Abstract
Objective
To identify risk factors for the development of postoperative diabetes insipidus (DI) in a modern cohort of endoscopic endonasal transsphenoidal surgery.
Methods
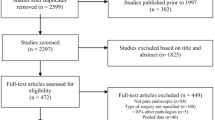
Analysis of prospectively collected data of 449 consecutive patients operated on for anterior skull base pathology. DI was defined as a polyuria (> 250 ml/h for ≥ 2 consecutive hours) polydipsia syndrome associated with hypotonic urine with or without hypernatraemia. Multivariate logistic regression was used to identify predictors of postoperative DI. A simple scoring system was then created.
Results
Postoperative DI occurred in 46 (10.2%) patients. The development of DI did not affect quality of life. Predictors of DI on multivariate analysis included suprasellar extension (OR 2.2; p = 0.04), age < 50 years (OR 2.8; p = 0.003), craniopharyngioma histology (OR 6.7; p = 0.002), and Kelly grade 3 intraoperative CSF leak (OR 2.1; p = 0.04). The SALT score was created based on these characteristics, with one point awarded for each feature present, and predicted DI with fair to good predictive value in our cohort (AUROC 0.735 (95%CI 0.65–0.82)). The rates of postoperative DI were 4.0%, 6.5%, 15.0%. 36.8% and 85.7% for SALT scores of zero, one, two, three, and four, respectively.
Conclusions
The SALT score predicts postoperative DI with fair to good accuracy, and now requires prospective external validation. Improved prediction of DI could optimize resource allocation and facilitate individualised preoperative patient counselling. We also provide our algorithm for diagnosis and treatment of DI.




Similar content being viewed by others
References
Hendricks BL, Shikary TA, Zimmer LA (2015) Causes for 30-day readmission following transsphenoidal surgery. Otolaryngol Neck Surg. https://doi.org/10.1177/0194599815617130
Parasher AK, Lerner DK, Glicksman JT et al (2019) In-hospital costs associated with diabetes insipidus following pituitary surgery. J Neurol Surg B
Ammirati M, Wei L, Ciric I (2013) Short-term outcome of endoscopic versus microscopic pituitary adenoma surgery: a systematic review and meta-analysis. J Neurol Neurosurg Psychiatry 84:843–849. https://doi.org/10.1136/jnnp-2012-303194
Burke WT, Cote DJ, Penn DL et al (2020) Diabetes insipidus after endoscopic transsphenoidal surgery. Neurosurgery 87:949–955. https://doi.org/10.1093/neuros/nyaa148
Nayak P, Montaser AS, Hu J et al (2018) Predictors of postoperative diabetes insipidus following endoscopic resection of pituitary adenomas. J Endocr Soc 2:1010–1019. https://doi.org/10.1210/js.2018-00121
Sigounas DG, Sharpless JL, Cheng DML et al (2008) Predictors and incidence of central diabetes insipidus after endoscopic pituitary surgery. Neurosurgery 62:71–79. https://doi.org/10.1227/01.NEU.0000311063.10745.D8
Schreckinger M, Walker B, Knepper J et al (2013) Post-operative diabetes insipidus after endoscopic transsphenoidal surgery. Pituitary 16:445–451. https://doi.org/10.1007/s11102-012-0453-1
Ajlan AM, Abdulqader SB, Achrol AS et al (2018) Diabetes insipidus following endoscopic transsphenoidal surgery for pituitary adenoma. J Neurol Surg Part B Skull Base 79:117–122
Aho CJ, Liu C, Zelman V et al (2005) Surgical outcomes in 118 patients with Rathke cleft cysts. J Neurosurg 102:189–193. https://doi.org/10.3171/jns.2005.102.2.0189
Park HR, Kshettry VR, Farrell CJ et al (2017) Clinical outcome after extended endoscopic endonasal resection of craniopharyngiomas: two-institution experience. World Neurosurg 103:465–474. https://doi.org/10.1016/j.wneu.2017.04.047
