Abstract
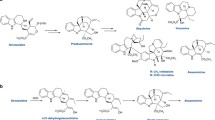
Benzylisoquinoline alkaloids (BIAs) are a group of specialized metabolites found predominantly in the plant order Ranunculales. Approximately 2500 naturally occurring BIAs have been identified, many of which possess a variety of potent biological and pharmacological properties. The initial BIA skeleton is formed via condensation by a unique enzyme, norcoclaurine synthase, of the l-tyrosine derivatives dopamine and 4-hydroxyphenylacetaldehyde, yielding (S)-norcoclaurine as a central intermediate. The vast diversity of BIA structures is subsequently derived from (1) transformation of the basic BIA backbone by oxidative enzymes, particularly cytochromes P450 and FAD-linked oxidases, and (2) further structural and functional group modification by tailoring enzymes, which also include various reductases, dioxygenases, acetyltransferases, and carboxylesterases. Most of the biosynthetic enzymes responsible for the biosynthesis of major BIAs (i.e. morphine, noscapine, papaverine, and sanguinarine) in opium poppy (Papaver somniferum), and other compounds (e.g. berberine) in related plants, have been isolated and partially characterized. Diversity in BIA metabolism is driven by the modular and repetitive recruitment, and subsequent neo-functionalization, of a limited number of ancestral enzymes. In this review, BIA biosynthetic enzymes are discussed in the context of their respective families, facilitating exploration of common phylogeny and biochemical mechanisms.











Similar content being viewed by others
Abbreviations
- 2-ODD:
-
2-Oxoglutarate-dependent dioxygenase
- 2-OG:
-
2-Oxoglutarate
- 4′OMT:
-
3′-Hydroxy-N-methylcoclaurine 4′-hydroxylase
- 4-HPAA:
-
4-Hydroxyphenylacetaldehyde
- 6OMT:
-
Norcoclaurine 6-O-methyltransferase
- 7OMT:
-
Reticuline 7-O-methyltransferase
- AKR:
-
Aldo-keto reductase
- AT1:
-
1,13-Dihydroxy-N-methylcanadine 13-O-acetyltransferase
- BBE:
-
Berbrine bridge enzyme
- BIA:
-
Benzylisoquinoline alkaloid
- BS:
-
Berbamunine synthase
- CAS:
-
Canadine synthase
- CFS:
-
Cheilanthifoline synthase
- CNMT:
-
Coclaurine N-methyltransferase
- CoA:
-
Coenzyme A
- CODM:
-
Codeine O-demethylase
- COR:
-
Codeinone reductase
- CPR:
-
Cytochrome P450 reductase
- CTS:
-
Corytuberine synthase
- CXE:
-
Carboxylesterase
- CYP:
-
Cytochrome P450
- DBOX:
-
Dihydrobenzophenanthridine oxidase
- DOPA:
-
3,4-Dihydroxyphenylalanine
- DRS:
-
1,2-Dehydroreticuline synthase
- DRR:
-
1,2-Dehydroreticuline reductase
- FAD:
-
Flavin adenine dinucleotide
- FADOX:
-
FAD-linked oxidoreductase
- MSH:
-
N-Methylstylopine 14-hydroxylase
- MT:
-
Methyltransferase
- NADPH:
-
Nicotinamide adenine dinucleotide phosphate
- N7OMT:
-
Norreticuline 7-O-methyltransferase
- NCS:
-
Norcoclaurine synthase
- NMCH:
-
(S)-N-Methylcoclaurine 3′-hydroxylase
- NMT:
-
N-Methyltransferase
- NOS:
-
Noscapine synthase
- OMT:
-
O-Methyltransferase
- P6H:
-
Protopine 6-hydroxylase
- P7ODM:
-
Papaverine 7-O-demethylase
- PavNMT:
-
Pavine N-methyltransferase
- PR10:
-
Pathogenesis-related protein 10
- PMT:
-
Putrescine N-methyltransferase
- PODA:
-
Protopine O-dealkylase
- REPI:
-
Reticuline epimerase
- RNMT:
-
Reticuline N-methyltransferase
- SalAT:
-
Salutaridinol 7-O-acetyltransferase
- SalR:
-
Salutaridine reductase
- SalSyn:
-
Salutaridine synthase
- SAM:
-
S-Adenosylmethionine
- SanR:
-
Sanguinarine reductase
- SDR:
-
Short-chain dehydrogenase/reductase
- SOMT:
-
Scoulerine 9-O-methyltransferase
- SPDS:
-
Spermidine synthase
- SPS:
-
Stylopine synthase
- STOX:
-
(S)-Tetrahydroprotoberberine oxidase
- T6ODM:
-
Thebaine 6-O-demethylase
- TIM:
-
Triosephosphate isomerase
- TNMT:
-
Tetrahydroprotoberberine N-methyltransferase
References
Agarwal P, Agarwal PK (2014) Pathogenesis related-10 proteins are small, structurally similar but with diverse role in stress signaling. Mol Biol Rep 41:599–611
Allen RS, Millgate AG, Chitty JA, Thisleton J, Miller JA, Fist AJ, Gerlach WL, Larkin PJ (2004) RNAi-mediated replacement of morphine with the nonnarcotic alkaloid reticuline in opium poppy. Nat Biotechnol 22:1559–1566
