Abstract
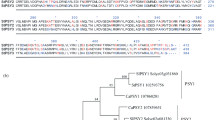
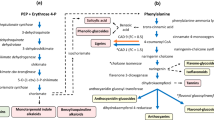
Isoprene is a volatile 5-carbon hydrocarbon derived from the chloroplastic methylerythritol 2-C-methyl-d-erythritol 4-phosphate isoprenoid pathway. In plants, isoprene emission is controlled by the enzyme isoprene synthase; however, there is still relatively little known about the genetics and regulation of this enzyme. Isoprene synthase gene structure was analysed in three poplar species. It was found that genes encoding stromal isoprene synthase exist as a small gene family, the members of which encode virtually identical proteins and are differentially regulated. Accumulation of isoprene synthase protein is developmentally regulated, but does not differ between sun and shade leaves and does not increase when heat stress is applied. Our data suggest that, in mature leaves, isoprene emission rates are primarily determined by substrate (dimethylallyl diphosphate, DMADP) availability. In immature leaves, where isoprene synthase levels are variable, emission levels are also influenced by the amount of isoprene synthase protein. No thylakoid isoforms could be identified in Populus alba or in Salix babylonica. Together, these data show that control of isoprene emission at the genetic level is far more complicated than previously assumed.





Similar content being viewed by others
References
Affek HP, Yakir D (2002) Protection by isoprene against singlet oxygen in leaves. Plant Physiol 129:269–277
Brilli F, Barta C, Fortunati A, Centritto M, Loreto F (2006) The contribution of different carbon sources to isoprene synthesis during and after drought in Populus alba leaves. Geophys Res Abs 8:EGU06-A-04113
Brüggemann N, Schnitzler JP (2002) Diurnal variation of dimethylallyl diphosphate concentrations in oak (Quercus robur) leaves. Physiol Plant 115:190–196
Chameides WL, Lindsay RW, Richardson J, Kiang CS (1998) The role of biogenic hydrocarbons in urban photochemical smog: Atlanta as a case study. Science 241:1473–1475
Cinege G, Louis S, Hänsch R, Schnitzler JP (2008) Regulation of isoprene synthase promoter by environmental and internal factors. Plant Mol Biol
Claeys M, Graham B, Vas G, Wang W, Vermeylen R, Pashynska V, Cafmeyer J, Guyon P, Andreae MO, Artaxo P, Maenhaut W (2004) Formation of secondary organic aerosols through photooxidation of isoprene. Science 303:1173–1176
Fortunati A, Barta C, Brilli F, Centritto M, Zimmer I, Schnitzler JP, Loreto F (2008) Isoprene emission is not temperature-dependent during and after severe drought-stress: a physiological and biochemical analysis. Plant J 55:687–697
Frohman MA, Dush MK, Martin GR (1988) Rapid production of full-length cDNAs from rare transcripts: amplification using a single gene-specific oligonucleotide primer. Proc Natl Acad Sci USA 85:8998–9002
Geiss-Friedlander R, Melchior F (2007) Concepts in sumoylation: a decade on. Nat Rev Mol Cell Biol 8:947–956
Granier C, Pétron G, Müller JF, Brasseur G (2000) The impact of natural and anthropogenic hydrocarbons on the tropospheric budget of carbon monoxide. Atmos Environ 34:5255–5270
Guenther A, Hewitt CN, Erickson D, Fall R, Geron C, Graedel T, Harley P, Klinger L, Lerdau M, McKay WA, Pierce T, Scholes B, Steinbrecher R, Tallamraju R, Taylor J, Zimmerman P (1995) A global model of natural volatile organic compound emissions. J Geophys Res 100:8873–8892
Guenther A, Karl T, Harley P, Wiedinmyer C, Palmer PI, Geron C (2006) Estimates of global terrestrial isoprene emissions using MEGAN (Model of Emissions of Gases and Aerosols from Nature). Atmos Chem Phys 6:3181–3210
Hannich JT, Lewis A, Kroetz MB, Li SJ, Heide H, Emili A, Hochstrasser M (2005) Defining the SUMO-modified proteome by multiple approaches in Saccharomyces cerevisiae. J Biol Chem 280:4102–4110
Harley P, Guenther A, Zimmerman P (1996) Effects of light, temperature and canopy position on net photosynthesis and isoprene emission from sweetgum (Liquidambar styraciflua) leaves. Tree Physiol 16:25–32