Nemergut EC, Zuo Z, Jane JAJ, Laws ERJ (2005) Predictors of diabetes insipidus after transsphenoidal surgery: a review of 881 patients. J Neurosurg 103:448–454. https://doi.org/10.3171/jns.2005.103.3.0448
Woods C, Thompson CJ (2008) Risk of diabetes insipidus after pituitary surgery. Expert Rev Endocrinol Metab 3:23–27. https://doi.org/10.1586/17446651.3.1.23
Hensen J, Henig A, Fahlbusch R et al (1999) Prevalence, predictors and patterns of postoperative polyuria and hyponatraemia in the immediate course after transsphenoidal surgery for pituitary adenomas. Clin Endocrinol (Oxf) 50:431–439
Mikhaylova D, Dzeranova L, Yu RO, et al (2020) The course and predictors of postoperative diabetes insipidus. In: Endocrine abstracts. Bioscientifica
Agam MS, Wedemeyer MA, Wrobel B et al (2018) Complications associated with microscopic and endoscopic transsphenoidal pituitary surgery: experience of 1153 consecutive cases treated at a single tertiary care pituitary center. J Neurosurg 130:1576–1583. https://doi.org/10.3171/2017.12.JNS172318
Broersen LHA, Biermasz NR, van Furth WR et al (2018) Endoscopic vs. microscopic transsphenoidal surgery for Cushing’s disease: a systematic review and meta-analysis. Pituitary 21:524–534. https://doi.org/10.1007/s11102-018-0893-3
Little AS, Kelly DF, White WL et al (2019) Results of a prospective multicenter controlled study comparing surgical outcomes of microscopic versus fully endoscopic transsphenoidal surgery for nonfunctioning pituitary adenomas: the Transsphenoidal Extent of Resection (TRANSSPHER) Study. J Neurosurg 62:1–11. https://doi.org/10.3171/2018.11.jns181238
Sheehan JM, Sheehan JP, Douds GL, Page RB (2006) DDAVP use in patients undergoing transsphenoidal surgery for pituitary adenomas. Acta Neurochir (Wien) 148:287–91-discussion 291. https://doi.org/10.1007/s00701-005-0686-0
Melmed S (2020) Pituitary-tumor endocrinopathies. N Engl J Med 382:937–950. https://doi.org/10.1056/NEJMra1810772
Casanueva FF, Barkan AL, Buchfelder M et al (2017) Criteria for the definition of Pituitary Tumor Centers of Excellence (PTCOE): a pituitary society statement. Pituitary 20:489–498. https://doi.org/10.1007/s11102-017-0838-2
Cappabianca P, Cavallo LM, Divitiis E de (2004) Endoscopic endonasal transsphenoidal surgery. Neurosurgery 55:933–40-discussion 940–1. https://doi.org/10.1227/01.neu.0000137330.02549.0d
Gil Z, Abergel A, Spektor S et al (2004) Development of a cancer-specific anterior skull base quality-of-life questionnaire. J Neurosurg 100:813–819. https://doi.org/10.3171/jns.2004.100.5.0813
Hardy J, Vezina JL (1976) Transsphenoidal neurosurgery of intracranial neoplasm. Adv Neurol 15:261–273
Knosp E, Steiner E, Kitz K, Matula C (1993) Pituitary adenomas with invasion of the cavernous sinus space: a magnetic resonance imaging classification compared with surgical findings. Neurosurgery 33:610–7-discussion 617–8. https://doi.org/10.1227/00006123-199310000-00008
Heinbecker P, White HL (1939) The role of the pituitary gland in water balance. Ann Surg 110:1037–1049
Lipsett M, Maclean JP, West CD et al (1956) An analysis of the polyuria induced by hypophysectomy in man. J Clin Endocrinol Metab 16:183–195. https://doi.org/10.1210/jcem-16-2-183
Thrasher TN, Keil LC, Ramsay DJ (1982) Lesions of the organum vasculosum of the lamina terminalis (OVLT) attenuate osmotically-induced drinking and vasopressin secretion in the dog. Endocrinology 110:1837–1839. https://doi.org/10.1210/endo-110-5-1837
Castle-Kirszbaum M, Kyi M, Wright C et al (2021) Hyponatraemia and hypernatraemia: disorders of water balance in neurosurgery. Neurosurg Rev. https://doi.org/10.1007/s10143-020-01450-9
de Vries F, Lobatto DJ, Verstegen MJT et al (2020) Postoperative diabetes insipidus: how to define and grade this complication? Pituitary. https://doi.org/10.1007/s11102-020-01083-7