Amann M, Wanner G, Zenk MH (1986) Intracellular compartmentation of two enzymes of berberine biosynthesis in plant cell cultures. Planta 167:310–320
Amann M, Nagakura N, Zenk MH (1988) Purification and properties of (S)-tetrahydroprotoberberine oxidase from suspension-cultured cells of Berberis wilsoniae. Eur J Biochem 175:17–25
Angelova S, Buchheim M, Frowitter D, Schierhorn A, Roos W (2010) Overproduction of alkaloid phytoalexins in California poppy cells is associated with the co-expression of biosynthetic and stress-protective enzymes. Mol Plant 3:927–939
Asai S, Mase K, Yoshioka H (2010) A key enzyme for flavin synthesis is required for nitric oxide and reactive oxygen species production in disease resistance. Plant J 62:911–924
Bak S, Beisson F, Bishop G, Hamberger B, Hofer R, Paquette S, Werck-Reichhart D (2011) Cytochromes P450. Arabidopsis B 9:e0144
Ballance GM, Dixon RA (1995) Medicago sativa cDNAs encoding chalcone reductase. Plant Physiol 107:1027–1028
Barreiro EJ, Kümmerle AE, Fraga CAM (2011) The methylation effect in medicinal chemistry. Chem Rev 111:5215–5246
Bassard JE, Richert L, Geerinck J, Renault H, Duval F, Ullmann P, Schmitt M, Meyer E, Mutterer J, Boerjan W, De Jaeger G, Mely Y, Goossens A, Werck-Reichhart D (2012) Protein-protein and protein-membrane associations in the lignin pathway. Plant Cell 24:4465–4482
Beaudoin GA, Facchini PJ (2013) Isolation and characterization of a cDNA encoding (S)-cis-N-methylstylopine 14-hydroxylase from opium poppy, a key enzyme in sanguinarine biosynthesis. Biochem Biophys Res Commun 431:597–603
Bomati EK, Austin MB, Bowman ME, Dixon RA, Noel JP (2005) Structural elucidation of chalcone reductase and implications for deoxychalcone biosynthesis. J Biol Chem 280:30496–30503
Bugg TDH (2003) Dioxygenase enzymes: catalytic mechanisms and chemical models. Tetrahedron 59:7075–7101
Burbulis IE, Winkel-Shirley B (1999) Interactions among enzymes of the Arabidopsis flavonoid biosynthetic pathway. Proc Natl Acad Sci USA 96:12929–12934
Chang YC, Chang FR, Khalil AT, Hsieh PW, Wu YC (2003) Cytotoxic benzophenanthridine and benzylisoquinoline alkaloids from Argemone mexicana. Z Naturforsch C 58:521–526
Chen X, Facchini PJ (2014) Short-chain dehydrogenase/reductase catalyzing the final step of noscapine biosynthesis is localized to laticifers in opium poppy. Plant J 77:173–184
Chen M, Jin Y, Penning TM (2015a) The rate-determining steps of aldo–keto reductases (AKRs), a study on human steroid 5-β-reductase (AKR1D1). Chem Biol Interact 234:360–365
Chen X, Dang TT, Facchini PJ (2015b) Noscapine comes of age. Phytochemistry 111:7–13
Choi K-B, Morishige T, Shitan N, Yazaki K, Sato F (2002) Molecular cloning and characterization of coclaurine N-methyltransferase from cultured cells of Coptis japonica. J Biol Chem 277:830–835
Custers JH, Harrison SJ, Sela-Buurlage MB, van Deventer E, Lageweg W, Howe PW, van der Meijs PJ, Ponstein AS, Simons BH, Melchers LS, Stuiver MH (2004) Isolation and characterisation of a class of carbohydrate oxidases from higher plants, with a role in active defence. Plant J 39:147–160
D’Auria JC (2006) Acyltransferases in plants: a good time to be BAHD. Curr Opin Plant Biol 9:331–340
Dang TT, Facchini PJ (2014a) Cloning and characterization of canadine synthase involved in noscapine biosynthesis in opium poppy. FEBS Lett 588:198–204
Dang TT, Facchini PJ (2014b) CYP82Y1 is N-methylcanadine 1-hydroxylase, a key noscapine biosynthetic enzyme in opium poppy. J Biol Chem 289:2013–2026
Dang TT, Chen X, Facchini PJ (2015) Acetylation serves as a protective group in noscapine biosynthesis in opium poppy. Nat Chem Biol 11:104–106
Dastmalchi M, Bernards MA, Dhaubhadel S (2016) Twin anchors of the soybean isoflavonoid metabolon: evidence for tethering of the complex to the endoplasmic reticulum by IFS and C4H. Plant J 85:689–706