Hayward S, Tani A, Hewitt CN (2002) Performance characteristics of a proton transfer reaction-mass spectrometer for measuring volatile organic compounds. Environ Sci Technol 36:1560
Hewitt CN, Hayward S, Tani A (2003) Application of proton transfer reaction mass spectrometry for the monitoring and measurement of volatile organic compounds in the atmosphere. J Environ Monitor 5:1–7
Kreuzwieser J, Graus M, Wisthaler A, Hansel A, Rennenberg H, Schnitzler JP (2002) Xylem-transported glucose as an additional carbon source for leaf isoprene formation in Quercus robur. New Phytol 156:171–178
Laemmli UK (1970) Cleavage of structural proteins during assembly of head of bacteriophage-T4. Nature 227:680–685
Landry LG, Pell EJ (1993) Modification of Rubisco and altered proteolytic activity in O3-stressed hybrid poplar (Populus maximowizii × trichocarpa). Plant Physiol 101:1355–1362
Laothawornkitkul J, Paul ND, Vickers CE, Possell M, Taylor JE, Mullineaux PM, Hewitt CN (2008) Isoprene emissions influence herbivore feeding decisions. Plant Cell Environ 31:1410–1415
Laothawornkitkul J, Taylor JE, Paul ND, Hewitt CN (2009) Biogenic volatile organic compounds in the Earth system. New Phytol 183:27–51
Lehning A, Zimmer I, Steinbrecher R, Brüggemann N, Schnitzler J-P (1999) Isoprene synthase activity and its relation to isoprene emission in Quercus robur L. leaves. Plant Cell Environ 22:495–504
Lexer C, Fay MF, Joseph JA, Nica MS, Heinze B (2005) Barrier to gene flow between two ecologically divergent Populus species, P. alba (white poplar) and P. tremula (European aspen): the role of ecology and life history in gene introgression. Mol Ecol 14:1045–1057
Litvak ME, Loreto F, Harley PC, Sharkey TD, Monson RK (1996) The response of isoprene emission rate and photosynthetic rate to photon flux and nitrogen supply in aspen and white oak trees. Plant Cell Environ 19:549–559
Loivamäki M, Louis S, Cinege G, Zimmer I, Fischbach RJ, Schnitzler JP (2006) Circadian rhythms of isoprene biosynthesis in Grey Poplar leaves. Plant Physiol 106
Loivamäki M, Mumm R, Dicke M, Schnitzler JP (2008) Isoprene interferes with the attraction of bodyguards by herbaceous plants. Proc Natl Acad Sci USA 104:17430–17435
Loreto F, Delfine S (2000) Emission of isoprene from salt-stressed Eucalyptus globulus leaves. Plant Physiol 123:1605–1610
Loreto F, Velikova V (2001) Isoprene produced by leaves protects the photosynthetic apparatus against ozone damage, quenches ozone products, and reduces lipid peroxidation of cellular membranes. Plant Physiol 127:1781–1787
Loreto F, Mannozzi M, Maris C, Nascetti P, Ferranti F, Pasqualini S (2001) Ozone quenching properties of isoprene and its antioxidant role in leaves. Plant Physiol 126:993–1000
Magel E, Mayrhofer S, Müller A, Zimmer I, Hampp R, Schnitzler J-P (2006) Photosynthesis and substrate supply for isoprene biosynthesis in poplar leaves. Atmos Environ 40(S1):138–151
Mayrhofer S, Teuber M, Zimmer I, Louis S, Fischbach RJ, Schnitzler JP (2005) Diurnal and seasonal variation of isoprene biosynthesis-related genes in Grey Poplar leaves. Plant Physiol 139:474–484
Mazumder B, Seshadri V, Fox PL (2007) Translational control by the 3′ UTR: the ends specify the means. Trends Biochem Sci 28:91–98
Mignone F, Gissi C, Liuni S, Pesole G (2002) Untranslated regions of mRNAs. Genome Biol 3:0004.1-0004.10
Miller B, Oschinski C, Zimmer W (2001) First isolation of an isoprene synthase gene from poplar and successful expression of the gene in Escherichia coli. Planta 213:483–487
Noctor G, Arisi AC, Jouanin L, Foyer CH (1998) Manipulation of glutathione and amino acid biosynthesis in the chloroplast. Plant Physiol 118:471–482
Pierce T, Geron C, Bender L, Dennis R, Tonnesen G, Guenther A (1998) Influence of increased isoprene emissions on regional ozone modeling. J Geophys Res-Atmos 103:25611–25629
