolated by laser microdissection. Planta. https://doi.org/10.1007/s00425-017-2786-5
Thimm O, Bläsing O, Gibon Y, Nagel A, Meyer S, Krüger P et al (2004) MAPMAN: a user-driven tool to display genomics data sets onto diagrams of metabolic pathways and other biological processes. Plant J 37:914–939. https://doi.org/10.1111/j.1365-313X.2004.02016.x
Troncoso-Ponce MA, Kilaru A, Cao X, Durrett TP, Fan J, Jensen JK, Thrower NA, Pauly M, Wilkerson C, Ohlrogge JB (2011) Comparative deep transcriptional profiling of four developing oilseeds. Plant J 68:1014–1027. https://doi.org/10.1111/j.1365-313X.2011.04751.x
Tuominen H, Puech L, Fink S, Sundberg B (1997) A radial concentration gradient of indole-3-acetic acid is related to secondary xylem development in hybrid aspen. Plant Physiol 115:577–585. https://doi.org/10.1104/PP.115.2.577
Verdaguer R, Soler M, Serra O, Garrote A, Fernández S, Company-Arumí D, Anticó E, Molinas M, Figueras M (2016) Silencing of the potato StNAC103 gene enhances the accumulation of suberin polyester and associated wax in tuber skin. J Exp Bot 67:5415–5427. https://doi.org/10.1093/jxb/erw305
Vishwanath SJ, Delude C, Domergue F, Rowland O (2015) Suberin: biosynthesis, regulation, and polymer assembly of a protective extracellular barrier. Plant Cell Rep 34:573–586. https://doi.org/10.1007/s00299-014-1727-z
Waisel Y (1995) Developmental and functional aspects of the periderm. In: Iqbal M (ed) The cambial derivatives. Gebruder Borntraeger, Stuttgart, pp 293–315
Wang X, Cnops G, Vanderhaeghen R, De Block S, Van Montagu M, Van Lijsebettens M (2001) AtCSLD3, a cellulose synthase-like gene important for root hair growth in Arabidopsis. Plant Physiol 126:575–586. https://doi.org/10.1104/pp.126.2.575
Wang L, Feng Z, Wang X, Wang X, Zhang X (2009) DEGseq: an R package for identifying differentially expressed genes from RNA-seq data. Bioinformatics 26:136–138. https://doi.org/10.1093/bioinformatics/btp612
Wang Z, Gerstein M, Snyder M (2010) RNA-Seq: a revolutionary tool for transcriptomics. Nat Rev Genet 10:57–63. https://doi.org/10.1038/nrg2484
Wang YZ, Zhang S, Dai MS, Shi ZB (2014) Pigmentation in sand pear (Pyrus pyrifolia) fruit: biochemical characterization, gene discovery and expression analysis with exocarp pigmentation mutant. Plant Mol Biol 85:123 –134. https://doi.org/10.1007/s11103-014-0173-1
Watanabe N, Lam E (2006) Arabidopsis Bax inhibitor-1 functions as an attenuator of biotic and abiotic types of cell death. Plant J 45:884–894. https://doi.org/10.1111/j.1365-313X.2006.02654.x
Wei Z, Qu Z, Zhang L, Zhao S, Bi Z, Ji X et al (2015) Overexpression of poplar xylem sucrose synthase in tobacco leads to a thickened cell wall and increased height. PLoS ONE 10:1–20. https://doi.org/10.1371/journal.pone.0120669
Zhong R, Ye ZH (2001) Alteration of auxin polar transport in the Arabidopsisifl1 mutants. Plant Physiol 126:549–563. https://doi.org/10.1104/pp.126.2.549
Acknowledgements
We would like to thank Professor M. Molinas (Departament de Biologia, UdG, Girona) for her useful advice and feedback during the analysis of the results and the drafting of the manuscript. The authors are grateful to Dr. R. Verdaguer, S. Fernández, S. Gómez and N. Salvatella for their help in cork harvesting. We thank Professor C. Pla (Departament de Biologia, UdG, Girona) for kindly lending the Thermocycler and Mr J. Blavia and Ms C. Carulla (Serveis Tècnics de Recerca, Universitat de Girona, Spain) for their highly skilled work with SEM. This work was supported by the Ministerio de Innovación y Ciencia [AGL2009-13745, FPI grant to P.B.], the Ministerio de Economía y Competitividad and FEDER funding [AGL2012-36725; AGL2015-67495-C2-1-R]. J.A.P.P. acknowledges the European Union’s Seventh Framework Programme for research, technological development and demonstration (EU FP7 Agreement No. 621321) and the Polish financial sources for education (2015–2019) allocated to Project No (W26/7.PR/2015).
Author information
Authors and Affiliations
Contributions
PB, OS, JP and MF designed the experiment; PB extracted the RNA and purified the mRNA; PB, CH and CN performed bioinformatics; PB and AS performed qPCR; all authors analyzed and discussed the data. PB, MS, OS and MF wrote the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
11103_2017_682_MOESM7_ESM.xlsx
Table S6. Classification in accordance with Arabidopsis databases for carbohydrates and acyl-lipid metabolism (XLSX 54 KB)
Rights and permissions
About this article
Cite this article
Boher, P., Soler, M., Sánchez, A. et al. A comparative transcriptomic approach to understanding the formation of cork. Plant Mol Biol 96, 103–118 (2018). https://doi.org/10.1007/s11103-017-0682-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11103-017-0682-9