Davies NJ, Hayden RE, Simpson PJ, Birtwistle J, Mayer K, Ride JP, Bunce CM (2009) AKR1C isoforms represent a novel cellular target for jasmonates alongside their mitochondrial-mediated effects. Cancer Res 69:4769–4775
Desgagné-Penix I, Facchini PJ (2012) Systematic silencing of benzylisoquinoline alkaloid biosynthetic genes reveals the major route to papaverine in opium poppy. Plant J 72:331–344
Diaz Chavez ML, Rolf M, Gesell A, Kutchan TM (2011) Characterization of two methylenedioxy bridge-forming cytochrome P450-dependent enzymes of alkaloid formation in the Mexican prickly poppy Argemone mexicana. Arch Biochem Biophys 507:186–193
Dittrich H, Kutchan TM (1991) Molecular cloning, expression, and induction of berberine bridge enzyme, an enzyme essential to the formation of benzophenanthridine alkaloids in the response of plants to pathogenic attack. Proc Natl Acad Sci USA 88:9969–9973
El-Masry S, El-Ghazooly MG, Omar AA, Khafagy SM, Phillipson JD (1981) Alkaloids from Egyptian Papaver rhoeas. Planta Med 41:61–64
Farrow SC, Facchini PJ (2013) Dioxygenases catalyze O-demethylation and O,O-demethylenation with widespread roles in benzylisoquinoline alkaloid metabolism in opium poppy. J Biol Chem 288:28997–29012
Farrow SC, Facchini PJ (2014) Functional diversity of 2-oxoglutarate/Fe(II)-dependent dioxygenases in plant metabolism. Front Plant Sci 5:524
Farrow SC, Facchini PJ (2015) Papaverine 7-O-demethylase, a novel 2-oxoglutarate/Fe2+-dependent dioxygenase from opium poppy. FEBS Lett 589:2701–2706
Farrow SC, Hagel JM, Beaudoin GA, Burns DC, Facchini PJ (2015) Stereochemical inversion of (S)-reticuline by a cytochrome P450 fusion in opium poppy. Nat Chem Biol 11:728–732
Fernandes H, Michalska K, Sikorski M, Jaskolski M (2013) Structural and functional aspects of PR-10 proteins. FEBS J 280:1169–1199
Filling C, Berndt KD, Benach J, Knapp S, Prozorovski T, Nordling E, Ladenstein R, Jornvall H, Oppermann U (2002) Critical residues for structure and catalysis in short-chain dehydrogenases/reductases. J Biol Chem 277:25677–25684
Frick S, Kramell R, Schmidt J, Fist AJ, Kutchan TM (2005) Comparative qualitative and quantitative determination of alkaloids in narcotic and condiment Papaver somniferum cultivars. J Nat Prod 68:666–673
Gershater MC, Edwards R (2007) Regulating biological activity in plants with carboxylesterases. Plant Sci 173:579–588
Gesell A, Rolf M, Ziegler J, Diaz Chavez ML, Huang FC, Kutchan TM (2009) CYP719B1 is salutaridine synthase, the C–C phenol-coupling enzyme of morphine biosynthesis in opium poppy. J Biol Chem 284:24432–24442
Gesell A, Chavez ML, Kramell R, Piotrowski M, Macheroux P, Kutchan TM (2011) Heterologous expression of two FAD-dependent oxidases with (S)-tetrahydroprotoberberine oxidase activity from Argemone mexicana and Berberis wilsoniae in insect cells. Planta 233:1185–1197
Grimshaw CE (1992) Aldose reductase: model for a new paradigm of enzymic perfection in detoxification catalysts. Biochemistry 31:10139–10145
Grothe T, Lenz R, Kutchan TM (2001) Molecular characterization of the salutaridinol 7-O-acetyltransferase involved in morphine biosynthesis in opium poppy Papaver somniferum. J Biol Chem 276:30717–30723
Groves JT (2006) High-valent iron in chemical and biological oxidations. J Inorg Biochem 100:434–447
Hagel JM, Facchini PJ (2010) Dioxygenases catalyze the O-demethylation steps of morphine biosynthesis in opium poppy. Nat Chem Biol 6:273–275
Hagel JM, Facchini PJ (2012) Subcellular localization of sanguinarine biosynthetic enzymes in cultured opium poppy cells. In Vitro Cell Dev Biol Plant 48:233–240
Hagel JM, Facchini PJ (2013) Benzylisoquinoline alkaloid metabolism: a century of discovery and a brave new world. Plant Cell Physiol 54:647–672
Hagel JM, Beaudoin GA, Fossati E, Ekins A, Martin VJ, Facchini PJ (2012) Characterization of a flavoprotein oxidase from opium poppy catalyzing the final steps in sanguinarine and papaverine biosynthesis. J Biol Chem 287:42972–42983