Poisson N, Kanakidou M, Crutzen PJ (2000) Impact of non-methane hydrocarbons on tropospheric chemistry and the oxidizing power of the global troposphere: 3-dimensional modelling results. J Atmos Chem 36:157–230
Rajora OP (1999) Genetic biodiversity impacts of silvicultural practices and phenotypic selection in white spruce. Theor Appl Genet 99:954–961
Rajora OP, Dancik BP (1992) Genetic characterization and relationships of Populus alba, P. tremula, and P. × canescens, and their clones. Theor Appl Genet V84:291–298
Rajora O, Rahman M (2003) Microsatellite DNA and RAPD fingerprinting, identification and genetic relationships of hybrid poplar (Populus × canadensis) cultivars. Theor Appl Genet 106:470–477
Rasulov B, Copolovici L, Laisk A, Niinemets U (2009) Postillumination isoprene emission: in vivo measurements of dimethylallyldiphosphate pool size and isoprene synthase kinetics in aspen leaves. Plant Physiol 108
Ratna WN, Oyeamalu C (2002) The upstream stem-loop domain of the 3′ untranslated region of apolipoprotein II mRNA binds the estrogen-regulated mRNA stabilizing factor. J Steroid Biochem Mol Biol 80:383–393
Rosenstiel TN, Fisher AJ, Fall R, Monson RK (2002) Differential accumulation of dimethylallyl diphosphate in leaves and needles of isoprene- and methylbutenol-emitting and nonemitting species. Plant Physiol 129:1276–1284
Sambrook J, Fritsch EF Maniatis, Maniatis T (1989) Molecular Cloning: a laboratory manual. Cold Spring Harbour Laboratory Press, New York
Sasaki K, Ohara K, Yazaki K (2005) Gene expression and characterization of isoprene synthase from Populus alba. FEBS Lett 579:2514–2518
Schaaf MJM, Cidlowski JA (2002) AUUUA motifs in the 3′ UTR of human glucocorticoid receptor alpha and beta mRNA destabilize mRNA and decrease receptor protein expression. Steroids 67:627–636
Schnitzler JP, Graus M, Kreuzwieser J, Heizmann U, Rennenberg H, Wisthaler A, Hansel A (2004) Contribution of different carbon sources to isoprene biosynthesis in poplar leaves. Plant Physiol 135:152–160
Schnitzler JP, Zimmer I, Bachl A, Arend M, Fromm J, Fischbach RJ (2005) Biochemical properties of isoprene synthase in poplar (Populus × canescens). Planta 1-10
Shallcross DE, Monks PS (2000) New Directions: a role for isoprene in biosphere-climate-chemistry feedbacks. Atmos Environ 34:1659–1660
Sharkey TD (1996) Isoprene synthesis by plants and animals. Endeavour 20:74–78
Sharkey TD, Loreto F (1993) Water stress, temperature, and light effects on the capacity for isoprene emission and photosynthesis of kudzu leaves. Oecologia 95:328–333
Sharkey TD, Singsaas EL (1995) Why plants emit isoprene. Nature 374:769
Sharkey TD, Yeh S (2001) Isoprene emission from plants. Annu Rev Plant Phys Plant Mol Biol 52:407–436
Sharkey TD, Loreto F, Delwiche CF (1991) High carbon dioxide and sun/shade effects on isoprene emission from oak and aspen tree leaves. Plant Cell Environ 14:333–338
Sharkey TD, Yeh S, Wiberley AE, Falbel TG, Gong D, Fernandez DE (2005) Evolution of the isoprene biosynthetic pathway in kudzu. Plant Physiol 137:700–712
Silver GM, Fall R (1991) Enzymatic synthesis of isoprene from dimethylallyl diphosphate in aspen leaf extracts. Plant Physiol 97:1588–1591
Silver GM, Fall R (1995) Characterization of aspen isoprene synthase, an enzyme responsible for leaf isoprene emission to the atmosphere. J Biol Chem 270:13010–13016
Singsaas EL, Lerdau M, Winter K, Sharkey TD (1997) Isoprene increases thermotolerance of isoprene-emitting species. Plant Physiol 115:1413–1420
Siwko ME, Marrink SJ, de Vries AH, Kozubek A, Schoot Uiterkamp AJ, Mark AE (2007) Does isoprene protect plant membranes from thermal shock? A molecular dynamics study. Biochem Biophys Acta 1768:198–206
Terry GN, Stokes NJ, Hewitt CN, Mansfield TA (1995) Exposure to isoprene promotes flowering in plants. J Exp Bot 46:1629–1631
Thompson AM (1992) The oxidizing capacity of the earth’s atmosphere—probable past and future changes. Science 256:1157–1165