Christ-Crain M, Bichet DG, Fenske WK et al (2019) Diabetes insipidus. Nat Rev Dis Primer 5:54–20. https://doi.org/10.1038/s41572-019-0103-2
Castle-Kirszbaum M, Goldschlager T, Ho B et al (2018) Twelve cases of pituitary metastasis: a case series and review of the literature. Pituitary 21:463–473. https://doi.org/10.1007/s11102-018-0899-x
Castle-Kirszbaum M, Beng Phung T, Luen SJ et al (2020) A pituitary metastasis, an adenoma and potential hypophysitis: a case report of tumour to tumour metastasis in the pituitary. J Clin Neurosci 81:161–166. https://doi.org/10.1016/j.jocn.2020.09.033
Se I, Saito T, Fukagawa A et al (2001) Close association of urinary excretion of aquaporin-2 with appropriate and inappropriate arginine vasopressin-dependent antidiuresis in hyponatremia in elderly subjects. J Clin Endocrinol Metab 86:1665–1671. https://doi.org/10.1210/jcem.86.4.7426
Gutkowska J, Jankowski M, Mukaddam-Daher S, McCann SM (2000) Corticotropin-releasing hormone causes antidiuresis and antinatriuresis by stimulating vasopressin and inhibiting atrial natriuretic peptide release in male rats. Proc Natl Acad Sci U S A 97:483–488
Honegger J, Grimm F (2018) The experience with transsphenoidal surgery and its importance to outcomes. Pituitary 21:545–555. https://doi.org/10.1007/s11102-018-0904-4
Ciric I, Ragin A, Baumgartner C, Pierce D (1997) Complications of transsphenoidal surgery: results of a national survey, review of the literature, and personal experience. Neurosurgery 40:225–36; discussion 236–237. https://doi.org/10.1097/00006123-199702000-00001
Zaidi HA, Awad A-W, Bohl MA et al (2016) Comparison of outcomes between a less experienced surgeon using a fully endoscopic technique and a very experienced surgeon using a microscopic transsphenoidal technique for pituitary adenoma. J Neurosurg 124:596–604. https://doi.org/10.3171/2015.4.JNS15102
Balanescu S, Kopp P, Gaskill MB et al (2011) Correlation of plasma copeptin and vasopressin concentrations in hypo-, iso-, and hyperosmolar states. J Clin Endocrinol Metab 96:1046–1052. https://doi.org/10.1210/jc.2010-2499
Winzeler B, Zweifel C, Nigro N et al (2015) Postoperative copeptin concentration predicts diabetes insipidus after pituitary surgery. J Clin Endocrinol Metab 100:2275–2282. https://doi.org/10.1210/jc.2014-4527
Berton AM, Gatti F, Penner F et al (2020) Early copeptin determination allows prompt diagnosis of post-neurosurgical central diabetes insipidus. Neuroendocrinology 110:525–534. https://doi.org/10.1159/000503145
Acknowledgements
The first author is undertaking a higher degree funded by an Australian Government Research Training Program (RTP) Scholarship.
Funding
None.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conclusion
DI is common after endoscopic TSS. Predictors of postoperative DI include craniopharyngioma histology, suprasellar extension, Kelly grade 3 intraoperative CSF leak, and age < 50 years. Together, these factors form the SALT score, which predicted DI in our cohort with fair to good predictive value. It requires external validation prior to wider implementation. We also describe our algorithm for diagnosis and treatment of DI.
The authors have no potential conflicts of interest to disclose.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Castle-Kirszbaum, M., Fuller, P., Wang, Y.Y. et al. Diabetes insipidus after endoscopic transsphenoidal surgery: multicenter experience and development of the SALT score. Pituitary 24, 867–877 (2021). https://doi.org/10.1007/s11102-021-01159-y
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11102-021-01159-y