Hagel JM, Morris JS, Lee EJ, Desgagné-Penix I, Bross CD, Chang L, Chen X, Farrow SC, Zhang Y, Soh J, Sensen CW, Facchini PJ (2015) Transcriptome analysis of 20 taxonomically related benzylisoquinoline alkaloid-producing plants. BMC Plant Biol 15:227
Hashimoto T, Shoji T, Mihara T, Oguri H, Tamaki K, Suzuki K, Yamada Y (1998) Intraspecific variability of the tandem repeats in Nicotiana putrescine N-methyltransferases. Plant Mol Biol 37:25–37
Higashi Y, Kutchan TM, Smith TJ (2011) Atomic structure of salutaridine reductase from the opium poppy (Papaver somniferum). J Biol Chem 286:6532–6541
Huang FC, Kutchan TM (2000) Distribution of morphinan and benzo[c]phenanthridine alkaloid gene transcript accumulation in Papaver somninferum. Phytochemistry 53:555–564
Huang CC, Smith CV, Glickman MS, Jocobs WR Jr, Sacchettini JC (2002) Crystal structures of mycolic acid cyclopropane synthases from Mycobacterium tuberculosis. J Biol Chem 277:11559–11569
Ibrahim RK, Bruneau A, Bantignies B (1998) Plant O-methyltransferases: molecular analysis, common signature and classification. Plant Mol Biol 36:1–10
Ikezawa N, Tanaka M, Nagayoshi M, Shinkyo R, Sakaki T, Inouye K, Sato F (2003) Molecular cloning and characterization of CYP719, a methylenedioxy bridge-forming enzyme that belongs to a novel P450 family, from cultured Coptis japonica cells. J Biol Chem 278:38557–38565
Ikezawa N, Iwasa K, Sato F (2008) Molecular cloning and characterization of CYP80G2, a cytochrome P450 that catalyzes an intramolecular C–C phenol coupling of (S)-reticuline in magnoflorine biosynthesis, from cultured Coptis japonica cells. J Biol Chem 283:8810–8821
Ilari A, Franceschini S, Bonamore A, Arenghi F, Botta B, Macone A, Pasquo A, Bellucci L, Boffi A (2009) Structural basis of enzymatic (S)-norcoclaurine biosynthesis. J Biol Chem 284:897–904
Jez JM, Bennett MJ, Schlegel BP, Lewis M, Penning TM (1997) Comparative anatomy of the aldo–keto reductase superfamily. Biochem J 326:625–636
Jirschitzka J, Schmidt GW, Reichelt M, Schneider B, Gershenzon J, D’Auria JC (2012) Plant tropane alkaloid biosynthesis evolved independently in the Solanaceae and Erythroxylaceae. Proc Natl Acad Sci USA 109:10304–10309
Jornvall H, Persson B, Krook M, Atrian S, Gonzalez-Duarte R, Jeffery J, Ghosh D (1995) Short-chain dehydrogenases/reductases (SDR). Biochemistry 34:6003–6013
Junker A, Fischer J, Sichhart Y, Brandt W, Drager B (2013) Evolution of the key alkaloid enzyme putrescine N-methyltransferase from spermidine synthase. Front Plant Sci 4:260
Kagan RM, Clarke S (1994) Widespread occurrence of three sequence motifs in diverse S-adenosylmethionine-dependent methyltransferases suggests a common structure for these enzymes. Arch Biochem Biophys 310:417–427
Kallberg Y, Persson B (2006) Prediction of coenzyme specificity in dehydrogenases/reductases. A hidden Markov model-based method and its application on complete genomes. FEBS J 273:1177–1184
Kanayama Y, Mori H, Imaseki H, Yamaki S (1992) Nucleotide sequence of a cDNA encoding NADP-sorbitol-6-phosphate dehydrogenase from apple. Plant Physiol 100:1607–1608
Kavanagh KL, Jornvall H, Persson B, Oppermann U (2008) Medium- and short-chain dehydrogenase/reductase gene and protein families—the SDR superfamily: functional and structural diversity within a family of metabolic and regulatory enzymes. Cell Mol Life Sci 65:3895–3906
Kawai Y, Ono E, Mizutani M (2014) Evolution and diversity of the 2-oxoglutarate-dependent dioxygenase superfamily in plants. Plant J 78:328–343
Kozbial PZ, Mushegian AR (2005) Natural history of S-adenosylmethionine-binding proteins. BMC Struct Biol 5:19
Kraus PF, Kutchan TM (1995) Molecular cloning and heterologous expression of a cDNA encoding berbamunine synthase, a C–O phenol-coupling cytochrome P450 from the higher plant Berberis stolonifera. Proc Natl Acad Sci USA 92:2071–2075
Kumar D, Klessig DF (2003) High-affinity salicylic acid-binding protein 2 is required for plant innate immunity and has salicylic acid-stimulated lipase activity. Proc Natl Acad Sci USA 100:16101–16106