Trainer M, Williams EJ, Parrish DD, Buhr MP, Allwine EJ, Westberg HH, Fehsenfeld FC, Liu SC (1987) Models and observations of the impact of natural hydrocarbons on rural ozone. Nature 329:705–707
Tuskan GA, DiFazio S, Jansson S, Bohlmann J, Grigoriev I, Hellsten U, Putnam N, Ralph S, Rombauts S, Salamov A, Schein J, Sterck L, Aerts A, Bhalerao RR, Bhalerao RP, Blaudez D, Boerjan W, Brun A, Brunner A, Busov V, Campbell M, Carlson J, Chalot M, Chapman J, Chen GL, Cooper D, Coutinho PM, Couturier J, Covert S, Cronk Q, Cunningham R, Davis J, Degroeve S, Dejardin A, dePamphilis C, Detter J, Dirks B, Dubchak I, Duplessis S, Ehlting J, Ellis B, Gendler K, Goodstein D, Gribskov M, Grimwood J, Groover A, Gunter L, Hamberger B, Heinze B, Helariutta Y, Henrissat B, Holligan D, Holt R, Huang W, Islam-Faridi N, Jones S, Jones-Rhoades M, Jorgensen R, Joshi C, Kangasjarvi J, Karlsson J, Kelleher C, Kirkpatrick R, Kirst M, Kohler A, Kalluri U, Larimer F, Leebens-Mack J, Leple JC, Locascio P, Lou Y, Lucas S, Martin F, Montanini B, Napoli C, Nelson, Nelson C, Nieminen K, Nilsson O, Pereda V, Peter G, Philippe R, Pilate G, Poliakov A, Razumovskaya J, Richardson P, Rinaldi C, Ritland K, Rouze P, Ryaboy D, Schmutz J, Schrader J, Segerman B, Shin H, Siddiqui A, Sterky F, Terry A, Tsai CJ, Uberbacher E, Unneberg P, Vahala J, Wall K, Wessler S, Yang G, Yin T, Douglas C, Marra M, Sandberg G, Van de Peer Y, Rokhsar D (2006) The genome of black cottonwood, Populus trichocarpa (Torr. & Gray). Science 313:1596–1604
Vickers CE, Possell M, Cojocariu C, Velikova V, Laothawornkitkul J, Ryan A, Mullineaux PM, Hewitt CN (2009a) Isoprene synthesis protects transgenic plants from oxidative stress. Plant Cell Environ 32:520–531
Vickers CE, Gershenzon J, Lerdau MT, Loreto F (2009b) A unified mechanism of action for volatile isoprenoids in plant abiotic stress. Nat Chem Biol 5:283–291
Walter MH, Hans J, Strack D (2002) Two distantly related genes encoding 1-deoxy-d-xylulose 5-phosphate synthases: differential regulation in shoots and apocarotenoid-accumulating mycorrhizal roots. Plant J 31:243–254
Wang KY, Shallcross DE (2000) Modelling terrestrial biogenic isoprene fluxes and their potential impact on global chemical species using a coupled LSM-CTM model. Atmos Environ 34:2909–2925
Wei Y, Lee JM, Richmond C, Blattner FR, Rafalski JA, LaRossa RA (2001) High-density microarray-mediated gene expression profiling of Escherichia coli. J Bacteriol 183:545–556
Wiberley AE, Linskey AR, Falbel TG, Sharkey TD (2005) Development of the capacity for isoprene emission in kudzu. Plant Cell Environ 28:898–905
Wiberley AE, Donohue AR, Westphal MM, Sharkey TD (2009) Regulation of isoprene emission from poplar leaves throughout a day. Plant Cell Environ 32:939–947
Wildermuth MC, Fall R (1996) Light-dependent isoprene emission (characterization of a thylakoid-bound isoprene synthase in Salix discolor chloroplasts). Plant Physiol 112:171–182
Wildermuth MC, Fall R (1998) Biochemical characterization of stromal and thylakoid-bound isoforms of isoprene synthase in willow leaves. Plant Physiol 116:1111–1123
Wilkie GS, Dickson KS, Gray NK (2003) Regulation of mRNA translation by 5′- and 3′-UTR-binding factors. Trends Biochem Sci 28:182–188
Acknowledgments
This research was funded by the UK Biotechnology and Biological Sciences Research Council (BBSRC) grant number BBS/B/12172. The full-length anti-isoprene synthase antibody was kindly provided by Dr Jörg-Peter Schnitzler, IMK-IFU, Garmisch, Germany. We thank Dr Oksana Zakhleniuk and Mr Daniel Exton for skilled technical assistance.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Vickers, C.E., Possell, M., Nicholas Hewitt, C. et al. Genetic structure and regulation of isoprene synthase in Poplar (Populus spp.). Plant Mol Biol 73, 547–558 (2010). https://doi.org/10.1007/s11103-010-9642-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11103-010-9642-3