Lallemand B, Erhardt M, Heitz T, Legrand M (2013) Sporopollenin biosynthetic enzymes interact and constitute a metabolon localized to the endoplasmic reticulum of tapetum cells. Plant Physiol 162:616–625
Lameira J, Bora RP, Chu ZT, Warshel A (2015) Methyltransferases do not work by compression, cratic, or desolvation effects, but by electrostatic preorganization. Prot Struct Funct Bioinform 72:181–204
Laursen T, Borch J, Knudsen C, Bavishi K, Torta F, Martens HJ, Silvestro D, Hatzakis NS, Wenk MR, Dafforn TR, Olsen CE, Motawia MS, Hamberger B, Møller BL, Bassard JE (2016) Characterization of a dynamic metabolon producing the defense compound dhurrin in sorghum. Science 354:890–893
Lee E-J, Facchini PJ (2010) Norcoclaurine synthase is a member of the pathogenesis-related 10/Bet v1 protein family. Plant Cell 22:3489–3503
Lee E-J, Hagel JM, Facchini PJ (2013) Role of the phloem in the biochemistry and ecophysiology of benzylisoquinoline alkaloid metabolism. Front Plant Sci 4:182
Li Y, Smolke CD (2016) Engineering biosynthesis of the anticancer alkaloid noscapine in yeast. Nat Commun 7:12137
Li J, Lee E-J, Chang L, Facchini PJ (2016) Genes encoding norcoclaurine synthase occur as tandem fusions in the Papaveraceae. Sci Rep 6:39256
Lichman BR, Gershater MC, Lamming ED, Pesnot T, Sula A, Keep NH, Hailes HC, Ward JM (2015) ‘Dopamine-first’ mechanism enables the rational engineering of the norcoclaurine synthase aldehyde activity profile. FEBS J 282:1137–1151
Liscombe DK, Facchini PJ (2007) Molecular cloning and characterization of tetrahydroprotoberberine cis-N-methyltransferase, an enzyme involved in alkaloid biosynthesis in opium poppy. J Biol Chem 282:14741–14751
Liscombe DK, MacLeod BP, Loukanina N, Nandi OI, Facchini PJ (2005) Evidence for the monophyletic evolution of benzylisoquinoline alkaloid biosynthesis in angiosperms. Phytochemistry 66:1374–1393
Liscombe DK, Ziegler J, Schmidt J, Ammer C, Facchini PJ (2009) Targeted metabolite and transcript profiling for elucidating enzyme function: isolation of novel N-methyltransferases from three benzylisoquinoline alkaloid-producing species. Plant J 60:729–743
Liscombe DK, Louie GV, Noel JP (2012) Architectures, mechanisms and molecular evolution of natural product methyltransferases. Nat Prod Rep 29:1238
Loeffler S, Deus-Neumann B, Zenk MH (1995) S-adenosyl-l-methionine:(S)-coclaurine-N-methyltransferase from Tinospora cordifolia. Phytochemistry 38:1387–1395
Loenarz C, Schofield CJ (2008) Expanding chemical biology of 2-oxoglutarate oxygenases. Nat Chem Biol 4:152–156
Ma X, Koepke J, Panjikar S, Fritzsch G, Stöckigt J (2005) Crystal structure of vinorine synthase, the first representative of the BAHD superfamily. J Biol Chem 280:13576–13583
Macheroux P, Kappes B, Ealick SE (2011) Flavogenomics—a genomic and structural view of flavin-dependent proteins. FEBS J 278:2625–2634
Mansuy D (1998) The great diversity of reactions catalyzed by cytochromes P450. Comp Biochem Physiol C 121:5–14
Mao G, Seebeck T, Schrenker D, Yu O (2013) CYP709B3, a cytochrome P450 monooxygenase gene involved in salt tolerance in Arabidopsis thaliana. BMC Plant Biol 13:169
Minami H, Kim J-S, Ikezawa N, Takemura T, Katayama T, Kumagai H, Sato F (2008) Microbial production of plant benzylisoquinoline alkaloids. Proc Natl Acad Sci USA 105:7393–7398
Mindrebo JT, Nartey CM, Seto Y, Burkart MD, Noel JP (2016) Unveiling the functional diversity of the alpha/beta hydrolase superfamily in the plant kingdom. Curr Opin Struct Biol 41:233–246
Morimoto S, Suemori K, Moriwaki J, Taura F, Tanaka H, Aso M, Tanaka M, Suemune H, Shimohigashi Y, Shoyama Y (2001) Morphine metabolism in the opium poppy and its possible physiological function. Biochemical characterization of the morphine metabolite, bismorphine. J Biol Chem 276:38179–38184
Morishige T, Tsujita T, Yamada Y, Sato F (2000) Molecular characterization of the S-adenosyl-l-methionine:3′-hydroxy-N-methylcoclaurine 4′-O-methyltransferase involved in isoquinoline alkaloid biosynthesis in Coptis japonica. J Biol Chem 275:23398–23405
Morris JS, Facchini PJ (2016) Isolation and characterization of reticuline N-methyltransferase involved in biosynthesis of the aporphine alkaloid magnoflorine in opium poppy. J Biol Chem 291:23416–23427
Moummou H, Kallberg Y, Tonfack LB, Persson B, van der Rest B (2012) The plant short-chain dehydrogenase (SDR) superfamily: genome-wide inventory and diversification patterns. BMC Plant Biol 12:219
Mudd SH (1973) S-Adenosylmethionine synthetase. Enzymes 8:21–154
Nakajima K, Hashimoto T, Yamada Y (1993) Two tropinone reductases with different stereospecificities are short-chain dehydrogenases evolved from a common ancestor. Proc Natl Acad Sci USA 90:9591–9595
Nelson DR (2009) The cytochrome P450 homepage. Hum Genomics 4:59–65
Nelson DR, Schuler MA, Paquette SM, Werck-Reichhart D, Bak S (2004) Comparative genomics of rice and Arabidopsis. Analysis of 727 cytochrome P450 genes and pseudogenes from a monocot and a dicot. Plant Physiol 135:756–772
Nielsen KA, Tattersall DB, Jones PR, Møller BL (2008) Metabolon formation in dhurrin biosynthesis. Phytochemistry 69:88–98
Ober D, Hartmann T (2000) Phylogenetic origin of a secondary pathway: the case of pyrrolizidine alkaloids. Plant Mol Biol 44:445–450
Oberschall A, Deak M, Torok K, Sass L, Vass I, Kovacs I, Feher A, Dudits D, Horvath GV (2000) A novel aldose/aldehyde reductase protects transgenic plants against lipid peroxidation under chemical and drought stresses. Plant J 24:437–446
Ollis DL, Cheah E, Cygler M, Dijkstra B, Frolow F, Franken SM, Harel M, Remington SJ, Silman I, Schrag J et al (1992) The alpha/beta hydrolase fold. Protein Eng 5:197–211
Oppermann U, Filling C, Hult M, Shafqat N, Wu X, Lindh M, Shafqat J, Nordling E, Kallberg Y, Persson B, Jörnvall H (2003) Short-chain dehydrogenases/reductases (SDR): the 2002 update. Chem Biol Interact 143–144:247–253
Ounaroon A, Decker G, Schmidt J, Lottspeich F, Kutchan TM (2003) (R,S)-Reticuline 7-O-methyltransferase and (R,S)-norcoclaurine 6-O-methyltransferase of Papaver somniferum—cDNA cloning and characterization of methyl transfer enzymes of alkaloid biosynthesis in opium poppy. Plant J 36:808–819
Pauli HH, Kutchan TM (1998) Molecular cloning and functional heterologous expression of two alleles encoding (S)-N-methylcoclaurine 3′-hydroxylase (CYP80B1), a new methyl jasmonate-inducible cytochrome P-450-dependent mono-oxygenase of benzylisoquinoline alkaloid biosynthesis. Plant J 13:793–801
Penning TM (2014) Human aldo–keto reductases and the metabolic activation of polycyclic aromatic hydrocarbons. Chem Res Toxicol 27:1901–1917
Penning TM (2015) The aldo–keto reductases (AKRs): overview. Chem Biol Interact 234:236–246
Persson B, Kallberg Y, Bray JE, Bruford E, Dellaporta SL, Favia AD, Duarte RG, Jornvall H, Kavanagh KL, Kedishvili N, Kisiela M, Maser E, Mindnich R, Orchard S, Penning TM, Thornton JM, Adamski J, Oppermann U (2009) The SDR (short-chain dehydrogenase/reductase and related enzymes) nomenclature initiative. Chem Biol Interact 178:94–98
Pesnot T, Gershater MC, Ward JM, Hailes HC (2011) Phosphate mediated biomimetic synthesis of tetrahydroisoquinoline alkaloids. Chem Commun 47:3242–3244
Petrossian TC, Clarke SG (2011) Uncovering the human methyltransferasome. Mol Cell Proteomics 10(M110):000976
Pienkny S, Brandt W, Schmidt J, Kramell R, Ziegler J (2009) Functional characterization of a novel benzylisoquinoline O-methyltransferase suggests its involvement in papaverine biosynthesis in opium poppy (Papaver somniferum L). Plant J 60:56–67
Pilka ES, Niesen FH, Lee WH, El-Hawari Y, Dunford JE, Kochan G, Wsol V, Martin HJ, Maser E, Oppermann U (2009) Structural basis for substrate specificity in human monomeric carbonyl reductases. PLoS ONE 4:e7113
Priestap HA, Velandia AE, Johnson JV, Barbieri MA (2012) Secondary metabolite uptake by the Aristolochia-feeding papilionoid butter fly Battus polydamas. Biochem Syst Ecol 40:126–137
Robin AY, Giustini C, Graindorge M, Matringe M, Dumas R (2016) Crystal structure of norcoclaurine-6-O-methyltransferase a key rate-limiting step in the synthesis of benzylisoquinoline alkaloids. Plant J 87:641–653
Runguphan W, Glenn WS, O’Connor SE (2012) Redesign of a dioxygenase in morphine biosynthesis. Chem Biol 19:674–678
Schubert HL, Blumenthal RM, Cheng X (2003) Many paths to methyltransfer: a chronicle of convergence. Trends Biochem Sci 28:329–335
Schuler MA, Werck-Reichhart D (2003) Functional genomics of P450s. Annu Rev Plant Biol 54:629–667
Schuler MA, Duan H, Bilgin M, Ali S (2006) Arabidopsis cytochrome P450s through the looking glass: a window on plant biochemistry. Phytochem Rev 5:205–237
Schütz I, Moritz GB, Roos W (2014) Alkaloid metabolism in thrips–Papaveraceae interaction: recognition and mutual response. J Plant Physiol 171:119–126
Simpson PJ, Tantitadapitak C, Reed AM, Mather OC, Bunce CM, White SA, Ride JP (2009) Characterization of two novel aldo-keto reductases from Arabidopsis: expression patterns, broad substrate specificity, and an open active-site structure suggest a role in toxicant metabolism following stress. J Mol Biol 392:465–480
Singleton C, Howard TP, Smirnoff N (2014) Synthetic metabolons for metabolic engineering. J Exp Bot 65:1947–1954
Sinnott M (1997) Comprehensive biological catalysis: a mechanistic reference. Academic Press, London
Sirikantaramas S, Morimoto S, Shoyama Y, Ishikawa Y, Wada Y, Shoyama Y, Taura F (2004) The gene controlling marijuana psychoactivity. Molecular cloning and heterologous expression of 1-tetrahydrocannabinolic acid synthase from Cannabis sativa L. J Biol Chem 279:39767–39774
Srere PA (1985) The metabolon. Trends Biochem Sci 10:109–110
Stafford HA (1974) Activation of 4-hydroxycinnamate hydroxylase in extracts from sorghum. Plant Physiol 54:686–689
St-Pierre B, De Luca V (2000) Origin and diversification of the BAHD superfamily of acyltransferases involved in secondary metabolism. Recent Adv Phytochem 34:285–315
Tang H, Vasconcelos AC, Berkowitz GA (1995) Evidence that plant K+ channel proteins have two different types of subunits. Plant Physiol 109:327–330
Tani C, Tagahara K (1977) Studies on the alkaloids of papeveraceous plants. XXVIII. The biosynthesis of rhoeadine. Yakugaku Zasshi 97:93–102
Taura F, Sirikantaramas S, Shoyama Y, Yoshikai K, Shoyama Y, Morimoto S (2007) Cannabidiolic-acid synthase, the chemotype-determining enzyme in the fiber-type Cannabis sativa. FEBS Lett 581:2929–2934
Thodey K, Galanie S, Smolke CD (2014) A microbial biomanufacturing platform for natural and semisynthetic opioids. Nat Chem Biol 10:837–844
Torres MA, Hoffarth E, Eugenio L, Savtchouk J, Chen X, Morris JS, Facchini PJ, Ng KK (2016) Structural and functional studies of pavine N-methyltransferase from Thalictrum flavum reveal novel insights into substrate recognition and catalytic mechanism. J Biol Chem 291:23403–23415
Unterlinner B, Lenz R, Kutchan TM (1999) Molecular cloning and functional expression of codeinone reductase: the penultimate enzyme in morphine biosynthesis in the opium poppy Papaver somniferum. Plant J 18:465–475
Vekemans D, Proost S, Vanneste K, Coenen H, Viaene T, Ruelens P, Maere S, Van de Peer Y, Geuten K (2012) Gamma paleohexaploidy in the stem lineage of core eudicots: significance for MADS-box gene and species diversification. Mol Biol Evol 29:3793–3806
Vogel M, Lawson M, Sippl W, Conrad U, Roos W (2010) Structure and mechanism of sanguinarine reductase, an enzyme of alkaloid detoxification. J Biol Chem 285:18397–18406
Wallner S, Winkler A, Riedl S, Dully C, Horvath S, Gruber K, Macheroux P (2012) Catalytic and structural role of a conserved active site histidine in berberine bridge enzyme. Biochemistry 51:6139–6147
Wang CF, Fan L, Tian M, Du SS, Deng ZW, Feng JB, Wang YY, Su X (2015) Cytotoxicity of benzophenanthridine alkaloids from the roots of Zanthoxylum nitidum (Roxb.) DC. var. fastuosum How ex Huang. Nat Prod Res 29:1380–1383
Weiss D, Baumert A, Vogel M, Roos W (2006) Sanguinarine reductase, a key enzyme of benzophenanthridine detoxification. Plant Cell Environ 29:291–302
Werck-Reichhart D, Feyereisen R (2000) Cytochromes P450: a success story. Genome Biol 1:reviews3003.1–reviews3003.9
Werck-Reichhart D, Bak S, Paquette S (2002) Cytochromes P450. Arabidopsis Book 1:e0028
Wermuth B (1981) Purification and properties of an NADPH-dependent carbonyl reductase from human brain. Relationship to prostaglandin 9-ketoreductase and xenobiotic ketone reductase. J Biol Chem 256:1206–1213
Wessjohann LA, Keim J, Weigel B, Dippe M (2013) Alkylating enzymes. Curr Opin Chem Biol 17:229–235
Wijekoon CP, Facchini PJ (2012) Systematic knockdown of morphine pathway enzymes in opium poppy using virus-induced gene silencing. Plant J 69:1052–1063
Williams PA, Cosme J, Sridhar V, Johnson EF, McRee DE (2000) Mammalian microsomal cytochrome P450 monooxygenase: structural adaptations for membrane binding and functional diversity. Mol Cell 5:121–131
Wilmouth RC, Turnbull JJ, Welford RW, Clifton IJ, Prescott AG, Schofield CJ (2002) Structure and mechanism of anthocyanidin synthase from Arabidopsis thaliana. Structure 10:93–103
Winkler W, Awe W (1961) On the structure of rhoeadine isomers isolated from Papaver rhoeas. Arch Pharm 294(66):301–306
Winkler A, Hartner F, Kutchan TM, Glieder A, Macheroux P (2006) Biochemical evidence that berberine bridge enzyme belongs to a novel family of flavoproteins containing a bi-covalently attached FAD cofactor. J Biol Chem 281:21276–21285
Winkler A, Lyskowski A, Riedl S, Puhl M, Kutchan TM, Macheroux P, Gruber K (2008) A concerted mechanism for berberine bridge enzyme. Nat Chem Biol 4:739–741
Winkler A, Motz K, Riedl S, Puhl M, Macheroux P, Gruber K (2009) Structural and mechanistic studies reveal the functional role of bicovalent flavinylation in berberine bridge enzyme. J Biol Chem 284:19993–20001
Winzer T, Gazda V, He Z, Kaminski F, Kern M, Larson TR, Li Y, Meade F, Teodor R, Vaistij FE, Walker C, Bowser TA, Graham IA (2012) A Papaver somniferum 10-gene cluster for synthesis of the anticancer alkaloid noscapine. Science 336:1704–1708
Winzer T, Kern M, King AJ, Larson TR, Teodor RI, Donninger SL, Li Y, Dowle AA, Cartwright J, Bates R, Ashford D, Thomas J, Walker C, Bowser TA, Graham IA (2015) Morphinan biosynthesis in opium poppy requires a P450-oxidoreductase fusion protein. Science 349:309–312
Wlodarski T, Kutner J, Towpik J, Knizewski L, Rychlewski L, Kudlicki A, Rowicka M, Dziembowski A, Ginalski K (2011) Comprehensive structural and substrate specificity classification of the Saccharomyces cerevisiae methyltransferome. PLoS ONE 6:e23168
Xu F (2005) Applications of oxidoreductases: recent progress. Ind Biotechnol 1:38–50
Ziegler J, Voigtlander S, Schmidt J, Kramell R, Miersch O, Ammer C, Gesell A, Kutchan TM (2006) Comparative transcript and alkaloid profiling in Papaver species identifies a short chain dehydrogenase/reductase involved in morphine biosynthesis. Plant J 48:177–192
Zubieta C, He X-Z, Dixon RA, Noel JP (2001) Structures of two natural product methyltransferases reveal the basis for substrate specificity in plant O-methyltransferases. Nat Struct Mol Biol 8:271–279
Acknowledgements
This work was supported by financial contributions from a Natural Sciences and Engineering Research Council of Canada Discovery Grant to PJF. MRP is the recipient of a University of Calgary Eyes High Postdoctoral Scholarship. JSM is the recipient of a Natural Sciences and Engineering Reserach Council of Canada Postgraduate Scholarship.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
PJF owns stock in, serves on the Board of Directors of, and is provided compensation by Epimeron Inc. MD, MRP and JSM also receive compensation from Epimeron as contractors.
Rights and permissions
About this article
Cite this article
Dastmalchi, M., Park, M.R., Morris, J.S. et al. Family portraits: the enzymes behind benzylisoquinoline alkaloid diversity. Phytochem Rev 17, 249–277 (2018). https://doi.org/10.1007/s11101-017-9519-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11101-017-9519-z